Fine chemical
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|

In
Fine chemicals are produced in limited volumes (< 1000 tons/year) and at relatively high prices (> $10/kg) according to exacting specifications, mainly by traditional
Since the late 1970s, fine chemicals have become an important part of the chemical industry. Their global total production value of $85 billion is split about 60-40 between in-house production in the life-science industry—the products' main consumers—and companies producing them for sale. The latter pursue both a "supply push" strategy, whereby standard products are developed in-house and offered ubiquitously, and a "demand pull" strategy, whereby products or services determined by the customer are provided exclusively on a "one customer / one supplier" basis. The products are mainly used as building blocks for proprietary products. The hardware of the top tier fine chemical companies has become almost identical. The design, lay-out and equipment of the plants and laboratories have become practically the same globally. Most chemical reactions performed go back to the days of the dyestuff industry. Numerous regulations determine the way labs and plants must be operated, thereby contributing to the uniformity.
History
The term "fine chemicals" was in use as early as 1908.

In subsequent years, the business developed and Lonza was the first fine chemical company entering in a
The growing complexity and potency of new pharmaceuticals and agrochemicals requiring production in multipurpose, instead of dedicated plants and, more recently,[when?] the advent of biopharmaceuticals had a major impact on the demand for fine chemicals and the evolution of the fine chemical industry as a distinct entity. For many years, the life science industry continued considering captive production of the active ingredients of their drugs and agrochemicals as a core competency. Outsourcing was used only in exceptional cases, such as capacity shortfalls, processes requiring hazardous chemistry or new products, where uncertainties existed about the chance of a successful launch.
Products
In terms of molecular structure, one distinguishes first between low-molecular-weight (LMW) and high-molecular-weight (HMW) products. The generally accepted threshold between LMW and HMW is a
Small molecules
As aromatic compounds have been exhausted to a large extent as building blocks for life science products, N-heterocyclic structures prevail nowadays. They are found in many natural products, such as chlorophyll, hemoglobin, and the vitamins
(vitamin B1). In synthetic life science products, N-heterocyclic moieties are widely used in both pharmaceuticals and agrochemicals. Thus,
A vast array of pharmaceuticals and agrochemicals are based on
Big molecules
Big molecules, also called high molecular weight (HMW) molecules, are mostly oligomers or polymers of small molecules or chains of amino acids. Thus, within pharmaceutical sciences,
Apart from pharmaceuticals, peptides are also used for diagnostics and vaccines. The total production volume (excl. Aspartame) of chemically synthesized, pure peptides is about 1500 kilograms and sales approach $500 million on the active pharmaceutical (API) level and $10 billion on the finished drug level, respectively. The bulk of the production of peptide drugs, which comprise also the first generation anti-AIDS drugs, the "...navirs", is outsourced to a few specialized contract manufacturers, such as
Proteins are "very high-molecular-weight" (MW > 100,000) organic compounds, consisting of amino acid sequences linked by peptide bonds. They are essential to the structure and function of all living cells and viruses and are among the most actively studied molecules in biochemistry. They can be made only by advanced biotechnological processes; primarily
Oligonucleotides are a third category of big molecules. They are oligomers of
Technologies
Several key technologies are used for the production of fine chemicals, including
- Chemical synthesis, either from petrochemical starting materials or from natural products extracts
- Biotechnology, for small molecules biocatalysis (enzymatic methods), biosynthesis (fermentation), and, for big molecules, cell culture technology
- Extraction from animals, microorganisms, or plants; isolation and purification, used, for example, for alkaloids, antibacterials(especially penicillins), and steroids
- Hydrolysis of proteins, especially when combined with ion exchange chromatography, used, for instance, for amino acids
Chemical synthesis and biotechnology are most frequently used; sometimes also in combination.
Traditional chemical synthesis
A large toolbox of chemical reactions is available for each step of the synthesis of a fine chemical. The reactions have been developed on laboratory scale by academia over the last two centuries and subsequently adapted to industrial scale, for instance for the manufacture of dyestuffs & pigments. The most comprehensive handbooks describing organic synthetic methods is Methods of Molecular Transformations.
Since the mid-1990s the commercial importance of
For peptides three main types of methods are used, namely chemical synthesis, extraction from natural substances, and biosynthesis. Chemical synthesis is used for smaller peptides made of up to 30–40 amino acids. One distinguishes between "liquid phase" and "solid phase" synthesis. In the latter, reagents are incorporated in a resin that is contained in a reactor or column. The synthesis sequence starts by attaching the first amino acid to the reactive group of the resin and then adding the remaining amino acids one after the other. In order to ascertain a full selectivity, the amino groups have to be protected in advance. Most developmental peptides are synthesized by this method, which lends itself to automation. As the intermediate products resulting from individual synthetic steps cannot be purified, a selectivity of effectively 100% is essential for the synthesis of larger-peptide molecules. Even at a selectivity of 99% per reaction step, the purity will drop to less than 75% for a
With the exception of some stereospecific reactions, particularly biotechnology, mastering these technologies does not represent a distinct competitive advantage. Most reactions can be carried out in standard multipurpose plants. Reaction-specific equipment, such as ozone or phosgene generators, is readily available. The installation generally is not a critical path on the overall project for developing an industrial-scale process of a new molecule.
Whereas the overall demand for outsourced pharmaceutical fine chemicals is expected to increase moderately (see Chapter 8), the estimated annual growth rates for the above-mentioned niche technologies are much higher. Microreactors and the SMB separation technology are expected to grow at a rate of even 50–100% per year. The total size of the accessible market typically does not exceed a few hundred tons per year at best.
Biotechnology
Industrial biotechnology, also called "
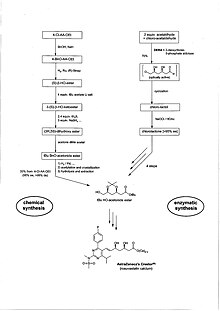
Further examples of modern drugs, where enzymes are used in the synthesis, are
Biosynthesis i.e. the conversion of organic materials into fine chemicals by microorganisms, is used for the production of both small molecules (using enzymes in whole cell systems) and less complex, non-glycosylated big molecules, including peptides and simpler proteins. The technology has been used for 10,000 years to produce food products, like alcoholic beverages, cheese, yogurt, and vinegar. In contrast to biocatalysis, a biosynthetic process does not depend on chemicals as starting materials, but only on cheap natural feedstock, such as glucose, to serve as nutrient for the cells. The enzyme systems triggered in the particular microorganism strain lead to the excretion of the desired product into the medium, or, in the case of HMW peptides and proteins, to the accumulation within so-called inclusion bodies in the cells. The key elements of fermentation development are strain selection and optimization, as well as media and process development. Dedicated plants are used for large-scale industrial production. As the volume productivity is low, the bioreactors, called fermenters, are large, with volumes that can exceed 250 m3. Product isolation was previously based on large-volume extraction of the medium containing the product. Modern isolation and membrane technologies, like reverse osmosis, ultra- and nano-filtration, or affinity chromatography can help to remove salts and by-products, and to concentrate the solution efficiently and in an environmentally friendly manner under mild conditions. The final purification is often achieved by conventional chemical crystallization processes. In contrast to the isolation of small molecules, the isolation and purification of microbial proteins is tedious and often involves a number of expensive large-scale chromatographic operations. Examples of large-volume LMW products made by modern industrial microbial biosynthetic processes are
Cell Cultures
Animal or plant cells, removed from tissues, will continue to grow if cultivated under the appropriate nutrients and conditions. When carried out outside the natural habitat, the process is called cell culture.
| Mammalian cell technology | Chemical technology | ||
|---|---|---|---|
| Worldwide reactor volume | ≈ 3000 m3 (fermenters) | ≈ 80,000 m3 | |
| Investment per m3 reactor volume | ≈ $5 million | ≈ $500,000 | |
| Production per m3 reactor volume and year | several 10 kg | several 1000 kg | |
| Sales per m3 reactor volume and year | ≈ $5 – 10 million | ≈ $250,000 - 500,000 | |
| Value of 1 batch | ≈ $5 million (20,000 liter fermenter) | ≈ $500,000 | |
| Product concentration in reaction mixture | ≈ 2 - 6 (-10) g / Liter | ≈ 100 g / Liter (10%) | |
| Typical reaction time | ≈ 20 days | ≈ 6 hours | |
| Process development time | ≈ 3 years (one step) | 2 – 3 months per step | |
| Capacity expansion projects | many, doubling of actual capacity | few, mainly in Far East | |
| Governing rules | cGMP, BLA [Biological License Application (product specific)] | cGMP, ISO 14000 | |
| Scale-up factor (1st lab process to industrial scale) | ≈ 109 (μg → 1 ton) | ≈ 106 (10 g → 10 tons) | |
| Plant construction time | 4 – 6 years | 2 – 3 years | |
| share of outsourcing | early stage | 55% | 25% of chemical production |
| commercial | 20% | 45% of chemical production | |
The mammalian cell production process, as used for most biopharmaceuticals, is divided into the four main steps: (1) Cultivation, i.e. reproduction of the cells; (2) Fermentation, i.e. the actual production of the protein, typically in 10,000 Liter, or multiples, bioreactors; (3) Purification, i.e. separation of the cells from the culture medium and purification, mostly by chromatography, (4) Formulation, i.e. conversion of the sensitive proteins to a stable form. All steps are fully automated. The low productivity of the animal culture makes the technology expensive and vulnerable to contamination. Actually, as a small number of bacteria would soon outgrow a larger population of animal cells. Its main disadvantages are low volume productivity and the animal provenance. It is conceivable that other technologies, particularly plant cell production, will gain importance in future. Given the fundamental differences between the two process technologies, plants for mammalian cell culture technologies have to be built ex novo.
The pros and cons of an involvement of a fine chemical company in cell culture technology are listed below:
Pros:
- Strong growth of demand: Today, biopharmaceuticals account for about $55–$80 billion, or 15% of the total pharmaceutical market. They are growing by 15% per year, i.e. three times faster than LMW drugs and are expected to pass the $150 billion per year threshold by 2015. Whereas just one out of the world's top ten drugs was a biopharmaceutical in 2001, the number went up to five in 2010 (see table 6) and is expected to increase further to eight by 2016[14] (see Table 2).
| Proprietary name | Generic Name | Company | |
|---|---|---|---|
| Small Molecular Weight (conventional chemical) | |||
| 1 | Crestor | rosuvastatin | AstraZeneca |
| 2 | Advair/Seretide | Salmeterol /fluticasone | GlaxoSmithKline |
| High Molecular Weight (biopharmaceuticals) | |||
| 1 | Humira | adalimumab | AbbVie (Before: Abbott) |
| 2 | Enbrel | etanecept | Amgen |
| 3 | Prolia | denosumab | Amgen |
| 4 | Rituxan | rituximab | Roche/Biogen Idec |
| 5 | Avastatin | bevacizumab | Roche |
| 6 | Herceptin | trastuzumab | Roche |
| 7 | Remicade | infliximab | J&J/Merck & Co. |
| 8 | Lantus | insulin glargine | Sanofi-Aventis |
- The likelihood of developing a new biopharmaceutical successfully is significantly greater than in traditional drug development. 25% of biopharmaceuticals that enter Phase I of the regulatory process eventually are granted approval. The corresponding figure for conventional drugs is less than 6%.
- The traditionally large share of outsourcing.
- Small number of custom manufacturers with industrial-scale manufacturing capabilities in this demanding technology. In the Western hemisphere, primarily Nicholas Piramal of India (through the acquisition of a former Avecia operation) and the joint ventures between AutekBio and Beijing E-Town Harvest International in China and between Biocon in India and Celltrionin South Korea.
- Same customer category: life science, especially the pharmaceutical industry.
- Similar business types: custom manufacturing of proprietary drugs; opportunities for generic versions, called biosimilars.
- Similar regulatory environment: FDA regulations, especially GMP.
- Existing infrastructure (utilities, etc.) can be used.
Cons:
- High entry barriers because of demanding technology. The construction of a large-scale plant for the production of biopharmaceuticals by cell culture fermentation costs around $500 million and takes four to six years.
- As the specifications of the plant and process types for biopharmaceuticals differ substantially from traditional chemical synthesis, they cannot be produced in conventional multipurpose fine chemical plants.
- High financial exposure: (1) high capital intensity ('massive investments are needed at a time when chances of success are still very low' and (2) risk of batch failures (contamination).
- Unlike the biopharmaceutical start-ups, the emerging big biopharmaceutical companies are adopting the same opportunistic outsourcing policy as larger pharmaceutical companies. Thus, are investing heavily in in-house manufacturing capacity. With three plants in the US, two in Japan and one each in Germany and Switzerland, Roche has the largest production capacity.
- New developments in expression systems for mammalian and plant cell technology could reduce capacity requirements substantially. Actually, the titer in large-scale mammalian production, actually 2–3 grams/liter. is expected to double to 5–7 by 2015 and once more to 10 by 2020. Furthermore, the widespread application of 'single-use disposable bioprocessing technology', considered by experts as 'the hottest buzz in town'. It advantageously substitutes for stainless steel production trains, at least for short production campaigns.
- New transgenic animalsand plants, such as tobacco plants possess the potential to become economically and industrially successful.
- Legislation and regulation of biotechnology is not well defined yet and leads to differences in interpretation and other uncertainties. In the US, legislation is not yet in place for biosimilars, the generic counterpart of generics in small molecule pharmaceuticals.
The inherent risks of the mammalian cell technology led several companies to opt out of mammalian cell technology or to substantially reduce their stake. Examples are Cambrex and Dowpharma in the US, Avecia, DSM and Siegfried in Europe and WuXi App Tech in China. In conclusion, biocatalysis should be, or become, part of the technology toolbox of any fine chemical company. Mammalian cell culture fermentation, on the other hand, should be considered only by large fine chemical companies with a full war chest and a long-term strategic orientation.
The industry
Within the chemical universe, the fine chemical industry is positioned between the commodity, their suppliers, and specialty chemical industries, their customers. Depending on the services offered, there are two types of fine chemical companies. The Fine Chemical Companies are active in industrial scale production, both of standard and exclusive products. If the latter prevails, they are referred to as Fine Chemical / Custom Manufacturing Organizations (CMOs). The main assets of the Contract Research Organizations (CROs) are their research laboratories. CRAMS; Contract Research and Manufacturing Organizations[15] are hybrids (see section 4.2).
Fine Chemical / Custom Manufacturing Companies
Fine chemical / Custom Manufacturing companies in the narrower sense are active in process scale up, pilot plant (trial) production, industrial-scale exclusive and non-exclusive manufacture and marketing. Their product portfolios comprise exclusive products, produced by custom manufacturing, as main activity, non-exclusive products, e.g. API-for Generics, and standard products. Characteristics are high asset intensity, batch production in campaigns in multipurpose plants, above-industry-average R&D expenditures and close, multi-level and multi-functional relationships with industrial customers. The industry is very fragmented. 2000 – 3000 fine chemical companies exist globally, extending from small, "garage-type" outfits in China making just one product, all the way to the big, diversified enterprises, resp. units. The main reason for the fragmentation is the lack of economy of scale (see below).
The industry is subject to a high degree of regulation
| Company | Location | Sales 2009 ($ million) | F.C. unit | Sales 2009 ($ million) | Remarks | |
|---|---|---|---|---|---|---|
| 1 | Lonza | Switz. | 2600 | Custom. Manuf. | 1370 | HMW/LMW~55/45 |
| 2 | Boehringer-Ingelheim |
Germany | 18,300 | Fine Chem.1 | 950 | HMW/LMW=84/16 |
| 3 | DSM | The Netherlands | 11,300 | Fine Chem.1 | 850aE | |
| 4 | Sumitomo Chemical | Japan | 17,420 | Fine Chem.1 | 730 | incl. some polymer additives |
| 5 | Merck KGaA |
Germany | 11,200 | Life Science Solutions | 580 | #1 in liquid crystals |
| 6 | Sigma-Aldrich | USA | 2148 | SAFC | 570E | |
| 7 | BASF | Germany | 73,000 | Fine Chem.1 | 5502E | incl. some excipients |
| 8 | CSPC Shijiazhuang Pharmaceutical Group |
China | 1500 | Fine Chem.1 | 550E | API-for-Generics, e.g. HIV / AIDS, sartans |
| 9 | Lanxess | Germany | 7280 | Saltigo | 550E | a.o. agrochemicals |
| 10 | Albemarle | USA | 2005 | Fine Chem.1 | 5002 | a.o. ibuprofen |
| Total Top Ten | ~7200 | |||||
| 1 as per author's definition
2 part of the sales do not derive from fine chemicals, e.g., generics, catalysts, excipients E Author's estimate (non figures published by the company) HMW, high molecular weight, LMW, low molecular weight fine chemicals 11.-20.: Jubilant Organosys. India,800E/470; Dr. Reddy's, India, 1370/370; Evonik-Degussa, Germany, 18,900/350E; Johnson Matthey, UK 12,500/350; Aurobinda, India 665/340; NCPC , North China Pharmaceutical, China, 718/300E; Divi's Laboratories, India, 250/250; Pfizer, US, 50,000/250E; Cambrex, US, 235/235; F.I.S., Italy, 230/230
Σ11-20 ~ 2,900 million; ΣΣ1-20 ~ $10,000 million note: The first number refers to the total sales, the second one to the fine chemical sales. Both are in $ million | ||||||
The combined revenues of the top 20 fine chemical companies amounted to $10 billion in 2009, representing about 30% of the figure for the whole industry. The leading companies are typically divisions of large, diversified chemical companies. In terms of geography, 9 of the top 20 are located in Europe, which is recognized as the cradle of the fine chemical industry. This is e.g. the case for the world's #1 company, Lonza, headquartered in Basel. Switzerland. Custom manufacturing prevails in northern Europe; the manufacture of active substances for generics, in southern Europe. The second largest geographic area is Asia, housing 7 of the top 20. With 4 large companies, the US rank last.
Whereas the European and U.S. pharma industry constitutes the main customer base for most fine chemical companies, some have a significant share of products and services for the agrochemical industry. Examples are Archimica, CABB, Saltigo (all Germany), DSM (The Netherlands) and Hikal, India. Several large pharmaceutical companies market fine chemicals as subsidiary activity to their production for captive use, e.g.
- A Lack of Economy in Size. As most fine chemicals are produced in quantities of not more than a few 10 tons per year in multipurpose plants, there is little or no economy of size. The reactor trains of these plants are similar throughout the industry (see production train of a multipurpose plant). Regardless of the size of the companies, their main constituents, the reaction vessels, have a median size of the 4–6 m3. Various products are made throughout a year in campaigns. Therefore, the unit cost per m3 per hour does practically not vary with the size of the company.
- A Dichotomy between Ownership and Management. The company's shares are listed on stock exchanges, and their performance is scrutinized by the financial community. Postponement of a single important shipment can affect a quarterly result. In the small and mid-sized companies the owners typically are the major shareholders, often members of the same family. Their shares are not traded publicly and fluctuations in their financial performance are more easily coped with.
- Complicated Business Processes. Flexibility and Responsiveness are in jeopardy. Customer complaints, for instance, are difficult to resolve in a straightforward manner.
- A Heterogeneous portfolio of small companies, accumulated over time through M&A activities. The key functions, such as production, R&D, and M&S, are located on different sites, often in different countries.
- A Cohabitation with Other Units.
A comprehensive list of about 1400 fine chemical companies (including traders) can be found in the "event catalogue" of the CPhI exhibition.[19]
The second tier consists of several dozens of midsized companies with sales in the range of $100–$250 million per year. Their portfolios comprise both custom manufacturing and API-for-generics. They include both independents and subsidiaries of major companies. A number of these companies are privately owned and have grown mainly by reinvesting the profits. Examples are Bachem, Switzerland; Dishman, India; F.I.S. and Poli Industria Chimica, Italy; Hikal, India, and Hovione, Portugal. Customers prefer to do business with mid-sized companies, because communications are easier —they typically deal directly with the decision maker— and they can better leverage their purchasing power. The third tier includes thousands of small independents with sales below $100 million per year. Most of them are located in Asia. They often specialize in niche technologies. The minimum economical size of a fine chemical company depends on the availability of infrastructure. If a company is located in an industrial park, where analytical services; utilities, safety, health, and environmental (SHE) services, and warehousing are readily available, there is practically no lower limit. New fine chemical plants have come on-stream mostly in Far East countries over the past few years. Their annual turnover rate rarely exceeds $25 million. All big and medium-size fine chemical companies have cGMP-compliant plants that are suitable for the production of pharmaceutical fine chemicals. With the exception of biopharmaceuticals, which are manufactured by only a few selected fine chemical companies, (see section 3.2.2), the technology toolboxes of all these companies are similar. This means that they can carry out practically all types of chemical reactions. They differentiate on the basis of the breadth and quality of the service offering.
Contract research organizations
Contract research organizations (CROs) provide services to the life science industries along product development. There are more than 2000 CROs operating worldwide, representing revenues of more than $20 billion. One distinguishes between "Product" and "Patient" CROs. Whereas the production sites of CMOs are multipurpose plants, allowing for the production of tens to hundreds of tons of fine chemicals, the work places of patient CROs are the test persons (volunteers) for the clinical trials and those of the product CROs are the laboratory benches. Major customers for CRO services are the large global pharmaceutical companies. Half a dozen companies (
- In North America: Innocentive; Irix Pharmaceuticals, PharmEco, all USA.
- In Europe; ChemDiv, Russia; Clauson-Kaas, Denmark; Enamine Ltd, Ukraine; Girindus, Germany; Nerviano Medical Sciences, Italy; Recipharm, Sweden; Serichim, Italy; Solvias, Switzerland, Netherlands.
- In Asia: BioDuro, Medicilon, Pharmaron; WuXi AppTec, all China; Acoris; Aptuit Laurus; Biocon / Syngene; Chembiotek; Chempartner; ProCitius, all India; NARD Institute, Riken, both Japan.
The business of CROs is usually done through a "pay for service" arrangement. Contrary to manufacturing companies, invoicing of CROs is not based on unit product price, but on full-time equivalents (FTEs), that is, the cost of a scientist working one year on a given customer assignment. Companies offering both contract research and manufacturing services (CRAMS) combine the activities of CROs and CMOs. Their history is either a forward integration of a CRO, which adds industrial scale capabilities or backwards integration of a CMO. As there are only limited synergies (e.g. > 90% of the projects end at the sample preparation stage). It is questionable, though, whether one-stop shops really fulfil a need. Actually, the large fine chemical companies consider the preparation of samples more as marketing tool (and expense ...) rather than a profit contributor.
The offerings of Patient CROs (Clinical CROs) comprise more than 30 tasks addressing the clinical part of pharmaceutical development at the interface between drugs, physicians, hospitals, and patients, such as the clinical development and selection of lead new drug compounds. As
Research and development
The overall emphasis of fine chemical R&D is more on development than on research. The main tasks are (1) designing, respectively duplicating and adapting in case of custom manufacture, and developing laboratory procedures for new products or processes; (2) transferring the processes from the laboratory via pilot plant to the industrial scale (the scale up factor from a 10g sample to a 1-ton batch is 100,000); and (3) to optimize existing processes. At all times during this course of action it has to be ensured that the four critical constraints, namely, economics, timing, safety, ecology and sustainability are observed . R&D expenditures in the fine chemical industry are higher than in the commodities industry. They represent around 5–10% versus 2–5% of sales. On the business side, product innovation must proceed at a more rapid pace, because life cycles of fine chemicals are shorter than those of commodities. Therefore, there is an ongoing need for substitution of obsolete products. On the technical side, the higher complexity of the products and the more stringent regulatory requirements absorb more resources. Many economic and technical parameters have been proposed to enable a meaningful assessment of single projects and project portfolios. Examples are attractiveness, strategic fit, innovation, gross/net present value, expected profits, R&D expenditures, development stage, probability of success, technology fit, potential conflicts with other activities of the company and realization time. Most of these parameters cannot be determined quantitatively, at least during the early phases of a project. The best way to take advantage of a project portfolio is to develop and use it in an iterative way. By comparing the entries at regular intervals, for instance, every 3 months, the directions that the projects take can be visualized. If a negative trend persists with one particular project, the project should be put on the watch list.
Objectives
R&D has to manage the following functions in order to deliver the requested services:
| Task | Laboratory synthesis | Industrial scale process |
|---|---|---|
| Operator | Laboratory chemist | Chemical engineer |
| Economy | Yield | Throughput (kg/m3/hour) |
| Units | G, mL, mol; min. hours | Kg, ton, hours, shift |
| Equipment | Glass flask | Stainless steel, glass lined |
| Process control | Manual | Automatic [reaction vessel] |
| Critical path | Reaction time | Heating / cooling |
| Liquid handling | Pouring | Pumping |
| Liquid / solid sep. | Filtration | Centrifugation |
In case of cGMP fine chemicals also a process validation is required. It consists of the three elements process design, process qualification and continued process verification.
Project management
There are two main sources of new research projects, namely ideas originating from the researchers themselves ("supply push") and those coming from customers ("demand pull"). Ideas for new processes typically originate from researchers, ideas for new products from customers, respectively customer contacts. Particularly in custom manufacturing, "demand pull" prevails industrial reality. The "new product committee" is the body of choice for evaluating new and monitoring ongoing research activities. It has the assignment to evaluate all new product ideas. It decides whether a new product idea should be taken up in research, reassesses a project at regular intervals and, last but not least decides also about the abandonment of a project, once it becomes evident that the objectives cannot be reached. In a typical project the overall responsibility for the economic and technical success lies with the
Markets
Fine chemicals are used as starting materials for
Market size
Fine chemicals account for about 4% of the universe of chemicals. The latter, valued at $2,500 billion, is dominated mainly by oil-, gas-, and mineral-derived commodities (~40%) on one hand and a large variety of specialty chemicals at the interface between industry and the public on the other hand (~55%). The global production value of fine chemicals is estimated at $85 billion, of which about 2/3, or $55 billion are produced captively and $30 billion represent the global revenues of the fine chemical industry. The corresponding figures for the major user, the pharmaceutical industry, are $32 billion and $23 billion, respectively. For a number of reasons, such as the lack of statistical data and the somewhat equivocal definition it is not possible to exactly determine the size of the fine chemical market.
| Size ($ billion) | ||||
| total A.I. | captive | merchant | ||
| Life sciences | Pharmaceuticals | 55 | 32 | 23 |
| Agrochemicals | 15 | 11 | 4 | |
| Various specialty chemicals | 15 | 10 | 5 | |
| Total fine-chemical industry | 85 | 53 | 32 | |
In Table 5, the approximately $85 billion fine chemical market is subdivided into major applications according to their relevance, namely, fine chemicals for pharmaceuticals, agrochemicals and specialty chemicals outside life sciences. Furthermore, a distinction is made between captive (in-house) production and merchant market. Pharmaceutical fine chemicals (PFCs) account for two-thirds of the total. Out of the PFC value of $55 billion, about $23 billion (~40%) are traded, and $32 billion (~60%) are the production value of the pharma industry's in-house production. Within life science products, fine chemicals for agro, and —at a distance— for veterinary drugs follow in importance. The production value for fine chemicals used for specialty chemicals other than pharmaceuticals and agrochemicals is estimated at $15 billion. As the leading specialty chemical companies,
Target markets
Pharmaceuticals
The pharmaceutical industry constitutes the most important customer base for the fine chemical industry (see Table 4). The largest companies are
| Brand | API | Company | sales 2010 ($ bn) | |
|---|---|---|---|---|
| 1 | Lipitor | atorvastatin | Pfizer | 11.8 |
| 2 | Plavix | clopidogrel | Bristol-Myers Squibb Sanofi-Aventis | 9.4 |
| 3 | Remicade* | infliximab | J&J, Merck, Mitsubishi, Tanabe | 8.0 |
| 4 | Advair/ Seretide | salmeterol + fluticasone | Glaxo SmithKline | 8.0 |
| 5 | Enbrel* | etanecerpt | Amgen, Pfizer, Takeda | 7.4 |
| 6 | Avastin* | bevacizumab | Roche | 6.8 |
| 7 | Abilify | aripiprazole | Bristol-Myers Squibb Otsuka | 6.8 |
| 8 | Mabthera/ Rituxan* | rituximab | Roche | 6.7 |
| 9 | Humira* | adalimumab | AbbVie (Before: Abbott) | 6.5 |
| 10 | Diovan & Co-Diovan | valsartan | Novartis | 6.1 |
| Total Top 10 | 77.5 | |||
Sales of the top 20 blockbuster drugs are reported in Table 6. The APIs of 12 of them are "small" (LMW) molecules. Averaging a MW of 477, they have quite complex structures. They typically show three cyclic moieties. 10 of them exhibit at least one N-heterocyclic moiety. Five of the top 10, up from none in 2005, are biopharmaceuticals. The largest-selling non-proprietary drugs are
| Pro's | Con's |
|---|---|
|
|
Several thousand small or virtual pharma companies focus on R&D. albeit on just a few lead compounds. They typically originate mostly from academia. Therefore, their R&D strategy is more focused on the elucidation of the biological roots of diseases rather than developing synthesis methods.
Agrochemicals
Agrochemical companies are the second largest users of fine chemicals. Most products have a "pharmaceutical heritage". As a consequence of an intensive M&A activity (Mergers and Acquisitions) over the past 10–20 years, the industry now is more consolidated than the pharmaceutical industry. The top 10 companies, led by
Generics play a bigger role in the agro than in the pharma industry. They represent some 70% of the global market. China National Chemical Corp, a.k.a. ChemChina Group, is the world's largest supplier of generic farm chemicals. Mahkteshim Agan, Israel, and Cheminova, Denmark follow on the ranks 2 and 3. Apart from these multibillion-dollar companies, there are hundreds of smaller firms with sales of less than $50 million per year, mainly in India and China. The incidence of the cost of the active ingredient is about 33%; i.e., much higher than in drugs. Depending on the climatic conditions affecting crop yields, consumption and prices of agrochemicals are subject to wide fluctuations from year to year, impacting also the suppliers.
The molecular structures of modern agrochemicals are much more complex than in older products, but lower than of their pharma counterparts.
Other specialty chemical industries
Apart from life sciences, specialty chemicals -and therefore also their active ingredients, commodities or fine chemicals, as the case may be- are used ubiquitously, in both industrial applications, such as
| Industry | Sales ($ billion) | Attrac-tiveness | Products |
|---|---|---|---|
| Animal Health | ~ 20 | ♦♦♦ | Typical a.h. products derive from human medicines, e.g. Reconzile, dubbed "puppy Prozac". Paraciticides are the largest product category. Good growth prospects in fish farming. |
| Adhesives & Sealants | ~ 60 | ♦♦ | Uses extend from household, e.g. paper gluing, to high-tech specialty products for assembling electronic parts, automotive and aircraft construction. |
| Biocides | ~ 3 | ♦ | Largest applications are wood conversation and water treatment. A.I.'s mainly commodities |
| Catalysts & Enzymes | ~ 15 | ♦ | Catalysts (automotive, polymers, petroleum processing, chemicals) / enzymes (detergents / technical enzymes, food & feed) = 80 / 20 |
| Dyestuffs & Pigments | ~ 10 | ♦ | Mostly based on large volume aromatic compounds, e.g., letter acids .Asian dyestuff prod, > 106 mtpa. Some niche products, e.g. colour shifting pigments |
| Electronic Chemicals | ~ 30 | ♦♦♦ | Sizeable and growing demand for fine chemicals, e.g. octafluorocyclobutane for etching. for liquid crystals and organic light emitting diodes (OLED). |
| Flavours & Fragrances | ~ 20 | ♦♦ | ~3000 molecules are used, e.g. (-) menthol [20,000 tons],polycyclic musks [10,000 tons], vanilline, linalool, geraniol, heterocyclics, 2-phenylethanol) |
| Food & Feed Additives | 40-50 | ♦♦ | Mainly amino acids (L-lysine [106 tons], L-methionine, ...), vitamins (C [> 105 tons], niacin, riboflavin, ...), artificial sweeteners (aspartame, splenda) and carotenoids |
| Specialty Polymers | NA | ♦♦ | Aerospace: Fluorinated poly ethylene/propylene,[30,000 tons], Polyether ether ketones [PEEK], Polyimides, Precision parts: Aramides [25,000 t], polybenzazoles |
*fine chemicals merchant market size, growth potential
Examples of applications in eight areas, ranging from
Target products and services
Global sales of proprietary drugs are estimated $735 billion in 2010, or almost 90% of the total pharma market. Global sales of generics are about $100 billion, or just over 10% of the total pharma market. Due to the much lower unit price, their market share will be close to 30% on an API volume/volume basis.
Custom manufacturing
The products and services offered by the fine chemical industry fall into two broad categories: (1) "Exclusives", a.k.a. custom manufacturing (CM) and (2) "standard" or "catalogue" products. "Exclusives", provided mostly under contract research or custom manufacturing arrangements, prevail in business with life science companies; "standards" prevail in other target markets. Service-intense custom manufacturing (CM) constitutes the most prominent activity of the fine chemical industry. CM is the antonym of outsourcing. In custom manufacturing, a specialty-chemicals company outsources the process development, pilot plant, and, finally, industrial-scale production of an active ingredient, or a predecessor thereof, to one, or a few, fine chemical companies. The intellectual property of the product, and generally also the manufacturing process, stay with the customer. The customer-supplier relationship is governed by an exclusive supply agreement. At the beginning of cooperation, the customer provides a "tech package", which in its simplest version, includes a laboratory synthesis description and SHE recommendations. In this case, the whole scale up, which comprises a factor of about one million (10 gram → 10 ton quantities), is done by the fine chemical company.
Standard products
Non-exclusives, "standard" or "catalogue products" constitute the second most important outlet for fine chemicals after custom manufacturing. API-for-Generics are the most important sub-category. Because of patent expiries, over 60 of the top 200 drugs alone, representing aggregated sales of over $150 billion, have fallen into the public domain within the past decade. This, along with government-backed incentives, are causing global sales of generics to rapidly increase.[26] Asian companies currently dominate the API-for-Generics business. They have multiple advantages of their low cost basis, their large home markets, and significant previous manufacturing experience compared to western manufacturers in producing for their domestic and other non-regulated markets.
Financials
Investment costs
Investment costs for multipurpose plants are high in comparison with product output. However, they vary considerably, depending on the location, size of equipment and degree of sophistication (e.g., automation, containment, quality of equipment, complexity of infrastructure). An example for a cGMP multipurpose plant built in the US is shown in Table 9. The investment cost of $21 million comprises just the equipment and installation. The building, property and external services are excluded. For comparison purposes, the investment cost per m3 reactor volume is used. In this case, it is $0.9 million. The amount includes the cost of the reaction vessel itself plus an equitable part of the ancillary equipment, like feeding tanks, piping, pumps & process control. If larger or smaller reactors were installed, the unit cost per m3 would decrease or decrease with the exponent 0.5, respectively. Hence, by increasing the equipment size manufacturing costs on a per kilogram (kg−1) basis typically decrease substantially. Also, costs for a plant that is used for the production of non regulated intermediates only would be substantially lower. Pharma companies tend to spend up to ten times more for a plant with the same capacity. In contrast, investment costs in developing countries, particularly in India or China, are considerably lower.
| Equipment / Investment | Numbers |
|---|---|
| Description of main equipment | |
| Production trains
Reactor vessels (volume = 4 m3)
|
2
6
|
| Capital investment | |
Total capital investment
|
$21 million
|
Manufacturing costs
The raw material consumption and the conversion cost are the two elements that establish the manufacturing cost for a particular fine chemical. The former is determined primarily by the unit consumption and the purchasing cost of the materials used; the latter, by the throughput in kilograms per day in a given production bay. A precise calculation of the conversion cost is a demanding task. Different products with widely differing throughputs are produced in campaigns in multipurpose plants, occupying the equipment to different extents. Therefore, both the production capacity and the equipment utilization for a specific fine chemical are difficult to determine. Moreover, cost elements such as labor, capital, utilities, maintenance, waste disposal, and quality control cannot be allocated unambiguously.
An approximative calculation can be done by an experienced process development or pilot plant chemist on the basis of (1) the laboratory synthesis procedure and (2) by breaking down the process into unit operations, the standard costs of which have been determined previously Controlling has to be involved for a more in-depth costing.. The problems it has to address are how to fairly allocate costs for production capacity, which is not used. This can be due to the fact that part of a production bay is idle, because of lack of demand or because, e.g., a reactor is not required for a particular process.
Manufacturing costs usually are reported on a per kilogram product basis. For the purpose of benchmarking (both internal and external), the volume x time / output (VTO), as mentioned above, is a useful aid.
| Cost elements | Details | Share | ||
|---|---|---|---|---|
| raw materials | inclusive solvents | 30 % | ||
| conversion cost | plant specific | utilities and energy | electric power, steam, brine | 4–5 % |
| plant labor | shift and daytime work | 10–15 % | ||
| capital cost | depreciation and interest on capital | 15 % | ||
| plant overhead | QC, maintenance, waste disposal, etc. | 10 % | ||
| research & development | inclusive pilot plant | 8 % | ||
| marketing & sales | inclusive promotion | 5 % | ||
| general overhead | administrative services | 15 % | ||
An indicative cost structure for a fine chemical company is shown in Table 10. Nowadays, a full 7-day/week operation, consisting of four or five shift teams, each working 8h per day, has become the standard. In terms of production costs, this is the most advantageous scheme. Higher salaries for night work are more than offset by better fixed cost absorption. As part of the budgeting process, standard costs for a production campaign of a particular fine chemical are determined on the basis of past experience. The actual results of the campaign are then compared with the standard. The capability of a fine chemical company to make dependable manufacturing cost forecasts is a distinct competitive advantage.
Profitability
The fine chemical industry has undergone several boom and bust phases during its almost 30 years of existence. The biggest boom took place in the late 1990s, when high-dosage, high volume anti-AIDS drugs and COX-2 inhibitors gave a big boost to custom manufacturing. After the end of the "irrational exuberance" in 2000, the industry suffered a first bust in 2003, as a result of capacity expansions, the advent of Asian competitors and a ruinous M&A activity, several billion dollars of shareholder value were destroyed. The most recent –minor- boom is associated with stockpiling of GlaxoSmithKline's
At the end of the "irrational exuberance" at the turn of the millennium and again in 2009 almost half of the industry achieved a return on sales (ROS) of more than 10%, and less than 10% an ROS below 5%. In the worst years, 2003 and 2009, almost half of the companies suffered from an ROS of less than 5%. Whereas during the period under review, 2000–2009. the average EBITDA / sales and EBIT / sales ratios of representative companies, resp. divisions were 15% and 71⁄2%, respectively, in the period 2000–2009, the numbers were 20% and 10–13% in the boom, and 10% and 5% in the bust phases. The factor 2 between the high and low numbers reflects the volatility of the industry's profitability. All in all, the average Western fine-chemical firms have been making a return below the cost of capital, i.e. they are not reinvestment grade.
Outlook
This section possibly contains unsourced reliable published sources. (March 2022) ) |
Two main trends impinge on the industry. On the supply side, biotechnology is rapidly gaining importance.[citation needed] In the synthesis of small molecule fine chemicals, the use of biocatalysts and microbial fermentation enable both a more sustainable and economic production than conventional organic chemistry. In the synthesis of big molecules, such as biopharmaceuticals, it is the method of choice. Biopharmaceuticals are expected to grow 15% per year, three times as fast as small molecule drugs. Five of the top ten drugs were biopharmaceuticals in 2010 (see table 6), and this is expected to grow to eight by 2016 (see table 2).
On the demand side, the main customer base for fine chemicals, the pharmaceutical industry, is faced with slower growth of demand, patent expirations of many lucrative blockbuster drugs and stalling new product launches. In order to restrain these challenges, the leading companies are implementing restructuring programs. They comprise a reduction of in-house chemical manufacturing and plant eliminations. Outsourcing is moving up from a purely opportunistic to a strategic approach. It is difficult to make a judgment, whether the positive or negative effects of these initiatives will prevail. In a worst-case scenario, a condition could develop, whereby even top-tier mid-sized, family-owned[29] fine-chemical companies with state-of-the-art plants and processes could be relegated to producing small quantities of fine chemicals for new life-science products in late stage of development. In agro fine chemicals, the active ingredients become more sophisticated and performing. Therefore, they require multipurpose instead of dedicated plants prevailing in the industry so far. At the same token, outsourcing is gaining ground.[citation needed]
Globalization results in a shift of fine chemical production from the industrialized to developing countries. The latter benefit not only from a "low cost / high skill" advantage, but also from a rapidly rising domestic demand for Western medicine. Despite the mantras of Western industry leaders, the cost advantage of the Asian producers is going to persist.[30] As the pharmemerging countries mainly use generics, their market share continues to grow to the detriment of originator pharmaceuticals and agrochemicals. This is also the case for biosimilars, the generic versions of biopharmaceuticals. As a consequence of the harsh business climate, many Western fine chemical companies or divisions created during the "irrational exuberance" at the end of the 20th century already have exited from the sector.[citation needed] Others will follow suit or will be acquired by private equity firms. Survival strategies include implementation of lean production principles originally developed by the automotive industry and extending the business model to include also contract research at the beginning and active drug formulation towards the end of the added value chain. This latter strategy, however, is not finding unanimous approval by industry experts.[citation needed]
Although the demand for fine chemicals on the merchant market has not grown to the extent originally anticipated, fine chemicals still provide attractive opportunities for well-run companies, which are fostering the critical success factors, namely running fine chemicals as a core business, pursuing niche technologies—primarily biotechnology—and taking advantage of the opportunities offered by the Asian market.[citation needed]
See also
- Chemical industry
- Commercial classification of chemicals
- Commodity chemicals
- Petrochemical
- Speciality chemicals
Bibliography
Pollak, Peter (2011). Fine Chemicals – The Industry and the Business (2nd. rev. ed.). J. Wiley & Sons.
References
- ^ Stahl, A. F. (1908). "XX.- Fine Chemicals, Alkaloids, Essential Oils, & Extracts". The Journal of the Society of Chemical Industry. 27: 956.
- ISBN 9781621983774.
- ISBN 9781621983774.
- ^ E. Reeder; L. H. Sternbach (1968). US 3371085.
- ^ Hughes, Andrew B. (2011). Amino Acids, Peptides and Proteins in Organic Chemistry. Volumes 1-5: John Wiley & Sons, Hoboken.
{{cite book}}: CS1 maint: location (link) - ^ D. Bellus, S. V. Ley, R. Noyori et al. (Series editors) (2010). Science of Synthesis: Houben-Weyl Methods of Molecular Transformations. Thieme Verlag, Stuttgart.
{{cite book}}:|last=has generic name (help)CS1 maint: multiple names: authors list (link) - ^ Examples: Swiss Federal Institute of Technology (ETHZ), Switzerland; Massachusetts Institute of Technology (MIT), USA; Institut für Mikrotechnik (IMM), Germany; University of Washington (WU), USA; Micro-Chemical ProcessTechnology Research Association (MCPT), Japan.
- ^ V. Hessel; A. Renken; J.C. Shouten; J. Yoshida (2009). Micro Process Engineering. Wiley-VCH Verlag, Weiheim.
- ^ Wim Soetaert; Erick J. Vandamme (2010). ), Industrial Biotechnology: Sustainable Growth and Economic Success. J. Wiley & Sons, Hoboken NJ.
- ^ Secretary: Prof. M.P. Walsh. University of Calgary, Calgary, Canada T2N 4N1
- ISBN 978-1-118-00222-3.
- ^ Victor A. Vinci; Sarad R. Parekh (2010). Handbook of Industrial Cell Culture: Mammalian, Microbial, and Plant Cells. Humana Press, New York.
- ^ C. Chassin; P. Pollak (January–February 2004). "Outlook for chemical and biochemical manufacturing, PharmaChem". 1–2: 23–26.
{{cite journal}}: Cite journal requires|journal=(help) - ^ "Expected Top Ten Drugs by 2016". Retrieved 11 December 2011.
- ^ A. Gosh; S. Ray; G. Jain; A. Arora (2011). CRAMS India: Overview & Outlook. ICRA Ltd. Mumbai.
- ^ Samuel L. Tuthill, Norman C. Jamieson, Kirk-Othmer (2000). Encyclopedia of Chemical Technology (4th ed.). John Wiley & Sons, Hoboken NJ. p. 857.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ The U.S. Pharmacopeia 34 (USP 34 –NF29). The U.S Pharmacopeial Convention, Inc., Rockville, MD. 2011.
- ISBN 978-0-470-62767-9.)
{{cite book}}: CS1 maint: location (link - ^ CPhI Worldwide, 25-27 October 2011. Messe Frankfurt, UBM plc., London.
- ^ Stanley H. Nusim (2009). Active Pharmaceutical Ingredients: Development, Manufacturing, and Regulation (2nd ed.). Taylor&Francis Group, Boca Raton FL. pp. 9–91.
- ISBN 978-0-470-62767-9.)
{{cite book}}: CS1 maint: location (link - ^ D. Aboody; B. Lev (2001). R&D Productivity in the Chemical Industry. New York University, Stern School of Business.
- ^ B. Lev (Winter 1999). "Journal of Applied Corporate Finance". 21–35.
{{cite journal}}: Cite journal requires|journal=(help) - ISBN 978-0-470-62767-9.)
{{cite book}}: CS1 maint: location (link - ^ C.D.S. Tomlin (2011). The Pesticide Manual: A World Compendium (15th ed.). BCPC Publications, Alton, Hampshire, UK.
- ^ [1] 29 August 2017
- ISBN 978-0-470-62767-9.)
{{cite book}}: CS1 maint: location (link - ISBN 978-0-470-62767-9.)
{{cite book}}: CS1 maint: location (link - ^ Guy Villax (Nov–Dec 2008). "Family owned businesses". Chimica Oggi / Chemistry Today. 26 (6): 8.
- ^ P. Pollak; A. Badrot; R. Dach; A. Swadi (Nov–Dec 2011). Costs of Asian Fine Chemical Producers close-up to European Levels – Facts or Fiction?. Contract Pharma.
