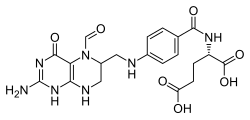
Folinic acid
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | Leucovorin /ˌljuːkoʊˈvɔːrɪn/ |
| Trade names | Many |
| Other names | citrovorum factor, 5-formyltetrahydrofolate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608038 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Intravenous, IM, by mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Dose dependent |
| Protein binding | ~15% |
| Elimination half-life | 6.2 hours |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| Melting point | 245 °C (473 °F) decomp |
| Solubility in water | ~0.3[1] mg/mL (20 °C) |
| |
| |
| | |
Folinic acid, also known as leucovorin, is a medication used to decrease the toxic effects of
Side effects may include trouble sleeping,
Folinic acid was first made in 1945.[5] It is on the World Health Organization's List of Essential Medicines.[6]
Medical use

Folinic acid is given following methotrexate as part of a total chemotherapeutic plan, where it may protect against bone marrow suppression or gastrointestinal mucosa inflammation. No apparent effect is seen on pre-existing methotrexate-induced nephrotoxicity.[7] Folinic acid can be taken as a pill (orally) or injected into a vein (intravenously) or muscle (intramuscularly).[8]
While not specifically an antidote for methotrexate, folinic acid may also be useful in the treatment of acute methotrexate overdose. Different dosing protocols are used, but folinic acid should be redosed until the methotrexate level is less than 5 x 10−8 M.[9]
Additionally, folinic acid is sometimes used to reduce the side effects of methotrexate in rheumatoid arthritis patients. This includes reductions in nausea, abdominal pain, abnormal liver blood tests, and mouth sores.[10]
Folinic acid is also used in combination with the chemotherapy agent
Folinic acid is also sometimes used to prevent toxic effects of high doses of antimicrobial dihydrofolate reductase inhibitors such as trimethoprim and pyrimethamine, although its value for this indication has not been clearly established.[11] It may be prescribed in the treatment of toxoplasmosis retinitis, in combination with the folic acid antagonists pyrimethamine and sulfadiazine.
Folinic acid is also used in the treatment of
In pyridoxine-dependent epilepsy, folinic acid may be used as additional therapy if pyridoxine or pyridoxal phosphate fails to fully control the seizures.[13]
Side effects
Folinic acid should not be administered
In cancer patients, rare hypersensitivity reactions to folinic acid have been described.[15]
Drug interactions
Fluorouracil: Folinic acid may increase the toxicity associated with fluorouracil if the two are administered together. Some adverse effects that have occurred, particularly in elderly patients, include severe enterocolitis, diarrhea, and dehydration.
Mechanism of action
Folinic acid is a 5-formyl derivative of
Although the mechanism is not very clear, the polyglutamylation of methotrexate and dihydrofolate in malignant cells is considered to play an important role in the selective reactivation of dihydrofolate reductase by folinic acid in normal cells.[17]
Folinic acid, therefore, allows for some purine/pyrimidine synthesis to occur in the presence of dihydrofolate reductase inhibition, so some normal DNA replication processes can proceed.
Folinic acid has dextro- and levorotary isomers. Both levoleucovorin (the levorotary isomer) and racemic folinic acid (a mixture of both isomers) have similar efficacy and tolerability.[18] Levoleucovorin was approved by the FDA in 2008.[19]
History
Folinic acid was discovered as a needed growth factor for the bacterium
Names
Folinic acid should be distinguished from
It is generally administered as the
References
- ^ "Safety Data Sheet Folinic Acid (calcium salt)" (PDF). Retrieved 25 January 2018.
- ^ ISBN 9780857111562.
- ^ a b c d e f "Leucovorin Calcium". The American Society of Health-System Pharmacists. Archived from the original on 10 May 2017. Retrieved 8 December 2016.
- ISBN 9789350250747. Archivedfrom the original on 10 May 2017.
- ISBN 9780471899792. Archivedfrom the original on 10 May 2017.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Therapeutic Information Resources Australia (2004). Calcium Folinate (Systemic) in AUSDI: Australian Drug Information for the Health Care Professional. Castle Hill: Therapeutic Information Resources Australia.[page needed]
- PMID 3257913.
- ^ "Leucovorin" (PDF). CCO Formulary. Archived from the original (PDF) on 10 October 2008. Retrieved 7 January 2014.
- PMID 23728635.
- ISBN 9781498747967.
- S2CID 7373721.
- PMID 35053812.
- S2CID 43280375.
- S2CID 25420102.
- S2CID 35513636.
- PMID 2448654.
- PMID 19822510.
- ^ "FDA Approves Levoleucovorin". Drugs.com. 7 May 2008. Archived from the original on 2 July 2009. Retrieved 7 June 2009.
External links
- "Leucovorin". MedlinePlus.
