GSK-3
| Glycogen synthase kinase 3, catalytic domain | |
|---|---|
| Identifiers | |
| Symbol | STKc_GSK3 |
| InterPro | IPR039192 |
| CDD | cd14137 |
Chr. 19 q13.2 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Chr. 3 q13.33 | |||||||
|---|---|---|---|---|---|---|---|
| |||||||
Glycogen synthase kinase 3 (GSK-3) is a
GSK-3 is a serine/threonine protein kinase that
As of 2019[update], GSK-3 is the only type of glycogen synthase kinase named and recognized. The
Mechanism
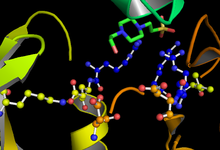
GSK-3 functions by phosphorylating a serine or threonine residue on its target substrate. A positively charged pocket adjacent to the active site binds a "priming" phosphate group attached to a serine or threonine four residues C-terminal of the target phosphorylation site. The active site, at residues 181, 200, 97, and 85, binds the terminal phosphate of ATP and transfers it to the target location on the substrate (see figure 1).[14]
Glycogen synthase
Function
Phosphorylation of a protein by GSK-3 usually inhibits the activity of its downstream target.[16][17][18] GSK-3 is active in a number of central intracellular signaling pathways, including cellular proliferation, migration, glucose regulation, and apoptosis.
GSK-3 was originally discovered in the context of its involvement in regulating
GSK-3 has also been shown to regulate immune and migratory processes. GSK-3 participates in a number of signaling pathways in the innate immune response, including pro-inflammatory cytokine and interleukin production.
GSK-3 is also integrally tied to pathways of cell proliferation and apoptosis. GSK-3 has been shown to phosphorylate
GSK-3 is also involved in nuclear transcriptional activator kappa B (NFκB) signaling pathway, Hedgehog signaling pathway, Notch signaling pathway, and epithelial-mesenchymal transition.[25]
Regulation
Due to its importance across numerous cellular functions, GSK-3 activity is subject to tight regulation.
The speed and efficacy of GSK-3 phosphorylation is regulated by several factors. Phosphorylation of certain GSK-3 residues can increase or decrease its ability to bind substrate. Phosphorylation at tyrosine-216 in GSK-3β or tyrosine-279 in GSK-3α enhances the enzymatic activity of GSK-3, while phosphorylation of autoinhibitory serine-9 in GSK-3β or serine-21 in GSK-3α significantly decreases active site availability (see figure).[22] Further, GSK-3 is unusual among kinases in that it usually requires a "priming kinase" to first phosphorylate a substrate. A phosphorylated serine or threonine residue located four amino acids C-terminal to the target site of phosphorylation allows the substrate to bind a pocket of positive charge formed by arginine and lysine residues.[19][30]
Depending on the pathway in which it is being utilized, GSK-3 may be further regulated by cellular localization or the formation of protein complexes. The activity of GSK-3 is far greater in the nucleus and mitochondria than in the cytosol in cortical neurons,
Insulin indirectly inactivates GSK3 via downstream phosphorylation of the specific serine residues Ser21 and Ser9 in GSK-3 isoforms α and β, respectively, via the
Disease relevance
Due to its involvement in a great number of signaling pathways, GSK-3 has been associated with a host of high-profile diseases. GSK-3 inhibitors are currently being tested for therapeutic effects in
It has now been shown that lithium, which is used as a treatment for bipolar disorder, acts as a mood stabilizer by selectively inhibiting GSK-3. The mechanism through which GSK-3 inhibition stabilizes mood is not known, though it is suspected that the inhibition of GSK-3's ability to promote inflammation contributes to the therapeutic effect.[22] Inhibition of GSK-3 also destabilises Rev-ErbA alpha transcriptional repressor, which has a significant role in the circadian clock.[33] Elements of the circadian clock may be connected with predisposition to bipolar mood disorder.[34]
GSK-3 activity has been associated with both pathological features of Alzheimer's disease, namely the buildup of
In a similar fashion, targeted inhibition of GSK-3 may have therapeutic effects on certain kinds of cancer. Though GSK-3 has been shown to promote
GSK-3 inhibitors have also shown promise in the treatment of T2DM.[19] Though GSK-3 activity under diabetic conditions can differ radically across different tissue types, studies have shown that introducing competitive inhibitors of GSK-3 can increase glucose tolerance in diabetic mice.[22] GSK-3 inhibitors may also have therapeutic effects on hemorrhagic transformation after acute ischemic stroke.[38] GSK-3 can negatively regulate the insulin signaling pathway by inhibiting IRS1 via phosphorylation of serine-332,[20] rendering the insulin receptor incapable of activating IRS1 and further initiating the canonical PI3K/Akt pathway. The role that inhibition of GSK-3 might play across its other signaling roles is not yet entirely understood.
GSK-3 inhibition also mediates an increase in the transcription of the transcription factor Tbet (Tbx21) and an inhibition of the transcription of the inhibitory co-receptor programmed cell death-1 (PD-1) on T-cells.[39] GSK-3 inhibitors increased in vivo CD8(+) OT-I CTL function and the clearance of viral infections by murine gamma-herpesvirus 68 and lymphocytic choriomeningitis clone 13 as well as anti-PD-1 in immunotherapy.
Inhibitors
Glycogen synthase kinase inhibitors are different
Benefits of GSK-3β inhibitors
In diabetes, GSK-3β inhibitors increase insulin sensitivity, glycogen synthesis, and glucose metabolism in skeletal muscles, and reduce obesity by affecting the adipogenesis process.[41] GSK-3β is also over expressed in several types of cancers, like colorectal, ovarian, and prostate cancer.[40] GSK-3β inhibitors also aid in the treatment of Alzheimer's disease,[citation needed] stroke,[38] and mood disorders, including bipolar disorder.[42]
Specific agents
Inhibitors of GSK-3 include:[43][44][45][46]
Metal cations
ATP-competitive
Marine organism-derived
- 6-BIO (IC50=1.5μM)
- Dibromocantharelline (IC50=3μM)
- Hymenialdesine (IC50=10nM)
- Indirubin (IC50=5-50nM)
- Meridianin
Aminopyrimidines
Arylindolemaleimide
Thiazoles
- AR-A014418 (IC50=104nM)
- AZD-1080 (IC50=6.9nM-31nM)
Paullones
IC50=4-80nM:
Aloisines
IC50=0.5-1.5μM:
Non-ATP competitive
Marine organism-derived
- Manzamine A (IC50=1.5μM)
- Palinurine (IC50=4.5μM)
- Tricantine (IC50=7.5μM)
Thiazolidinediones
- TDZD-8 (IC50=2μM)
- NP00111 (IC50=2μM)
- NP031115 (IC50=4μM)
- Tideglusib (IC50=60nM)
Halomethylketones
- HMK-32 (IC50=1.5μM)
Peptides
Unknown Mechanism (small-molecule inhibitors)
Lithium
Lithium which is used in the treatment of bipolar disorder was the first natural GSK-3 inhibitor discovered. It inhibits GSK-3 directly by competition with magnesium ions and indirectly by phosphorylation and auto-regulation of serine. Lithium has been found to have insulin-like effects on glucose metabolism, including stimulation of glycogen synthesis in fat cells, skin, and muscles, increasing glucose uptake, and activation of GS activity. In addition to inhibition of GSK-3, it also inhibits other enzymes involved in the regulation of glucose metabolisms, such as myo-inositol-1-monophosphatase and 1,6 bisphosphatase. Also, it has shown therapeutic benefit in Alzheimer's and other neurodegenerative diseases such as epileptic neurodegeneration.[45]
Naproxen and Cromolyn
Naproxen is a non-steroidal anti-inflammatory drug while cromolyn is an anti-allergic agent which acts as a mast cell stabilizer. Both drugs have demonstrated the anticancer effect in addition to hypoglycemic effect due to inhibition of glycogen synthase kinase-3β (GSK-3β).
To validate the anti-GSK-3β hypothesis of naproxen and cromolyn, docking of the two structures against GSK-3β binding pocket and comparing their fitting with known GSK-3β inhibitor ARA014418 was performed, in addition to measuring the serum glucose, serum insulin, serum C-peptide, weight variation and hepatic glycogen levels for normal and diabetic fasting animal's models to assess their in vitro hypoglycemic effects.[citation needed]
Naproxen and cromolyn were successfully docked into the binding site of GSK-3β (both were fitted into its binding pocket). They exhibited electrostatic, hydrophobic, and hydrogen-bonding interactions with key amino acids within the
Antidiabetic effects of naproxen and cromolyn: In normal animal models, both drugs have showed dose-dependent reduction in blood glucose levels and rise in glycogen levels. In chronic type II diabetic model, glucose levels were also reduced, and glycogen level and insulin levels were elevated in a dose-dependent manner with a reduction in plasma glucose.[citation needed]
Anti-obesity effects of naproxen and cromolyn: Both drugs showed significant anti-obesity effects as they reduce body weight, resistin, and glucose levels in a dose-dependent manner. They were also found to elevate adiponectin, insulin, and C-peptide levels in a dose-dependent manner.[41]
Famotidine
Famotidine is a specific, long-acting
The study of famotidine binding to the enzyme has showed that famotidine can be docked within the binding pocket of GSK-3β making significant interactions with key points within the GSK-3β binding pocket. Strong hydrogen bond interactions with the key amino acids PRO-136 and VAL -135 and potential hydrophobic interaction with LEU-188 were similar to those found in the ligand binding to the enzyme (AR-A014418).[citation needed]
Furthermore, famotidine showed high GSK-3β binding affinity and inhibitory activity due to interactions that stabilize the complex, namely hydrogen bonding of guanidine group in famotidine with the sulfahydryl moiety in CYS-199; and electrostatic interactions between the same guanidine group with the carboxyl group in ASP-200, the hydrogen bond between the terminal NH2 group, the OH of the TYR-143, and the hydrophobic interaction of the sulfur atom in the thioether with ILE-62. In vitro studies showed that famotidine inhibits GSK-3β activity and increases liver glycogen reserves in a dose dependent manner. A fourfold increase in the liver glycogen level with the use of the highest dose of famotidine (4.4 mg/kg) was observed. Also, famotidine has been shown to decrease serum glucose levels 30, and 60 minutes after oral glucose load in healthy individuals.[47]
Curcumin
Curcumin, which Is a constituent of turmeric spice, has flavoring and coloring properties.[48] It has two symmetrical forms: enol (the most abundant forms) and ketone.[49][50]
Curcumin has wide pharmacological activities: anti-inflammatory,[51] anti-microbial,[52] hypoglycemic, anti-oxidant, and wound healing effects.[53] In animal models with Alzheimer disease, it has anti-destructive effect of beta amyloid in the brain,[54] and recently it shows anti-malarial activity.[55]
Curcumin also has chemo preventative and anti-cancer effects.[citation not found], and it has been shown to attenuate oxidative stress and renal dysfunction in diabetic animals with chronic use.[56]
Curcumin's mechanism of action is anti-inflammatory; it inhibits the nuclear transcriptional activator kappa B (NF-KB) that is activated whenever there is inflammatory response.[citation needed]
Among its two forms, experimental and theoretical studies show that the enol form is the favored form due to its intra-molecular hydrogen bonding, and an NMR experiment show that enol form exist in a variety of solvents.[citation needed]
Olanzapine
Antipsychotic medications are increasingly used for schizophrenia, bipolar disorder, anxiety, and other psychiatric conditions[59] Atypical antipsychotics are more commonly used than first generation antipsychotics because they decrease the risk of extrapyramidal symptoms, such as tardive dyskinesia, and have better efficacy.[60]
Olanzapine and atypical antipsychotics induce weight gain through increasing body fat.[61] It also affects glucose metabolism, and several studies shows that it may worsen diabetes.[62]
A recent study shows that olanzapine inhibits GSK3 activity, suggesting olanzapine permits glycogen synthesis. A study of the effect of olanzapine on mouse blood glucose and glycogen levels showed a significant decrease in blood glucose level and elevation of glycogen level in mice, and the IC50% of olanzapine were 91.0 nm, which is considered a potent inhibitor. The study also illustrates that sub-chronic use of olanzapine results in potent inhibition of GSK3.[42]
Pyrimidine derivatives
Pyrimidine analogues are antimetabolites that interfere with nucleic acid synthesis.[63] Some of them have been shown to fit the ATP-binding pocket of GSK-3β to lower blood glucose levels and improve some neuronal diseases.[64]
See also
References
- PMID 14993667.
- ^ PMID 6249596.
- PMID 25435019.
- ^ PMID 15102436.
- ISSN 2673-4087.
- PMID 26941849.
- PMID 11749387.
- PMID 11879773.
- PMID 18088381.
- ^ PMID 28712664. Retrieved 15 December 2023.
- ^ PMID 31553910.
- ^ Glycogen+synthase+kinase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- ^ GSK1, GSK2. NCBI Gene.
- S2CID 17401752.
- PMID 21035757.
- PMID 7803763.
- S2CID 19052833.
- S2CID 12925005.
- ^ PMID 19366350.
- ^ PMID 15574412.
- PMID 11334436.
- ^ PMID 16944320.
- ^ PMID 21095632.
- PMID 22275880.
- ^ PMID 32767962.
- PMID 12048243.
- PMID 11579131.
- ^ PMID 18701488.
- PMID 22675363.
- PMID 12615961.
- S2CID 43633965.
- PMID 29306837.
- S2CID 11240826.
- PMID 24679394.
- PMID 19038340.
- PMID 18806775.
- PMID 22201186.
- ^ PMID 26671619.
- PMID 26885856.
- ^ PMID 16452634.
- ^ S2CID 46597394.
- ^ PMID 18295757.
- PMID 31553649.
- S2CID 34207388.
- ^ PMID 22065134.
- PMID 24931005.
- S2CID 11890710.
- PMID 16413584.
- PMID 19127718.
- PMID 17315954.
- hdl:1807/8668.
- PMID 10552805.
- S2CID 21440334.
- PMID 15590663.
- PMID 18194869.
- S2CID 25193929.
- PMID 12871932.
- ^ S2CID 23137441.
- ^ "Antipsychotics A-Z". Mind.org.uk. 2018.[unreliable medical source?]
- ^ "Antipsychotic Medication for Bipolar Disorder". WebMD.
- S2CID 23589812.
- PMID 20856920.
- ISBN 978-0-444-59499-0.
- PMID 22888461.
External links
- Glycogen Synthase Kinase 3 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
