Fries rearrangement
| Fries rearrangement | |
|---|---|
| Named after | Karl Theophil Fries |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| Organic Chemistry Portal | fries-rearrangement |
| RSC ontology ID | RXNO:0000444 |
The Fries rearrangement, named for the German chemist
It involves migration of an
ortho and para selective and one of the two products can be favoured by changing reaction conditions, such as temperature and solvent
.
Mechanism
Despite many efforts, a definitive reaction mechanism for the Fries rearrangement has not been determined. Evidence for inter- and intramolecular mechanisms have been obtained by crossover experiments with mixed reactants.[citation needed]The reaction progress is not dependent on solvent or substrate. A widely accepted mechanism involves a carbocation intermediate.

In the first reaction step a
ortho product prevails, this can be rationalised as exhibiting classic thermodynamic versus kinetic reaction control as the ortho product can form a more stable bidentate complex with the aluminium.[5] Formation of the ortho product is also favoured in non-polar solvents; as the solvent polarity increases, the ratio of the para product also increases.[6]
Scope
catalysts altogether research into alternative heterogeneous
catalysts is actively pursued.
Limits
In all instances only
Friedel–Crafts acylation
.
Photo-Fries rearrangement
In addition to the ordinary thermal phenyl ester reaction a
photochemical variant is possible. The photo-Fries rearrangement can likewise give [1,3] and [1,5] products,[7][8] which involves a radical reaction mechanism. This reaction is also possible with deactivating substituents on the aromatic group. Because the yields are low this procedure is not used in commercial production. However, photo-Fries rearrangement may occur naturally, for example when a plastic object made of aromatic polycarbonate, polyester or polyurethane, is exposed to the sun (aliphatic carbonyls undergo Norrish reactions, which are somewhat similar). In this case, photolysis of the ester groups would lead to leaching of phthalate from the plastic.[9]
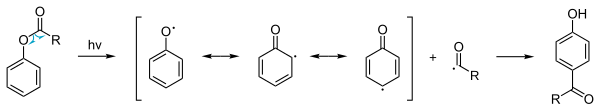
Anionic Fries rearrangement
In the anionic Fries rearrangement
carbamates and carbonates with a strong base results in a rearrangement to give ortho-carbonyl species.[10]
See also
- Friedel–Crafts alkylation-like reactions:
- Duff reaction
References
- .
- .
- ^ March, J. Advanced Organic Chemistry, 3rd Ed.; John Wiley & Sons: Chichester, 1985; S. 499ff.
- ^ Blatt, A. H. Org. React. 1942, 1.
- ISBN 0198556748.
- ISBN 0123694833.
- ISBN 9780470133385.
- ISBN 9780471721550.
- S2CID 206131063.
