Gabriel–Colman rearrangement
The Gabriel–Colman rearrangement

Mechanism

The
The displacement of the methoxide is analogous to the displacement seen in the

Furthermore,
Applications
The major application of the Gabriel–Colman rearrangement is in the formation of isoquinolines, due to the relatively high yield of the desired products. Therefore, studies in which either the product or an
Phthalimide derivatives were seen to be inactive, while saccharin derivatives were seen to be fair inhibitors of these enzymes.
In a study
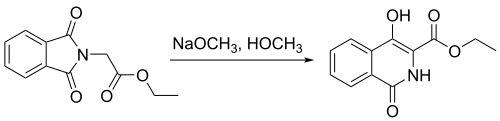
In another study,[10] N-phthalimidoglycine ethyl ester was used to synthesize 4-hydroxyisoquinoline through use of a Gabriel–Colman rearrangement, as shown above. This reaction has shown a percent yield of 91%. The formation of this product was an important step in the study of the synthesis of 4,4′-functionalized 1,1′-biisoquinolines.
See also
- Dieckmann reaction
- Claisen condensation
- Chan rearrangement
References
- .
- .
- PMID 24538878.
- .
- .
- ISBN 978-3-642-01052-1.
- PMID 5116229.
- PMID 7796053.
- .
- .
