Flagellum
| Flagellum | |
|---|---|
bacterial flagellum. | |
 | |
| Identifiers | |
| MeSH | D005407 |
| TH | H1.00.01.1.01032 |
| FMA | 67472 |
| Anatomical terminology] | |
A flagellum (.
A microorganism may have from one to many flagella. A
Across the
Eukaryotic flagella and
Types
The three types of flagella are bacterial, archaeal, and eukaryotic.
The flagella in eukaryotes have dynein and microtubules that move with a bending mechanism. Bacteria and archaea do not have dynein or microtubules in their flagella, and they move using a rotary mechanism.[11]
Other differences among these three types are:
- Bacterial flagella are helical filaments, each with a rotary motor at its base which can turn clockwise or counterclockwise.[12][13][14] They provide two of several kinds of bacterial motility.[15][16]
- Archaeal flagella (archaella) are superficially similar to bacterial flagella in that it also has a rotary motor, but are different in many details and considered non-homologous.[17][18][19]
- Eukaryotic flagella—those of animal, plant, and protist cells—are complex cellular projections that lash back and forth. Eukaryotic flagella and motile cilia are identical in structure, but have different lengths, waveforms, and functions. Primary cilia are immotile, and have a structurally different 9+0 axoneme rather than the 9+2 axonemefound in both flagella and motile cilia.
Bacterial
Structure and composition
The bacterial flagellum is made up of
The flagellar filament is the long, helical screw that propels the bacterium when rotated by the motor, through the hook. In most bacteria that have been studied, including the gram-negative
The basal body has several traits in common with some types of
The atomic structure of both bacterial flagella as well as the TTSS
| Part of a series on |
| Microbial and microbot movement |
|---|
 |
| Microswimmers |
| Molecular motors |
Motor
The bacterial flagellum is driven by a rotary engine (
The cylindrical shape of flagella is suited to locomotion of microscopic organisms; these organisms operate at a low Reynolds number, where the viscosity of the surrounding water is much more important than its mass or inertia.[29]
The rotational speed of flagella varies in response to the intensity of the proton-motive force, thereby permitting certain forms of speed control, and also permitting some types of bacteria to attain remarkable speeds in proportion to their size; some achieve roughly 60 cell lengths per second. At such a speed, a bacterium would take about 245 days to cover 1 km; although that may seem slow, the perspective changes when the concept of scale is introduced. In comparison to macroscopic life forms, it is very fast indeed when expressed in terms of number of body lengths per second. A cheetah, for example, only achieves about 25 body lengths per second.[30]
Through use of their flagella, bacteria are able to move rapidly towards attractants and away from repellents, by means of a
Assembly
During flagellar assembly, components of the flagellum pass through the hollow cores of the basal body and the nascent filament. During assembly, protein components are added at the flagellar tip rather than at the base.[32] In vitro, flagellar filaments assemble spontaneously in a solution containing purified flagellin as the sole protein.[33]
Evolution
At least 10 protein components of the bacterial flagellum share homologous proteins with the
Flagella and the intelligent design debate
Some authors have argued that flagella cannot have evolved, assuming that they can only function properly when all proteins are in place. In other words, the flagellar apparatus is "irreducibly complex".[38] However, many proteins can be deleted or mutated and the flagellum still works, though sometimes at reduced efficiency.[39] Moreover, with many proteins unique to some number across species, diversity of bacterial flagella composition was higher than expected.[40] Hence, the flagellar apparatus is clearly very flexible in evolutionary terms and perfectly able to lose or gain protein components. For instance, a number of mutations have been found that increase the motility of E. coli.[41] Additional evidence for the evolution of bacterial flagella includes the existence of vestigial flagella, intermediate forms of flagella and patterns of similarities among flagellar protein sequences, including the observation that almost all of the core flagellar proteins have known homologies with non-flagellar proteins.[34] Furthermore, several processes have been identified as playing important roles in flagellar evolution, including self-assembly of simple repeating subunits, gene duplication with subsequent divergence, recruitment of elements from other systems ('molecular bricolage') and recombination.[42]
Flagellar arrangements
Different species of bacteria have different numbers and arrangements of flagella,[43][44] named using the term tricho, from the Greek trichos meaning hair.[45]
- Monotrichous bacteria such as Vibrio cholerae have a single polar flagellum.[46]
- Amphitrichous bacteria have a single flagellum on each of two opposite ends (e.g., Alcaligenes faecalis)—only one flagellum operates at a time, allowing the bacterium to reverse course rapidly by switching which flagellum is active.
- Lophotrichous bacteria (lopho Greek combining term meaning crest or tuft)[47] have multiple flagella located at the same spot on the bacterial surface such as Helicobacter pylori, which act in concert to drive the bacteria in a single direction. In many cases, the bases of multiple flagella are surrounded by a specialized region of the cell membrane, called the polar organelle.[citation needed]
- Peritrichous bacteria have flagella projecting in all directions (e.g., E. coli).
Counterclockwise rotation of a monotrichous polar flagellum pushes the cell forward with the flagellum trailing behind, much like a corkscrew moving inside cork. Water on the microscopic scale is highly
In certain large forms of Selenomonas, more than 30 individual flagella are organized outside the cell body, helically twining about each other to form a thick structure (easily visible with the light microscope) called a "fascicle".
In some Vibrio spp. (particularly Vibrio parahaemolyticus[51]) and related bacteria such as Aeromonas, two flagellar systems co-exist, using different sets of genes and different ion gradients for energy. The polar flagella are constitutively expressed and provide motility in bulk fluid, while the lateral flagella are expressed when the polar flagella meet too much resistance to turn.[52][53][54][55][56][57] These provide swarming motility on surfaces or in viscous fluids.
Bundling
Bundling is an event that can happen in multi-flagellated cells, bundling the flagella together and causing them to rotate in a coordinated manner.
Flagella are left-handed helices, and when rotated counter-clockwise by their rotors, they can bundle and rotate together. When the rotors reverse direction, thus rotating clockwise, the flagellum unwinds from the bundle. This may cause the cell to stop its forward motion and instead start twitching in place, referred to as tumbling. Tumbling results in a stochastic reorientation of the cell, causing it to change the direction of its forward swimming.
It is not known which stimuli drive the switch between bundling and tumbling, but the motor is highly adaptive to different signals. In the model describing chemotaxis ("movement on purpose") the clockwise rotation of a flagellum is suppressed by chemical compounds favorable to the cell (e.g. food). When moving in a favorable direction, the concentration of such chemical attractants increases and therefore tumbles are continually suppressed, allowing forward motion; likewise, when the cell's direction of motion is unfavorable (e.g., away from a chemical attractant), tumbles are no longer suppressed and occur much more often, with the chance that the cell will be thus reoriented in the correct direction.
Even if all flagella would rotate clockwise, however, they often cannot form a bundle due to geometrical and hydrodynamic reasons.[58][59]
Eukaryotic
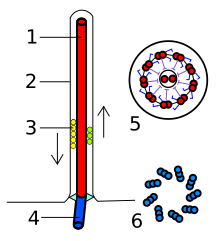


Terminology
Aiming to emphasize the distinction between the bacterial flagella and the eukaryotic cilia and flagella, some authors attempted to replace the name of these two eukaryotic structures with "
Internal structure
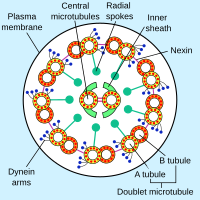
The core of a eukaryotic flagellum, known as the
Besides the axoneme and basal body, relatively constant in morphology, other internal structures of the flagellar apparatus are the transition zone (where the axoneme and basal body meet) and the root system (microtubular or fibrilar structures that extend from the basal bodies into the cytoplasm), more variable and useful as indicators of phylogenetic relationships of eukaryotes. Other structures, more uncommon, are the paraflagellar (or paraxial, paraxonemal) rod, the R fiber, and the S fiber.[65]: 63–84 For surface structures, see below.
Mechanism
Each of the outer 9 doublet microtubules extends a pair of dynein arms (an "inner" and an "outer" arm) to the adjacent microtubule; these produce force through ATP hydrolysis. The flagellar axoneme also contains radial spokes, polypeptide complexes extending from each of the outer nine microtubule doublets towards the central pair, with the "head" of the spoke facing inwards. The radial spoke is thought to be involved in the regulation of flagellar motion, although its exact function and method of action are not yet understood.[66]
Flagella versus cilia

The regular beat patterns of eukaryotic
Although eukaryotic
Intraflagellar transport
Evolution and occurrence
Eukaryotic flagella or cilia, probably an ancestral characteristic,[69] are widespread in almost all groups of eukaryotes, as a relatively perennial condition, or as a flagellated life cycle stage (e.g., zoids, gametes, zoospores, which may be produced continually or not).[70][71][62]
The first situation is found either in specialized cells of multicellular organisms (e.g., the
Flagellated lifecycle stages are found in many groups, e.g., many
Flagella or cilia are completely absent in some groups, probably due to a loss rather than being a primitive condition. The loss of cilia occurred in
Typology
A number of terms related to flagella or cilia are used to characterize eukaryotes.[71][74][65]: 60–63 [75][76] According to surface structures present, flagella may be:
- whiplash flagella (= smooth, acronematic flagella): without hairs, e.g., in Opisthokonta
- hairy flagella (= tinsel, flimmer, pleuronematic flagella): with hairs (= mastigonemes sensu lato), divided in:
- with fine hairs (= non-tubular, or simple hairs): occurs in Pavlovales)
- with stiff hairs (= tubular hairs, retronemes, mastigonemes sensu stricto), divided in:
- bipartite hairs: with two regions. Occurs in Heterokonta
- tripartite (= straminipilous) hairs: with three regions (a base, a tubular shaft, and one or more terminal hairs). Occurs in most Heterokonta
- bipartite hairs: with two regions. Occurs in
- with fine hairs (= non-tubular, or simple hairs): occurs in
- stichonematic flagella: with a single row of hairs
- pantonematic flagella: with two rows of hairs
- acronematic: flagella with a single, terminal mastigoneme or flagellar hair (e.g., bodonids);[77] some authors use the term as synonym of whiplash
- with scales: e.g., Prasinophyceae
- with spines: e.g., some brown algae
- with undulating membrane: e.g., some kinetoplastids, some parabasalids
- with proboscis (trunk-like protrusion of the cell): e.g.,
According to the number of flagella, cells may be: (remembering that some authors use "ciliated" instead of "flagellated")[62][79]
- uniflagellated: e.g., most Opisthokonta
- biflagellated: e.g., all metazoans[73]
- triflagellated: e.g., the gametes of some Foraminifera
- quadriflagellated: e.g., some Prasinophyceae, Collodictyonidae
- octoflagellated: e.g., some Prasinophyceae
- multiflagellated: e.g.,
According to the place of insertion of the flagella:[80]
- opisthokont: cells with flagella inserted posteriorly, e.g., in Haptophyceae, flagella are laterally to terminally inserted, but are directed posteriorly during rapid swimming.[81]
- akrokont: cells with flagella inserted apically
- subakrokont: cells with flagella inserted subapically
- pleurokont: cells with flagella inserted laterally
According to the beating pattern:
- gliding: a flagellum that trails on the substrate[78]
- heterodynamic: flagella with different beating patterns (usually with one flagellum functioning in food capture and the other functioning in gliding, anchorage, propulsion or "steering")[82]
- isodynamic: flagella beating with the same patterns
Other terms related to the flagellar type:
- isokont: cells with flagella of equal length. It was also formerly used to refer to the Chlorophyta
- anisokont: cells with flagella of unequal length, e.g., some Prasinophyceae
- heterokont: term introduced by Luther (1899) to refer to the Heterokonta
- stephanokont: cells with a crown of flagella near its anterior end, e.g., the gametes and spores of Oedogoniales
- akont: cells without flagella. It was also used to refer to taxonomic groups, as Aconta or Akonta: the Rhodophyceae(Christensen, 1962)
Archaeal
The archaellum possessed by some species of Archaea is superficially similar to the bacterial flagellum; in the 1980s, they were thought to be homologous on the basis of gross morphology and behavior.[83] Both flagella and archaella consist of filaments extending outside the cell, and rotate to propel the cell. Archaeal flagella have a unique structure which lacks a central channel. Similar to bacterial type IV pilins, the archaeal proteins (archaellins) are made with class 3 signal peptides and they are processed by a type IV prepilin peptidase-like enzyme. The archaellins are typically modified by the addition of N-linked glycans which are necessary for proper assembly or function.[3]
Discoveries in the 1990s revealed numerous detailed differences between the archaeal and bacterial flagella. These include:
- Bacterial flagella rotation is powered by the
- While bacterial cells often have many flagellar filaments, each of which rotates independently, the archaeal flagellum is composed of a bundle of many filaments that rotates as a single assembly.
- Bacterial flagella grow by the addition of flagellin subunits at the tip; archaeal flagella grow by the addition of subunits to the base.
- Bacterial flagella are thicker than archaella, and the bacterial filament has a large enough hollow "tube" inside that the flagellin subunits can flow up the inside of the filament and get added at the tip; the archaellum is too thin (12-15 nm) to allow this.[85]
- Many components of bacterial flagella share sequence similarity to components of the type IV pili, which are assembled through the action of type II secretion systems (the nomenclature of pili and protein secretion systems is not consistent).[85]
These differences support the theory that the bacterial flagella and archaella are a classic case of biological analogy, or convergent evolution, rather than homology.[86][87][88] Research into the structure of archaella made significant progress beginning in the early 2010s, with the first atomic resolution structure of an archaella protein, the discovery of additional functions of archaella, and the first reports of archaella in Nanoarchaeota and Thaumarchaeota.[89][90]
Fungal
The only
Additional images
-
Multiple flagella in lophotrichous arrangement on surface of Helicobacter pylori
-
Physical model of a bacterial flagellum
See also
References
- PMID 12624192.
- PMID 11743094.
- ^ ISBN 978-1-904455-48-6.
- PMID 17148374.
- PMID 11584108. Archived from the original(abstract page) on 7 February 2009. Retrieved 2 June 2008.
- PMID 15889148.
- PMID 25699024.
- PMID 33525831.
- PMID 6459327.
- PMID 18786541.
- ^ ISBN 9780815344643.)
{{cite book}}: CS1 maint: location missing publisher (link - S2CID 10370084.
- S2CID 4242129.
- S2CID 4173914.
- PMID 5318439.
- PMID 14527279.
- S2CID 30386932.
- S2CID 632440.
- ISBN 978-1-904455-48-6.
- PMID 12730325.
- S2CID 33886010.
- S2CID 7702002.
- S2CID 261963968.
- S2CID 4315167.
- ^ Dean T (2 August 2010). "Inside nature's most efficient motor: the flagellar". Australian Life Scientist.
- ^ Nagata Y (June 2014). "Unlocking the secrets of nature's nanomotor". Nikkei Asian Review.
- PMID 19851449.
- . Retrieved 17 May 2017.
- ISBN 978-0-674-03116-6.
- JSTOR 1376265. Although according to Hunter, Luke; Hamman, Dave (2003). Cheetah. Struik Publishers. pp. 37–38.
the cheetah's fastest recorded speed was 110 km/h (68 mph)
- PMID 21572986.
- PMID 18931786.
- PMID 14222895.
- ^ S2CID 24057949.
- ^ PMID 15001186.
- PMID 12909351.
- PMID 18078471.
- ^ Behe, M. (2007) The Edge of Evolution. Free Press, New York
- PMID 17667950.
- PMID 16936039.
- PMID 28489862.
- PMID 18753783.
- ^ "Bacterial flagella" (PDF). Archived (PDF) from the original on 9 October 2022. Retrieved 29 December 2021.
- PMID 23184985.
- ^ "tricho- prefix". Retrieved 26 March 2022.
- PMID 31119103.
- ^ "Lopho". Retrieved 26 March 2022.
- PMID 19820083.
- PMID 18468917.
- S2CID 19650892.
- PMID 10850984.
- PMID 8759871.
- S2CID 21963003.
- PMID 16978346.
- PMID 3013835.
- PMID 16428388.
- PMID 16385045.
- PMID 14671319.
- PMID 264676.
- PMID 14657097.
- PMID 23195228.
- ^ PMID 23020233.
- S2CID 40755371.
- ISBN 978-1-4822-6822-5.
- ^ ISBN 9780203492598.
- ^ S2CID 18673550.
- ^ ISBN 0-7167-3136-3.
- PMID 15466257.
- PMID 23398214.
- ISBN 9781482268225.
- ^ ISBN 9781139461504.
- PMID 21429931.
- ^ ISBN 9780521479967.
- ISBN 9781444314205.
- ISBN 9780323158237.
- ISBN 9781139469876.
lee tubular hairs.
- ISBN 1891276239.
- ^ PMID 24239731.
- ISBN 9780521428057.
- ^ Sparrow FK (1960). Aquatic phycomycetes (2nd ed.). Ann Arbor: Michigan: University of Michigan Press. p. 15.
- .
- ^ Sleigh MA (1985). "Origin and evolution of flagellar movement". Cell Motil. 5: 137–138.
- S2CID 38405158.[permanent dead link]
- ISBN 9781292235103.)
{{cite book}}: CS1 maint: location missing publisher (link - ^ S2CID 23810797.
- S2CID 34411164.
- PMID 25999922.
- PMID 34219289.
- PMID 35387068.
- PMID 37334237.
- ^ JSTOR 3761366.
Further reading
- Berg HC (January 2000). "Motile Behavior of Bacteria". Physics Today. 53 (1): 24–29. S2CID 178516210.
- Lindemann C (4 April 2008). "Mechanisms of sperm motility". Oakland University. Archived from the original on 16 May 2008. Retrieved 18 May 2008.
- Purcell EM (1977). "Life at Low Reynolds Number" (PDF). American Journal of Physics. 45 (1): 3–11. hdl:2433/226838. Archived from the original(PDF) on 5 June 2011. Retrieved 19 October 2009.
- Matzke NJ (10 November 2003). "Evolution in (Brownian) space: a model for the origin of the bacterial flagellum". talkdesign.org.
External links
![]() This article incorporates text from a publication now in the public domain: Chambers, Ephraim, ed. (1728). Cyclopædia, or an Universal Dictionary of Arts and Sciences (1st ed.). James and John Knapton, et al.
This article incorporates text from a publication now in the public domain: Chambers, Ephraim, ed. (1728). Cyclopædia, or an Universal Dictionary of Arts and Sciences (1st ed.). James and John Knapton, et al. {{cite encyclopedia}}: Missing or empty |title= (help)



