Flavan-3-ol
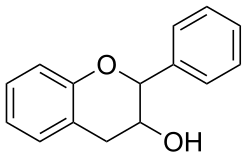
Flavan-3-ols (sometimes referred to as flavanols) are a subgroup of
Chemical structure
The single-molecule (monomer) catechin, or isomer epicatechin (see diagram), adds four hydroxyls to flavan-3-ol, making building blocks for concatenated polymers (proanthocyanidins) and higher order polymers (anthocyanidins).[2]
Flavan-3-ols possess two chiral carbons, meaning four
Catechin gallates are gallic acid esters of the catechins; an example is epigallocatechin gallate, which is commonly the most abundant catechin in tea. Proanthocyanidins and thearubigins are oligomeric flavan-3-ols.
In contrast to many other flavonoids, flavan-3-ols do not generally exist as glycosides in plants.[3]
Biosynthesis of (–)-epicatechin
The flavonoids are products from a cinnamoyl-CoA starter unit, with chain extension using three molecules of malonyl-CoA. Reactions are catalyzed by a type III PKS enzyme. These enzyme do not use ACPSs, but instead employ coenzyme A esters and have a single active site to perform the necessary series of reactions, e.g. chain extension, condensation, and cyclization. Chain extension of 4-hydroxycinnamoyl-CoA with three molecules of malonyl-CoA gives initially a polyketide (Figure 1), which can be folded. These allow Claisen-like reactions to occur, generating aromatic rings.[4][5] Fluorescence-lifetime imaging microscopy (FLIM) can be used to detect flavanols in plant cells.[6]
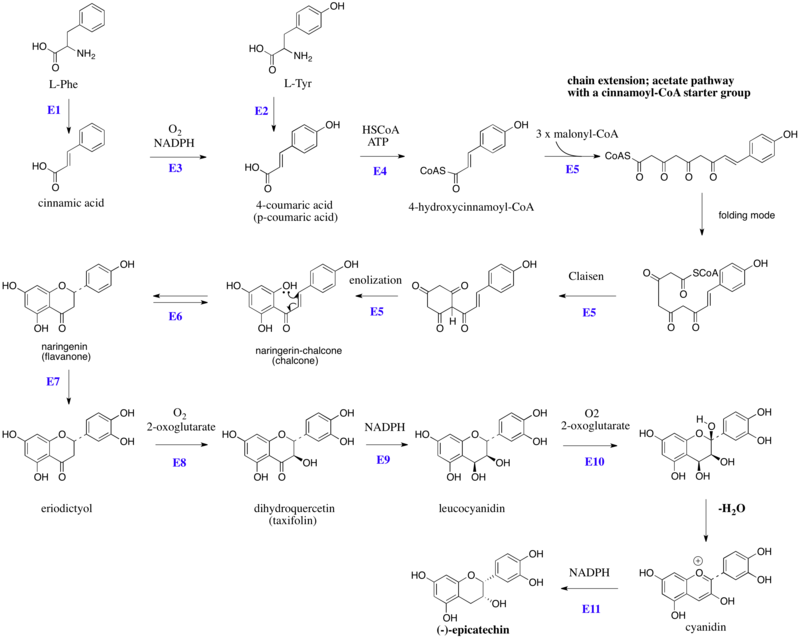
Figure 1:Schematic overview of the flavan-3-ol (–)-epicatechin biosynthesis in plants: Enzymes are indicated in blue, abbreviated as follows: E1,
Aglycones
| Image | Name | Formula | Oligomers |
|---|---|---|---|
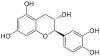 |
Catechin, C, (+)-Catechin | C15H14O6 | Procyanidins |
 |
Epicatechin , EC, (–)-Epicatechin (cis) |
C15H14O6 | Procyanidins |
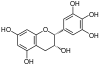 |
Epigallocatechin , EGC |
C15H14O7 | Prodelphinidins |
 |
Epicatechin gallate, ECG | C22H18O10 | |
 |
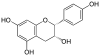
Epigallocatechin gallate, EGCG, (–)-Epigallocatechin gallate |
C22H18O11 | |
 |
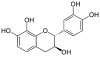
Epiafzelechin |
C15H14O5 | |
| Fisetinidol | C15H14O5 | ||
| Guibourtinidol | C15H14O4 | Proguibourtinidins | |
 |
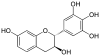
Mesquitol | C15H14O6 | |
 |
Robinetinidol | C15H14O6 | Prorobinetinidins
|
Dietary sources

Flavan-3-ols are abundant in teas derived from the tea plant Camellia sinensis, as well as in some cocoas (made from the seeds of Theobroma cacao), although the content is affected considerably by processing, especially in chocolate.[8][9] Flavan-3-ols are also present in the human diet in fruits, in particular pome fruits, berries, vegetables, and wine.[10] Their content in food is variable and affected by various factors, such as cultivar, processing, and preparation.[11]
Bioavailability and metabolism
The
Possible adverse effects
As catechins in green tea extract can be hepatotoxic, Health Canada and EFSA have advised for caution,[19] recommending intake should not exceed 800 mg per day.[20]
Research
Research has shown that flavan-3-ols may affect
Gallery
-
Schematic representation of the flavan-3-ol (−)-epicatechin metabolism in humans as a function of time post-oral intake. SREM: structurally related (−)-epicatechin metabolites. 5C-RFM: 5-carbon ring fission metabolites. 3/1C-RFM: 3- and 1-carbon-side chain ring fission metabolites. The structures of the most abundant (−)-epicatechin metabolites present in the systemic circulation and in urine are depicted.[17]
-
Flavan-3-ol precursors of the microbial metabolite 5-(3′/4′-dihydroxyphenyl)-γ-valerolactone (gVL). Only compounds with intact (epi)catechin moiety result in the formation of γVL by the intestinal microbiome. ECG, (−)-epicatechin-3-O-gallate; EGCG,Epigallocatechin.[18]
References
- PMID 29070515.
- ^ OCLC 45289285.
- PMID 22794138.
- ISBN 978-0-471-49641-0.
- PMID 11402179.
- S2CID 24094780.
- ^ "Database on polyphenol content in foods, v. 3.6". Phenol Explorer. 2016.
- PMID 10917927.
- PMID 20843086.
- ISBN 978-0-412-11960-6.
- PMID 15113710.
- ^ PMID 22794138.
- PMID 30358831.
- PMID 23864538.
- PMID 11032751.
- PMID 5132890.
- ^ PMID 27363516.
- ^ PMID 29959422.
- ^ Health Canada (12 December 2017). "Summary Safety Review - Green tea extract-containing natural health products - Assessing the potential risk of liver injury (hepatotoxicity)". Health Canada, Government of Canada. Retrieved 2022-05-06.
- PMID 32625874.
- PMID 28439881.
- PMID 31504087.
- ^ "Article 13 (5): Cocoa flavanols; Search filters: Claim status - authorised; search - flavanols". European Commission, EU Register. 31 March 2015. Retrieved 8 September 2022.
- .
- PMID 36190328.
External links
 Media related to Flavan-3-ols at Wikimedia Commons
Media related to Flavan-3-ols at Wikimedia Commons

![Schematic representation of the flavan-3-ol (−)-epicatechin metabolism in humans as a function of time post-oral intake. SREM: structurally related (−)-epicatechin metabolites. 5C-RFM: 5-carbon ring fission metabolites. 3/1C-RFM: 3- and 1-carbon-side chain ring fission metabolites. The structures of the most abundant (−)-epicatechin metabolites present in the systemic circulation and in urine are depicted.[17]](http://upload.wikimedia.org/wikipedia/commons/thumb/2/21/Schematic_representation_of_%28%E2%88%92%29-epicatechin_metabolism_in_humans_as_a_function_of_time_post-oral_intake.jpg/112px-Schematic_representation_of_%28%E2%88%92%29-epicatechin_metabolism_in_humans_as_a_function_of_time_post-oral_intake.jpg)
![Flavan-3-ol precursors of the microbial metabolite 5-(3′/4′-dihydroxyphenyl)-γ-valerolactone (gVL). Only compounds with intact (epi)catechin moiety result in the formation of γVL by the intestinal microbiome. ECG, (−)-epicatechin-3-O-gallate; EGCG, Epigallocatechin gallate; EGC, Epigallocatechin.[18]](http://upload.wikimedia.org/wikipedia/commons/thumb/f/f6/Flavan-3-ol_precursors_of_the_microbial_metabolite_5-%283%E2%80%B2-4%E2%80%B2-dihydroxyphenyl%29-%CE%B3-valerolactone.jpg/120px-Flavan-3-ol_precursors_of_the_microbial_metabolite_5-%283%E2%80%B2-4%E2%80%B2-dihydroxyphenyl%29-%CE%B3-valerolactone.jpg)