Fractional distillation
This article needs additional citations for verification. (November 2022) |
Fractional distillation is the
A crude oil distillation unit uses fractional distillation in the process of refining crude oil.
History
The fractional distillation of organic substances played an important role in the 9th-century works attributed to the Islamic alchemist
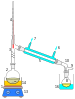
Laboratory setup
Fractional distillation in a laboratory makes use of common

As an example, consider the distillation of a mixture of water and ethanol. Ethanol boils at 78.4 °C (173.1 °F) while water boils at 100 °C (212 °F). So, by heating the mixture, the most volatile component (ethanol) will concentrate to a greater degree in the vapor leaving the liquid. Some mixtures form azeotropes, where the mixture boils at a lower temperature than either component. In this example, a mixture of 96% ethanol and 4% water boils at 78.2 °C (172.8 °F); the mixture is more volatile than pure ethanol. For this reason, ethanol cannot be completely purified by direct fractional distillation of ethanol–water mixtures.
The apparatus is assembled as in the diagram. (The diagram represents a batch apparatus as opposed to a continuous apparatus.) The mixture is put into the round-bottomed flask along with a few
The above explanation reflects the theoretical way fractionation works. Normal laboratory fractionation columns will be simple glass tubes (often vacuum-jacketed, and sometimes internally silvered) filled with a packing, often small glass helices of 4 to 7 millimetres (0.16 to 0.28 in) diameter. Such a column can be calibrated by the distillation of a known mixture system to quantify the column in terms of number of theoretical trays. To improve fractionation the apparatus is set up to return condensate to the column by the use of some sort of reflux splitter (reflux wire, gago, Magnetic swinging bucket, etc.) – a typical careful fractionation would employ a reflux ratio of around 4:1 (4 parts returned condensate to 1 part condensate take off).
In laboratory distillation, several types of condensers are commonly found. The
Alternate set-ups may use a multi-outlet distillation receiver flask (referred to as a "cow" or "pig") to connect three or four receiving
Vacuum distillation systems operate at reduced pressure, thereby lowering the boiling points of the materials. Anti-bumping granules, however, become ineffective at reduced pressures.
Industrial distillation

Fractional distillation is the most common form of separation technology used in
Industrial distillation is typically performed in large, vertical cylindrical columns known as "distillation or fractionation towers" or "distillation columns" with diameters ranging from about 0.65 to 6 meters (2 to 20 ft) and heights ranging from about 6 to 60 meters (20 to 197 ft) or more. The distillation towers have liquid outlets at intervals up the column which allow for the withdrawal of different fractions or products having different
For example, fractional distillation is used in
- have more carbon atoms
- have higher molecular weights
- are less branched-chain alkanes
- are darker in color
- are more viscous
- are more difficult to ignite and to burn

Large-scale industrial towers use reflux to achieve a more complete separation of products.[5] Reflux refers to the portion of the condensed overhead liquid product from a distillation or fractionation tower that is returned to the upper part of the tower as shown in the schematic diagram of a typical, large-scale industrial distillation tower. Inside the tower, the reflux liquid flowing downwards provides the cooling needed to condense the vapors flowing upwards, thereby increasing the effectiveness of the distillation tower. The more reflux is provided for a given number of theoretical plates, the better the tower's separation of lower boiling materials from higher boiling materials. Alternatively, the more reflux provided for a given desired separation, the fewer theoretical plates are required.
Fractional distillation is also used in air separation, producing liquid oxygen, liquid nitrogen, and highly concentrated argon. Distillation of chlorosilanes also enable the production of high-purity silicon for use as a semiconductor.
In industrial uses, sometimes a packing material is used in the column instead of trays, especially when low-pressure drops across the column are required, as when operating under vacuum. This packing material can either be random dumped packing (1–3 in (25–76 mm) wide) such as
Design of industrial distillation columns
Design and operation of a distillation column depends on the feed and desired products. Given a simple, binary component feed, analytical methods such as the McCabe–Thiele method[4][6][7] or the Fenske equation[4] can be used. For a multi-component feed, simulation models are used both for design and operation.
Moreover, the efficiencies of the vapor-liquid contact devices (referred to as plates or trays) used in distillation columns are typically lower than that of a theoretical 100% efficient
Reflux refers to the portion of the condensed overhead product that is returned to the tower. The reflux flowing downwards provides the cooling required for condensing the vapors flowing upwards. The reflux ratio, which is the ratio of the (internal) reflux to the overhead product, is conversely related to the theoretical number of stages required for efficient separation of the distillation products. Fractional distillation towers or columns are designed to achieve the required separation efficiently. The design of fractionation columns is normally made in two steps; a process design, followed by a mechanical design. The purpose of the process design is to calculate the number of required theoretical stages and stream flows including the reflux ratio, heat reflux, and other heat duties. The purpose of the mechanical design, on the other hand, is to select the tower internals, column diameter, and height. In most cases, the mechanical design of fractionation towers is not straightforward. For the efficient selection of tower internals and the accurate calculation of column height and diameter, many factors must be taken into account. Some of the factors involved in design calculations include feed load size and properties and the type of distillation column used.
The two major types of distillation columns used are tray and packing columns. Packing columns are normally used for smaller towers and loads that are corrosive or temperature-sensitive or for vacuum service where pressure drop is important. Tray columns, on the other hand, are used for larger columns with high liquid loads. They first appeared on the scene in the 1820s. In most oil refinery operations, tray columns are mainly used for the separation of petroleum fractions at different stages of oil refining.
In the oil refining industry, the design and operation of fractionation towers is still largely accomplished on an empirical basis. The calculations involved in the design of petroleum fractionation columns require in the usual practice the use of numerable charts, tables, and complex empirical equations. In recent years, however, a considerable amount of work has been done to develop efficient and reliable computer-aided design procedures for fractional distillation.[8]
See also
- Azeotropic distillation
- Batch distillation
- Extractive distillation
- Freeze distillation
- Steam distillation
References
- hdl:2078.1/211340. pp. 106, 111.
- ISBN 9780262082822. p. 44.
- ISBN 0-07-034909-6.
- ^ ISBN 0-07-049479-7.
- ^ "Reflux drum". Alutal. Retrieved 2020-09-18.
- ^ Beychok, Milton (May 1951). "Algebraic Solution of McCabe-Thiele Diagram". Chemical Engineering Progress.
- ISBN 0-471-58626-9.
- ISBN 978-953-51-1719-3.