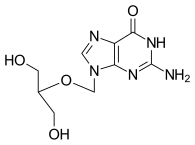
Ganciclovir
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɡænˈsaɪkləvɪər/ |
| Trade names | Cytovene; Cymevene; Vitrasert |
| Other names | gancyclovir; DHPG; 9-(1,3-dihydroxy-2-propoxymethyl)guanine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605011 |
| License data |
|
| Pregnancy category |
|
Intravenous, by mouth, intravitreal | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 5% (oral) |
| Metabolism | guanylate kinase (CMV UL97 gene product) |
| Elimination half-life | 2.5–5 hours |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| Melting point | 250 °C (482 °F) (dec.) |
| |
| |
| (verify) | |
Ganciclovir, sold under the brand name Cytovene among others, is an
Ganciclovir was patented in 1980 and approved for medical use in 1988.[3]
Medical use
Ganciclovir is indicated for:[4]
- Sight-threatening immunocompromisedpeople
- CMV bone marrow transplantrecipients
- Prevention of CMV disease in bone marrow and solid organ transplantrecipients
- Confirmed CMV retinitis in people with AIDS (intravitreal implant)
It is also used for acute CMV colitis in HIV/AIDS and CMV pneumonitis in immunosuppressed patients.[medical citation needed]
Ganciclovir has also been used with some success in treating Human herpesvirus 6 infections.[5]
Ganciclovir has also been found to be an effective treatment for
Veterinary use
Ganciclovir (in gel form) appears to be effective for treating the ophthalmic
Adverse effects
Ganciclovir is commonly associated with a range of serious
Toxicity
Ganciclovir is considered a potential human
Mechanism of action
Ganciclovir (9-[(1,3-dihydroxy-2-propoxy)methyl]guanine) is a potent inhibitor of viruses of the herpes family, including cytomegalovirus (CMV), that are pathogenic for humans and animals. The primary mechanism of ganciclovir action against CMV is inhibition of the replication of viral DNA by ganciclovir-5'-triphosphate (ganciclovir-TP). This inhibition includes a selective and potent inhibition of the viral DNA polymerase. Ganciclovir is metabolized to the triphosphate form by primarily three cellular enzymes: (1) a deoxyguanosine kinase induced by CMV-infected cells; (2) guanylate kinase; and (3) phosphoglycerate kinase. Other nucleotide-metabolizing enzymes may be involved as well. The selective antiviral response associated with ganciclovir treatment is achieved because of the much weaker inhibition of cellular DNA polymerases by ganciclovir-TP. Activity and selectivity are also amplified by the accumulation of ganciclovir-TP in CMV-infected cells.
Pharmacokinetics
Administration
Acute infections are treated in two phases:
- induction phase, 5 mg per kilogram intravenously every 12 hours for 14–21 days, the intravenous dose given as a 1-hour infusion
- maintenance phase, 5 mg per kg intravenously every day
Stable disease is treated with 1000 mg orally three times daily. Similar dosing is used to prevent disease in high-risk patients, such as those infected with human immunodeficiency virus (HIV) or those with organ transplants.
Ganciclovir is also available in slow-release formulations for insertion into the
A topical ophthalmic gel preparation of ganciclovir was recently[when?] approved for the treatment of acute herpes simplex keratitis.[citation needed]
See also
Valganciclovir – the prodrug of ganciclovir
References
- FDA. Retrieved 22 Oct 2023.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ISBN 9783527607495.
- ^ ISBN 0-9757919-2-3.
- PMID 19559728.
- PMID 25879115.
- PMID 35613418.
- ^ "Cymevene® (ganciclovir) Australian Approved Product Information". Pharmaco (Australia) Ltd. Australian Therapeutic Goods Administration. 4 November 2022.
Further reading
- Noble S, Faulds D (July 1998). "Ganciclovir. An update of its use in the prevention of cytomegalovirus infection and disease in transplant recipients". Drugs. 56 (1): 115–146. PMID 9664203.
- Spector SA (1999). "Oral Ganciclovir". Antiviral Chemotherapy 5. Advances in Experimental Medicine and Biology. Vol. 458. pp. 121–7. PMID 10549384.
- Couchoud-Heyer C (July 2007). "WITHDRAWN: Cytomegalovirus prophylaxis with antiviral agents for solid organ transplantation". The Cochrane Database of Systematic Reviews (4): CD001320. PMID 17636667.
