Fructose
This article's lead section contains information that is not included elsewhere in the article. (March 2023) |
| |||
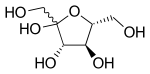
 Haworth projection of β-d-fructofuranose
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
D-arabino-Hex-2-ulose[3]
| |||
| Systematic IUPAC name
(3S,4R,5R)-1,3,4,5,6-Pentahydroxyhexan-2-one | |||
| Other names | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.000.303 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H12O6 | |||
| Molar mass | 180.156 g·mol−1 | ||
| Density | 1.694 g/cm3 | ||
| Melting point | 103 °C (217 °F; 376 K) | ||
| ~4000 g/L (25 °C) | |||
| −102.60×10−6 cm3/mol | |||
| Thermochemistry | |||
Std enthalpy of (ΔcH⦵298)combustion |
675.6 kcal/mol (2,827 kJ/mol) Higher heating value )
| ||
| Pharmacology | |||
| V06DC02 (WHO) | |||
| Hazards | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
15000 mg/kg (intravenous, rabbit)[5] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Fructose (
Fructose was discovered by French chemist
Commercially, fructose is derived from
Excessive consumption of sugars, including fructose, (especially from sugar-sweetened beverages) may contribute to
Etymology
The word "fructose" was coined in 1857 from the Latin for fructus (fruit) and the generic chemical suffix for sugars, -ose.[8][14] It is also called fruit sugar and levulose or laevulose, due to its ability to rotate plane polarised light in a laevorotary fashion (anti-clockwise/to the left) when a beam is shone through it in solution. Likewise, dextrose (an isomer of glucose) is given its name due to its ability to rotate plane polarised light in a dextrorotary fashion (clockwise/to the right).[14]
Chemical properties

Fructose is a 6-carbon polyhydroxyketone.
The distribution of d-fructose tautomers in solution is related to several variables, such as solvent and temperature.[17] d-Fructopyranose and d-fructofuranose distributions in water have been identified multiple times as roughly 70% fructopyranose and 22% fructofuranose.[18]
Reactions
Fructose and fermentation
Fructose may be anaerobically
Fructose and Maillard reaction
Fructose undergoes the
Dehydration
Fructose readily dehydrates to give hydroxymethylfurfural ("HMF", C
6H
6O
3), which can be processed into liquid dimethylfuran (C
6H
8O).
This process, in the future, may become part of a low-cost, carbon-neutral system to produce replacements for petrol and diesel from plants.[22]
Physical and functional properties
Sweetness of fructose
The primary reason that fructose is used commercially in foods and beverages, besides its low cost, is its high relative sweetness. It is the sweetest of all naturally occurring
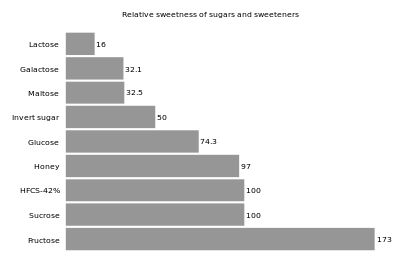
The sweetness of fructose is perceived earlier than that of sucrose or glucose, and the taste sensation reaches a peak (higher than that of sucrose), and diminishes more quickly than that of sucrose. Fructose can also enhance other flavors in the system.[23][25]
Fructose exhibits a sweetness synergy effect when used in combination with other sweeteners. The relative sweetness of fructose blended with sucrose, aspartame, or saccharin is perceived to be greater than the sweetness calculated from individual components.[28][25]
Fructose solubility and crystallization
Fructose has higher water solubility than other sugars, as well as other sugar alcohols. Fructose is, therefore, difficult to crystallize from an aqueous solution.[23] Sugar mixes containing fructose, such as candies, are softer than those containing other sugars because of the greater solubility of fructose.[29]
Fructose hygroscopicity and humectancy
Fructose is quicker to absorb moisture and slower to release it to the environment than sucrose, glucose, or other nutritive sweeteners.
Freezing point
Fructose has a greater effect on freezing point depression than disaccharides or oligosaccharides, which may protect the integrity of cell walls of fruit by reducing ice crystal formation. However, this characteristic may be undesirable in soft-serve or hard-frozen dairy desserts.[23]
Fructose and starch functionality in food systems
Fructose increases starch viscosity more rapidly and achieves a higher final viscosity than sucrose because fructose lowers the temperature required during gelatinizing of starch, causing a greater final viscosity.[30]
Although some artificial sweeteners are not suitable for home baking, many traditional recipes use fructose.[31]
Food sources
This section needs additional citations for verification. (December 2017) |

Natural sources of fructose include fruits, vegetables (including sugar cane), and honey.
The sugar contents of common fruits and vegetables are presented in Table 1. In general, in foods that contain free fructose, the ratio of fructose to glucose is approximately 1:1; that is, foods with fructose usually contain about an equal amount of free glucose. A value that is above 1 indicates a higher proportion of fructose to glucose and below 1 a lower proportion. Some fruits have larger proportions of fructose to glucose compared to others. For example, apples and pears contain more than twice as much free fructose as glucose, while for apricots the proportion is less than half as much fructose as glucose.
Apple and pear juices are of particular interest to
Table 1 also shows the amount of sucrose found in common fruits and vegetables. Sugarcane and sugar beet have a high concentration of sucrose, and are used for commercial preparation of pure sucrose. Extracted cane or beet juice is clarified, removing impurities; and concentrated by removing excess water. The end product is 99.9%-pure sucrose. Sucrose-containing sugars include common white sugar and powdered sugar, as well as brown sugar.[34]
| Food Item | Total carbohydrateA including "dietary fiber" |
Total sugars |
Free fructose |
Free glucose |
Sucrose | Fructose/ glucose ratio |
Sucrose as a % of total sugars |
Free fructose as a % of total sugars |
|---|---|---|---|---|---|---|---|---|
| Fruits | ||||||||
| Apple | 13.8 | 10.4 | 5.9 | 2.4 | 2.1 | 2.0? | 19.9 | 57 |
| Apricot | 11.1 | 9.2 | 0.9 | 2.4 | 5.9 | 0.7? | 63.5 | 10 |
| Banana | 22.8 | 12.2 | 4.9 | 5.0 | 2.4 | 1.0 | 20.0 | 40 |
| Fig, dried | 63.9 | 47.9 | 22.9 | 24.8 | 0.9? | 0.93 | 1.9 | 47.8 |
| Grapes | 18.1 | 15.5 | 8.1 | 7.2 | 0.2 | 1.1 | 1 | 52 |
Navel orange |
12.5 | 8.5 | 2.25 | 2.0 | 4.3 | 1.1 | 50.4 | 26 |
| Peach | 9.5 | 8.4 | 1.5 | 2.0 | 4.8 | 0.9? | 56.7 | 18 |
| Pear | 15.5 | 9.8 | 6.2 | 2.8 | 0.8 | 2.1? | 8.0 | 63 |
| Pineapple | 13.1 | 9.9 | 2.1 | 1.7 | 6.0 | 1.1 | 60.8 | 21 |
| Plum | 11.4 | 9.9 | 3.1 | 5.1 | 1.6 | 0.66 | 16.2 | 31 |
| Vegetables | ||||||||
Beet , Red |
9.6 | 6.8 | 0.1 | 0.1 | 6.5 | 1.0 | 96.2 | 1.5 |
| Carrot | 9.6 | 4.7 | 0.6 | 0.6 | 3.6 | 1.0 | 77 | 13 |
| Red Pepper, Sweet | 6.0 | 4.2 | 2.3 | 1.9 | 0.0 | 1.2 | 0.0 | 55 |
| Onion, Sweet | 7.6 | 5.0 | 2.0 | 2.3 | 0.7 | 0.9 | 14.3 | 40 |
Sweet Potato |
20.1 | 4.2 | 0.7 | 1.0 | 2.5 | 0.9 | 60.3 | 17 |
| Yam | 27.9 | 0.5 | tr | tr | tr | na | tr | |
Sugar Cane |
13–18 | 0.2 – 1.0 | 0.2 – 1.0 | 11–16 | 1.0 | high | 1.5-5.6 | |
Sugar Beet |
17–18 | 0.1 – 0.5 | 0.1 – 0.5 | 16–17 | 1.0 | high | 0.59-2.8 | |
| Grains | ||||||||
| Maize, Sweet | 19.0 | 6.2 | 1.9 | 3.4 | 0.9 | 0.61 | 15.0 | 31 |
- ^A The carbohydrate figure is calculated in FoodData Central and does not always correspond to the sum of the sugars, the starch, and the "dietary fiber".
All data with a unit of g (gram) are based on 100 g of a food item. The fructose/glucose ratio is calculated by dividing the sum of free fructose plus half sucrose by the sum of free glucose plus half sucrose.
Fructose is also found in the manufactured
Carbohydrate content of commercial sweeteners (percent on dry basis)
| Sugar | Fructose | Glucose | Sucrose (Fructose+Glucose) |
Other sugars |
|---|---|---|---|---|
| Granulated sugar | 0 | 0 | 100 | 0 |
| Caramel | 1 | 1 | 97 | 1 |
| HFCS-42 | 42 | 53 | 0 | 5 |
| HFCS-55 | 55 | 41 | 0 | 4 |
| HFCS-90 | 90 | 5 | 0 | 5 |
| Honey | 50 | 44 | 1 | 5 |
| Maple syrup | 1 | 4 | 95 | 0 |
| Molasses | 23 | 21 | 53 | 3 |
| Tapioca Syrup | 55 | 45 | 0 | 0 |
| Corn syrup | 0 | 98 | 0 | 2 |
[34] for HFCS, and USDA for fruits and vegetables and the other refined sugars.[35]
Cane and beet sugars have been used as the major sweetener in food manufacturing for centuries. However, with the development of HFCS, a significant shift occurred in the type of sweetener consumption in certain countries, particularly the United States.[37] Contrary to the popular belief, however, with the increase of HFCS consumption, the total fructose intake relative to the total glucose intake has not dramatically changed. Granulated sugar is 99.9%-pure sucrose, which means that it has equal ratio of fructose to glucose. The most commonly used forms of HFCS, HFCS-42, and HFCS-55, have a roughly equal ratio of fructose to glucose, with minor differences. HFCS has simply replaced sucrose as a sweetener. Therefore, despite the changes in the sweetener consumption, the ratio of glucose to fructose intake has remained relatively constant.[38]

Nutritional information
Providing 368 kcal per 100 grams of dry powder (table), fructose has 95% the caloric value of sucrose by weight.[39][40] Fructose powder is 100% carbohydrates and supplies no other nutrients in significant amount (table).
| Nutritional value per 100 g (3.5 oz) | |
|---|---|
| Energy | 368 kcal (1,540 kJ) |
100 g | |
0 g | |
0 g | |
| Minerals | Quantity %DV† |
| Calcium | 0% 0 mg |
| Iron | 1% 0.1 mg |
| Phosphorus | 0% 0 mg |
| Potassium | 0% 0 mg |
| Sodium | 1% 12 mg |
| †Percentages estimated using US recommendations for adults,[41] except for potassium, which is estimated based on expert recommendation from the National Academies.[42] | |
Fructose digestion and absorption in humans
This section needs additional citations for verification. (May 2020) |


Fructose exists in foods either as a monosaccharide (free fructose) or as a unit of a disaccharide (sucrose). Free fructose is absorbed directly by the intestine. When fructose is consumed in the form of sucrose, it is digested (broken down) and then absorbed as free fructose. As sucrose comes into contact with the membrane of the small intestine, the enzyme
The mechanism of fructose absorption in the small intestine is not completely understood. Some evidence suggests
Capacity and rate of absorption
The absorption capacity for fructose in monosaccharide form ranges from less than 5 g to 50 g (per individual serving) and adapts with changes in dietary fructose intake.
Malabsorption
Several studies have measured the intestinal absorption of fructose using the
Fructose metabolism
All three dietary monosaccharides are transported into the liver by the GLUT2 transporter.[54] Fructose and galactose are phosphorylated in the liver by fructokinase (Km= 0.5 mM) and galactokinase (Km = 0.8 mM), respectively. By contrast, glucose tends to pass through the liver (Km of hepatic glucokinase = 10 mM) and can be metabolised anywhere in the body. Uptake of fructose by the liver is not regulated by insulin. However, insulin is capable of increasing the abundance and functional activity of GLUT5, fructose transporter, in skeletal muscle cells.[55]
Fructolysis
The initial
Metabolism of fructose to DHAP and glyceraldehyde
The first step in the metabolism of fructose is the phosphorylation of fructose to fructose 1-phosphate by fructokinase, thus trapping fructose for metabolism in the liver. Fructose 1-phosphate then undergoes hydrolysis by aldolase B to form DHAP and glyceraldehydes; DHAP can either be isomerized to glyceraldehyde 3-phosphate by triosephosphate isomerase or undergo reduction to glycerol 3-phosphate by glycerol 3-phosphate dehydrogenase. The glyceraldehyde produced may also be converted to glyceraldehyde 3-phosphate by glyceraldehyde kinase or further converted to glycerol 3-phosphate by glycerol 3-phosphate dehydrogenase. The metabolism of fructose at this point yields intermediates in the gluconeogenic pathway leading to glycogen synthesis as well as fatty acid and triglyceride synthesis.
Synthesis of glycogen from DHAP and glyceraldehyde 3-phosphate
The resultant glyceraldehyde formed by aldolase B then undergoes phosphorylation to glyceraldehyde 3-phosphate. Increased concentrations of DHAP and glyceraldehyde 3-phosphate in the liver drive the gluconeogenic pathway toward glucose and subsequent glycogen synthesis.[56] It appears that fructose is a better substrate for glycogen synthesis than glucose and that glycogen replenishment takes precedence over triglyceride formation.[57] Once liver glycogen is replenished, the intermediates of fructose metabolism are primarily directed toward triglyceride synthesis.[58]

Synthesis of triglyceride from DHAP and glyceraldehyde 3-phosphate
Carbons from dietary fructose are found in both the

Potential health effects
In 2022, the European Food Safety Authority stated that there is research evidence that fructose and other added free sugars may be associated with increased risk of several chronic diseases:
Cardiometabolic diseases
When fructose is consumed in excess as a sweetening agent in foods or beverages, it may be associated with increased risk of obesity, diabetes, and cardiovascular disorders that are part of metabolic syndrome.[12]
Compared with sucrose
Fructose was found to increase triglycerides in type-2 but not type-1 diabetes and moderate use of it has previously been considered acceptable as a sweetener for diabetics,[61] possibly because it does not trigger the production of insulin by pancreatic β cells.[62] For a 50 gram reference amount, fructose has a glycemic index of 23, compared with 100 for glucose and 60 for sucrose.[63] Fructose is also 73% sweeter than sucrose at room temperature, allowing diabetics to use less of it per serving. Fructose consumed before a meal may reduce the glycemic response of the meal.[64] Fructose-sweetened food and beverage products cause less of a rise in blood glucose levels than do those manufactured with either sucrose or glucose.[11]
See also
References
- ^ "Fructose". m-w.com. Merriam-Webster. Archived from the original on 19 April 2011. Retrieved 10 December 2014.
- ^ Levulose comes from the Latin word laevus, "left"; levulose is the old word for the most occurring isomer of fructose. D-fructose rotates plane-polarised light to the left, hence the name."Levulose". Archived from the original on 2009-10-08. Retrieved 2010-01-28..
- ^ "2-Carb-10". Archived from the original on 2023-06-18. Retrieved 2023-06-18.
- ^ CRC Handbook of Chemistry and Physics (49th ed.). 1968–69. p. D-186.
- ^ Chambers, Michael. "ChemIDplus – 57-48-7 – BJHIKXHVCXFQLS-UYFOZJQFSA-N – Fructose [USP:JAN] – Similar structures search, synonyms, formulas, resource links, and other chemical information". chem.sis.nlm.nih.gov. US National Institutes of Health. Archived from the original on 10 December 2014. Retrieved 10 December 2014.
- ^ Dubrunfaut (1847). "Sur une propriété analytique des fermentations alcoolique et lactique, et sur leur application à l'étude des sucres" [On an analytic property of alcoholic and lactic fermentations, and on their application to the study of sugars]. Annales de Chimie et de Physique (in French). 21: 169–178. Archived from the original on 2014-06-27. On page 174, Dubrunfaut relates the discovery and properties of fructose.
- from the original on 2021-02-28. Retrieved 2021-02-05.
- ^ a b Miller, William Allen (1857). "Part III. Organic Chemistry". Elements of Chemistry: Theoretical and Practical. London: John W. Parker and son. pp. 52, 57.
- ISBN 978-0-85334-997-6.
- ISBN 9783527303854.
- ^ .
The Panel notes that these values support a significant decrease in post-prandial blood glucose responses when fructose replaces either sucrose or glucose.
- ^ from the original on 26 October 2023. Retrieved 3 October 2022 – via ESFA.
- ^ "Carbohydrates and Health" (PDF). Williams Lea, Norwich, UK: UK Scientific Advisory Committee on Nutrition, Public Health England, TSO. 2015. Archived (PDF) from the original on 19 March 2016. Retrieved 1 April 2016.
- ^ a b "Fructose. Origin and meaning of fructose". Online Etymology Dictionary, Douglas Harper. 2017. Archived from the original on 25 December 2017. Retrieved 24 December 2017.
- ^ "D-Fructose". PubChem, US National Library of Medicine. 20 February 2021. Archived from the original on 12 August 2020. Retrieved 24 February 2021.
- from the original on 28 February 2021. Retrieved 24 February 2021.
- from the original on 24 February 2022. Retrieved 24 February 2021.
- from the original on 28 February 2021. Retrieved 24 February 2021.
- ISBN 978-0-13-021282-5.
- ^ Keusch, P. "Yeast and Sugar- the Chemistry must be right". Archived from the original on December 20, 2010.
- PMID 8213610.
- from the original on 2023-03-26. Retrieved 2020-08-28.
- ^ from the original on 14 April 2016. Retrieved 7 February 2017 – via nutrition.org.
- ^ "Sugar Sweetness". food.oregonstate.edu. Oregon State University. Archived from the original on May 16, 2008. Retrieved 7 February 2017.
- ^ ISBN 978-0471238966.
- ^ Jana, A.H.; Joshi, N.S.S. (November 1994). "Sweeteners for frozen [desserts] success – a review". Australian Journal of Dairy Technology. 49. Archived from the original on 8 February 2017. Retrieved 7 February 2017.
- ISBN 978-0-7514-0150-9.
- ^ a b Nabors, LO (2001). American Sweeteners. pp. 374–375.
- ISBN 978-0-13-021282-5.
- ^ White, DC; Lauer GN (1990). "Predicting gelatinization temperature of starch/sweetener system for cake formulation by differential scanning calorimetry I. Development of a model". Cereal Foods World. 35: 728–731.
- ISBN 978-1580087506.
fructose traditional baking.
- PMID 8213605.
- PMID 8213606.
- ^ a b Kretchmer, N; Hollenbeck CB (1991). Sugars and Sweeteners. CRC Press, Inc.
- ^ a b Use link to FoodData Central (USDA) Archived 2019-10-25 at the Wayback Machine and then search for the particular food, and click on "SR Legacy Foods".
- ^ a b c "High Fructose Corn Syrup: Questions and Answers". US Food and Drug Administration. 5 November 2014. Archived from the original on 25 January 2018. Retrieved 18 December 2017.
- PMID 19064536.
- PMID 10646004.
- ^ "Calories and nutrient composition for fructose, dry powder per 100 g". USDA National Nutrient Database, version SR-28. May 2016. Archived from the original on 2017-02-08.
- ^ "Calories and nutrient composition for sucrose granules per 100 g". USDA National Nutrient Database, version SR-28. May 2016. Archived from the original on 2017-02-08.
- ^ United States Food and Drug Administration (2024). "Daily Value on the Nutrition and Supplement Facts Labels". Retrieved 2024-03-28.
- )
- ^ Stipanuk, Marsha H (2006). Biochemical, Physiological, and Molecular Aspects of Human Nutrition, 2nd Edition. W.B. Saunders, Philadelphia, PA.
- ^ from the original on 16 February 2022. Retrieved 15 February 2022.
- ^ PMID 2065911.
- PMID 7643250.
- PMID 11716754.
- ^ PMID 16183355.
- PMID 6847852.
- PMID 8213606.
- PMID 3781328.
- S2CID 12084142.
- PMID 8317393.
- ISBN 978-1-4160-0209-3.
- S2CID 25952139.
- PMID 3415647.
- PMID 24760443.
- PMID 21165569.
- ^ ISBN 978-1-4160-0209-3.
- ^ ISBN 978-1-4160-0209-3.
- PMID 21050460.
- PMID 20009031.
- ^ "Glycemic index". Glycemic Index Testing and Research, University of Sydney (Australia) Glycemic Index Research Service (SUGiRS). 2 May 2017. Archived from the original on 16 January 2021. Retrieved 23 February 2018.
- PMID 12221216.
External links
 Media related to Fructose at Wikimedia Commons
Media related to Fructose at Wikimedia Commons