Fujiwara–Moritani reaction
In organic chemistry, the Fujiwara–Moritani reaction is a type of cross coupling reaction where an aromatic C-H bond is directly coupled to an olefinic C-H bond, generating a new C-C bond. This reaction is performed in the presence of a transition metal, typically palladium. The reaction was discovered by Yuzo Fujiwara and Ichiro Moritani in 1967.
The need for prefunctionalization of either component is obviated in this reaction, which is desirable because it can shorten syntheses, provide atom economical routes, and enable late stage functionalization of complex molecules. Despite the potential of the Fujiwara-Moritani transformation, it is not often utilized by organic chemists due to the typically harsh reaction conditions, such as acidic, oxidative and high temperature conditions, that most functional groups can not survive.
Mechanism

The mechanism of the Fujiwara–Moritani reaction is not fully understood. The most widely accepted mechanism is as shown in Figure 1.
Industrial example
Latest examples
Even though the original conditions of the Fujiwara–Moritani reaction are not practical, this reaction has significant importance in the sense that it showed the possibility for other transformations that were later developed. Recent advancements have unveiled the mechanism of the Fujiwara–Moritani reaction to some extent which has allowed the development of new systems that enable similar transformations on complex substrates.[10][11][12]
One of the earliest examples of the Fujiwara–Moritani reaction in total synthesis is found in the enantioselective total synthesis of clavicipitic acid by the Murakami group. They used stoichiometric
Fagnou's group showed that direct C-H arylation of an indole is possible using palladium catalysis with a copper oxidant.[14] Although the reaction requires high temperature, acidic solvent, and solvent quantities of the coupling partner, this demonstration of a selective and direct hetero aryl-aryl coupling is notable.
The Yu group developed an Aryl C-H olefination reaction in which aryl carboxylic acids were directly coupled to olefins through the aryl C-H bond. Note: this is not the Fujiwara–Moritani reaction because it is directed by the free acid. The Fujiwara–Moritani reaction is typified by non-directed C–H palladation consistent with regioselectivity of Friedel-Crafts reactions. [15] They also applied their methodology for the total synthesis of (+)-Lithospermic acid.[16] The product yield is as high as 93% despite the complexity of both coupling partners. This is one of the best examples in which C-H olefination simplifies the retro synthesis and demonstrates a convergent synthesis of this complex natural product. 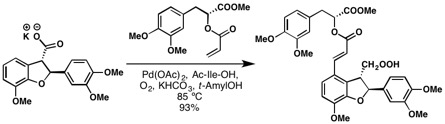
The Lipshutz group dramatically improved the conditions of the Fujiwara–Moritani reaction by developing reaction conditions that utilize water as the solvent and obviate the need for an exogenous acid.[17] Although the substrate scope is limited to p-methoxy aryl species, Lipshutz's report suggested that the Fujiwra-Moritani can be run under milder conditions.
References
- .
- .
- PMID 27462934.
- ISSN 0022-3263.
- PMID 16201772.
- PMID 16433509.
- ^ "OnTheWeb - 触媒学会 - ページビュー". www.shokubai.org. Retrieved 2017-04-21.
- .
- ^ "US Patent for Method for producing biphenyl and its derivatives Patent (Patent # 6,914,152 issued July 5, 2005) - Justia Patents Search". patents.justia.com. Retrieved 2017-04-21.
- PMID 11513570.
- PMID 19557755.
- PMID 21391561.
- ISSN 0022-3263.
- PMID 17880083.
- PMID 19965380.
- PMID 21443224.
- PMID 20364834.




