Garnet
| Garnet | ||
|---|---|---|
Crystal class | ||
Specific gravity | 3.1–4.3 | |
| Polish luster | vitreous to subadamantine[2] | |
| Optical properties | Single refractive, often anomalous double refractive[2] | |
| Refractive index | 1.72–1.94 | |
| Birefringence | None | |
| Pleochroism | None | |
| Ultraviolet fluorescence | variable | |
| Other characteristics | variable magnetic attraction | |
| Major varieties | ||
| Pyrope | Mg3Al2Si3O12 | |
| Almandine | Fe3Al2Si3O12 | |
| Spessartine | Mn3Al2Si3O12 | |
| Andradite | Ca3Fe2Si3O12 | |
| Grossular | Ca3Al2Si3O12 | |
| Uvarovite | Ca3Cr2Si3O12 | |
Garnets ( /ˈɡɑːrnɪt/) are a group of silicate minerals that have been used since the Bronze Age as gemstones and abrasives.
All species of garnets possess similar physical properties and crystal forms, but differ in
Etymology
The word garnet comes from the 14th-century
Physical properties
Properties

Garnet species are found in every colour, with reddish shades most common. Blue garnets are the rarest and were first reported in the 1990s.[7][8][9][10]
Garnet species' light transmission properties can range from the gemstone-quality transparent specimens to the opaque varieties used for industrial purposes as abrasives. The mineral's
Crystal structure
Garnets are
-
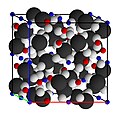
Crystal structure of pyrope garnet. White spheres are oxygen; black, silicon; blue, aluminium; and red, magnesium.
-
Same view, with ion sizes reduced to better show all ions
-
Silicon ion size exaggerated to emphasize silica tetrahedra
Hardness
Because the chemical composition of garnet varies, the atomic bonds in some species are stronger than in others. As a result, this mineral group shows a range of hardness on the
Magnetics used in garnet series identification
For gem identification purposes, a pick-up response to a strong neodymium magnet separates garnet from all other natural transparent gemstones commonly used in the jewelry trade. Magnetic susceptibility measurements in conjunction with refractive index can be used to distinguish garnet species and varieties, and determine the composition of garnets in terms of percentages of end-member species within an individual gem.[15]
Garnet group end member species
Pyralspite garnets – aluminium in Y site
- Almandine: Fe3Al2(SiO4)3
- Pyrope: Mg3Al2(SiO4)3
- Spessartine: Mn3Al2(SiO4)3
Almandine

Almandine, sometimes incorrectly called almandite, is the modern gem known as
Pyrope
Pyrope (from the Greek pyrōpós meaning "firelike")[3] is red in color and chemically an aluminium silicate with the formula Mg3Al2(SiO4)3, though the magnesium can be replaced in part by calcium and ferrous iron. The color of pyrope varies from deep red to black. Pyrope and spessartine gemstones have been recovered from the Sloan diamondiferous kimberlites in Colorado, from the Bishop Conglomerate and in a Tertiary age lamprophyre at Cedar Mountain in Wyoming.[20]
A variety of pyrope from Macon County, North Carolina is a violet-red shade and has been called rhodolite, Greek for "rose". In chemical composition it may be considered as essentially an isomorphous mixture of pyrope and almandine, in the proportion of two parts pyrope to one part almandine.[21] Pyrope has tradenames some of which are misnomers; Cape ruby, Arizona ruby, California ruby, Rocky Mountain ruby, and Bohemian ruby from the Czech Republic.[16]
Pyrope is an indicator mineral for high-pressure rocks. Mantle-derived rocks (peridotites and eclogites) commonly contain a pyrope variety.[22]
Spessartine

Spessartine or spessartite is manganese aluminium garnet, Mn3Al2(SiO4)3. Its name is derived from Spessart in Bavaria.[3] It occurs most often in skarns,[3] granite pegmatite and allied rock types,[23] and in certain low grade metamorphic phyllites. Spessartine of an orange-yellow is found in Madagascar.[24] Violet-red spessartines are found in rhyolites in Colorado[21] and Maine.[citation needed]
Pyrope–spessartine (blue garnet or color-change garnet)
Blue pyrope–spessartine garnets were discovered in the late 1990s in Bekily, Madagascar. This type has also been found in parts of the United States, Russia, Kenya, Tanzania, and Turkey. It changes color from blue-green to purple depending on the color temperature of viewing light, as a result of the relatively high amounts of vanadium (about 1 wt.% V2O3).[9]
Other varieties of color-changing garnets exist. In daylight, their color ranges from shades of green, beige, brown, gray, and blue, but in incandescent light, they appear a reddish or purplish/pink color.[25]
This is the rarest type of garnet. Because of its color-changing quality, this kind of garnet resembles alexandrite.[26]
Ugrandite group – calcium in X site
Andradite
Andradite is a calcium-iron garnet, Ca3Fe2(SiO4)3, is of variable composition and may be red, yellow, brown, green or black.
Grossular


Grossular is a calcium-aluminium garnet with the formula Ca3Al2(SiO4)3, though the calcium may in part be replaced by ferrous iron and the aluminium by ferric iron. The name grossular is derived from the
Grossular garnet from Kenya and Tanzania has been called tsavorite. Tsavorite was first described in the 1960s in the Tsavo area of Kenya, from which the gem takes its name.[32][33]
Uvarovite
Uvarovite is a calcium chromium garnet with the formula Ca3Cr2(SiO4)3. This is a rather rare garnet, bright green in color, usually found as small crystals associated with
Less common species
- Calcium in X site
- Goldmanite: Ca3(V3+,Al,Fe3+)2(SiO4)3
- Kimzeyite: Ca3(Zr, Ti)2[(Si,Al,Fe3+)O4]3
- Morimotoite: Ca3Ti4+Fe2+(SiO4)3
- Schorlomite: Ca3Ti4+2(SiO4)(Fe3+O4)2
- Hydroxide bearing – calcium in X site
- Hydrogrossular: Ca3Al2(SiO4)3−x(OH)4x
- Hibschite: Ca3Al2(SiO4)3−x(OH)4x (where x is between 0.2 and 1.5)
- Katoite: Ca3Al2(SiO4)3−x(OH)4x (where x is greater than 1.5)
- Hydrogrossular: Ca3Al2(SiO4)3−x(OH)4x
- Magnesium or manganese in X site
- Knorringite: Mg3Cr2(SiO4)3
- Majorite: Mg3(Fe2+Si)(SiO4)3
- Calderite: Mn3Fe3+2(SiO4)3
Knorringite
Knorringite is a magnesium-chromium garnet species with the formula Mg3Cr2(SiO4)3. Pure endmember knorringite never occurs in nature. Pyrope rich in the knorringite component is only formed under high pressure and is often found in kimberlites. It is used as an indicator mineral in the search for diamonds.[34]
Garnet structural group
- Formula: X3Z2(TO4)3 (X = Ca, Fe, etc., Z = Al, Cr, etc., T = Si, As, V, Fe, Al)
- All are cubic or strongly pseudocubic.
| IMA/CNMNC Nickel-Strunz Mineral class |
Mineral name | Formula | Crystal system | Point group | Space group |
|---|---|---|---|---|---|
| 04 Oxide | Bitikleite-(SnAl) | Ca3SnSb(AlO4)3 | isometric | m3m | Ia3d |
| 04 Oxide | Bitikleite-(SnFe) | Ca3(SnSb5+)(Fe3+O4)3 | isometric | m3m | Ia3d |
| 04 Oxide | Bitikleite-(ZrFe) | Ca3SbZr(Fe3+O4)3 | isometric | m3m | Ia3d |
| 04 Tellurate | Yafsoanite | Ca3Zn3(Te6+O6)2 | isometric | m3m or 432 |
Ia3d or I4132 |
| 08 Arsenate | Berzeliite | NaCa2Mg2(AsO4)3 | isometric | m3m | Ia3d |
| 08 Vanadate | Palenzonaite | NaCa2Mn2+2(VO4)3 | isometric | m3m | Ia3d |
| 08 Vanadate | Schäferite | NaCa2Mg2(VO4)3 | isometric | m3m | Ia3d |
- IMA/CNMNC – Nickel-Strunz – Mineral subclass: 09.A Nesosilicate
- Nickel-Strunz classification: 09.AD.25
| Mineral name | Formula | Crystal system | Point group | Space group |
|---|---|---|---|---|
| Almandine | Fe2+3Al2(SiO4)3 | isometric | m3m | Ia3d |
| Andradite | Ca3Fe3+2(SiO4)3 | isometric | m3m | Ia3d |
| Calderite | Mn+23Fe+32(SiO4)3 | isometric | m3m | Ia3d |
| Goldmanite | Ca3V3+2(SiO4)3 | isometric | m3m | Ia3d |
| Grossular | Ca3Al2(SiO4)3 | isometric | m3m | Ia3d |
| Henritermierite | Ca3Mn3+2(SiO4)2(OH)4 | tetragonal | 4/mmm | I41/acd |
| Hibschite | Ca3Al2(SiO4)(3−x)(OH)4x (x= 0.2–1.5) | isometric | m3m | Ia3d |
| Katoite | Ca3Al2(SiO4)(3−x)(OH)4x (x= 1.5–3) | isometric | m3m | Ia3d |
| Kerimasite | Ca3Zr2(Fe+3O4)2(SiO4) | isometric | m3m | Ia3d |
| Kimzeyite | Ca3Zr2(Al+3O4)2(SiO4) | isometric | m3m | Ia3d |
| Knorringite | Mg3Cr2(SiO4)3 | isometric | m3m | Ia3d |
| Majorite | Mg3(Fe2+Si)(SiO4)3 | tetragonal | 4/m or 4/mmm |
I41/a or I41/acd |
| Menzerite-(Y) | Y2CaMg2(SiO4)3 | isometric | m3m | Ia3d |
| Momoiite | Mn2+3V3+2(SiO4)3 | isometric | m3m | Ia3d |
| Morimotoite | Ca3(Fe2+Ti4+)(SiO4)3 | isometric | m3m | Ia3d |
| Pyrope | Mg3Al2(SiO4)3 | isometric | m3m | Ia3d |
Schorlomite |
Ca3Ti4+2(Fe3+O4)2(SiO4) | isometric | m3m | Ia3d |
| Spessartine | Mn2+3Al2(SiO4)3 | isometric | m3m | Ia3d |
| Toturite | Ca3Sn2(Fe3+O4)2(SiO4) | isometric | m3m | Ia3d |
| Uvarovite | Ca3Cr2(SiO4)3 | isometric | m3m | Ia3d |
- References: Mindat.org; mineral name, chemical formula and space group (American Mineralogist Crystal Structure Database) of the IMA Database of Mineral Properties/ RRUFF Project, Univ. of Arizona, was preferred most of the time. Minor components in formulae have been left out to highlight the dominant chemical endmember that defines each species.
Synthetic garnets
Also known as rare-earth garnets.
The crystallographic structure of garnets has been expanded from the prototype to include chemicals with the general formula A3B2(CO4)3. Besides silicon, a large number of elements have been put on the C site, including
Interesting magnetic properties arise when the appropriate elements are used. In
Lutetium aluminium garnet (LuAG), Al5Lu3O12, is an inorganic compound with a unique crystal structure primarily known for its use in high-efficiency laser devices. LuAG is also useful in the synthesis of transparent ceramics.[41] LuAG is particularly favored over other crystals for its high density and thermal conductivity; it has a relatively small lattice constant in comparison to the other rare-earth garnets, which results in a higher density producing a crystal field with narrower linewidths and greater energy level splitting in absorption and emission.[42]
Terbium gallium garnet (TGG), Tb3Ga5O12, is a Faraday rotator material with excellent transparency properties and is very resistant to laser damage. TGG can be used in optical isolators for laser systems, in optical circulators for fiber optic systems, in optical modulators, and in current and magnetic field sensors.[43]
Another example is gadolinium gallium garnet (GGG), Gd3Ga2(GaO4)3 which is synthesized for use as a substrate for liquid-phase epitaxy of magnetic garnet films for bubble memory and magneto-optical applications.[citation needed]
Geological importance


The mineral garnet is commonly found in metamorphic and to a lesser extent, igneous rocks. Most natural garnets are compositionally zoned and contain inclusions.
In addition to being used to devolve conditions of metamorphism, garnets can be used to date certain geologic events. Garnet has been developed as a U-Pb geochronometer, to date the age of crystallization[47] as well as a thermochronometer in the (U-Th)/He system[48] to date timing of cooling below a closure temperature.
Garnets can be chemically altered and most often alter to serpentine, talc, and chlorite.[45]
Uses

Gemstones
Red garnets were the most commonly used gemstones in the
Pure crystals of garnet are still used as gemstones. The gemstone varieties occur in shades of green, red, yellow, and orange.[52] In the US it is known as the birthstone for January.[2] The garnet family is one of the most complex in the gem world. It is not a single species, but is composed of multiple species and varieties.[53] It is the state mineral of Connecticut,[54] New York's gemstone,[55] and star garnet (garnet with rutile asterisms) is the state gemstone of Idaho.[56]
Industrial uses
Garnet sand is a good abrasive, and a common replacement for silica sand in sand blasting. Alluvial garnet grains which are rounder are more suitable for such blasting treatments. Mixed with very high pressure water, garnet is used to cut steel and other materials in water jets. For water jet cutting, garnet extracted from hard rock is suitable since it is more angular in form, therefore more efficient in cutting.[57]
Garnet paper is favored by cabinetmakers for finishing bare wood.[58]
Garnet sand is also used for
As an abrasive, garnet can be broadly divided into two categories; blasting grade and water jet grade. The garnet, as it is mined and collected, is crushed to finer grains; all pieces which are larger than 60 mesh (250 micrometers) are normally used for sand blasting. The pieces between 60 mesh (250 micrometers) and 200 mesh (74 micrometers) are normally used for water jet cutting. The remaining garnet pieces that are finer than 200 mesh (74 micrometers) are used for glass polishing and lapping. Regardless of the application, the larger grain sizes are used for faster work and the smaller ones are used for finer finishes.[citation needed]
There are different kinds of abrasive garnets which can be divided based on their origin. The largest source of abrasive garnet today is garnet-rich beach sand which is quite abundant on Indian and Australian coasts and the main producers today are Australia and India.[59]
This material is particularly popular due to its consistent supplies, huge quantities and clean material. The common problems with this material are the presence of ilmenite and chloride compounds. Since the material has been naturally crushed and ground on the beaches for past centuries, the material is normally available in fine sizes only. Most of the garnet at the Tuticorin beach in south India is 80 mesh, and ranges from 56 mesh to 100 mesh size.[citation needed]
River garnet is particularly abundant in Australia. The river sand garnet occurs as a placer deposit.[60]
Rock garnet is perhaps the garnet type used for the longest period of time. This type of garnet is produced in America, China and western India. These crystals are crushed in mills and then purified by wind blowing, magnetic separation, sieving and, if required, washing. Being freshly crushed, this garnet has the sharpest edges and therefore performs far better than other kinds of garnet. Both the river and the beach garnet suffer from the tumbling effect of hundreds of thousands of years which rounds off the edges. Gore Mountain Garnet from Warren County, New York, USA, is a significant source of rock garnet for use as an industrial abrasive.[3]
Cultural significance
Garnet is the
In Persia this birth gem was considered a talisman from nature's forces like storm and lightning. It was widely accepted that garnet could signal approaching danger by turning pale.[citation needed]United States
Garnet is
Since 2003 New York State has ranked first in industrial garnet-production in the United States. Since there are just a few companies that account for all U.S. industrial garnet production, published detailed production statistics for New York State are not available. However, generally speaking, Barton Mines in Warren County is the largest U.S. garnet producer.[69]
Collections
The New York State Museum in Albany, NY houses specimens from significant sites across the state, including 93 mineral species from the Balmat-Edwards mining district in St. Lawrence, super garnets from the Barton Mine in the Adirondack Mountains, and Herkimer diamonds from Herkimer County, New York[70]
Oldest garnet mine
The largest garnet mine in the world is located Near North Creek New York and is operated by Barton Mines Corporation who supplies about 90% of the world's garnet.[71] Barton Mines Corporation is the first and oldest industrial garnet mining operation in the world and the second oldest continuous mining operation in the United States under the same management and mining the same product throughout its history. The Gore Mountain Mine of the Barton Mines Corporation was first mined under the direction of H. H. Barton Sr. in 1878 to produce garnet as the primary product.[71]
Largest garnet crystal
The open-pit Barton Garnet Mine, located at Gore Mountain in the Adirondack Highlands, yields the world's largest single crystals of garnet; diameters range from 5 to 35 cm and commonly average 10–18 cm.[72]
Gore Mountain garnets are unique in many respects, and considerable effort has been made to determine the timing of garnet growth. The first dating was that of Basu et al. (1989), who used plagioclase-hornblende-garnet to produce a Sm/Nd isochron that yielded an age of 1059 ± 19 Ma. Mezger et al. (1992) conducted their own Sm/Nd investigation using hornblende and the drilled core of a 50 cm garnet to produce an isochron age of 1051 ± 4 Ma. Connelly (2006) utilized seven different fractions of a Gore Mountain garnet to obtain a Lu-Hf isochron age of 1046.6 ± 6 Ma. It is therefore concluded with confidence that the garnets formed at 1049 ± 5 Ma, the average of the three determinations. This is also the local age of peak metamorphism in the 1090–1040 Ma Ottawan phase of the Grenvillian orogeny and serves as a critical data point in ascertaining the evolution of the megacrystic garnet deposits.[72]
See also
- Abrasive blasting
References
- S2CID 235729616.
- ^ ISBN 0-87311-019-6
- ^ ISBN 047157452X.
- ^ pomegranate. Online Etymology Dictionary. Retrieved on 2011-12-25.
- ^ garnet. Online Etymology Dictionary. Retrieved on 2011-12-25.
- OCLC 33190408.
- ^ Klein & Hurlbut 1993, p. 600.
- .
- ^ (PDF) from the original on 2022-10-09. Retrieved 7 December 2020.
- .
- ^ Smyth, Joe. "Mineral Structure Data". Garnet. University of Colorado. Retrieved 2007-01-12.
- ISBN 9780195106916.
- ISBN 9780903056434.
- .
- ^ D. B. Hoover, B. Williams, C. Williams and C. Mitchell, Magnetic susceptibility, a better approach to defining garnets Archived 2011-10-05 at the Wayback Machine, The Journal of Gemmology, 2008, Volume 31, No. 3/4 pp. 91–103
- ^ a b c Lytvynov, L. A. (2011). "On the words used as names for ruby and sapphire" (PDF). Functional Materials. 18 (2): 274–277. Retrieved 7 December 2020.
- .
- ^ Nesse 2000, pp. 312, 320.
- ISBN 978-3-540-72795-8.
- ^ Hausel, W. Dan (2000). Gemstones and Other Unique Rocks and Minerals of Wyoming – Field Guide for Collectors. Laramie, Wyoming: Wyoming Geological Survey. pp. 268 p.
- ^ doi:10.3133/b1042G.
- ^ Klein & Hurlbut 1993, pp. 453, 587–588.
- ^ Nesse 2000, p. 312.
- .
- ^ "Color Change Garnet Value, Price, and Jewelry Information - Gem Society". International Gem Society. Retrieved 2022-10-13.
- S2CID 95448333.
- JSTOR 24077869.
- .
- S2CID 129908263.
- (PDF) from the original on 2022-10-09. Retrieved 7 December 2020.
- .
- ^ Mindat.org - Tsavorite
- .
- ^ Nixon, Peter H.; Hornung, George (1968). "A new chromium garnet end member, knorringite, from Kimberlite". American Mineralogist. 53 (11–12): 1833–1840. Retrieved 7 December 2020.
- ISBN 978-0-471-60997-1.
- S2CID 31557815.
- PMID 15354712.
- PMID 28570745.
- ^ "What is YIG and How Does It Work So Well?". www.microlambdawireless.com. Retrieved 2023-07-17.
- Bibcode:2015PhDT.......308M.
- ^ "Lutetium Aluminum Garnet - LuAG - Lu3Al5O12". scientificmaterials.com. Retrieved 2016-04-29.
- PMID 24150356.
- ISBN 978-0-19-984628-3.
- ^ ISBN 0-471-80580-7.
- ^ a b c "P-T-t Paths". Teaching Phase Equilibria. Retrieved 2020-03-19.
- ISSN 0009-2541.
- ISSN 0012-821X.
- ^ "Staffordshire Hoard Festival 2019". The Potteries Museum & Art Gallery. Retrieved 18 June 2019.
- ^ "A trail of garnet and gold: Sri Lanka to Anglo-Saxon England". The Historical Association. 22 June 2017. Retrieved 18 June 2019.
- ^ "Acquisitions of the month: June 2018". Apollo Magazine. 5 July 2018. Retrieved 18 June 2019.
- ^ Geological Sciences at University of Texas, Austin. Geo.utexas.edu. Retrieved on 2011-12-25.
- ^ "Garnet Value, Price, and Jewelry Information". International Gem Society. Retrieved 2021-11-16.
- ^ State of Connecticut, Sites º Seals º Symbols Archived 2008-07-31 at the Wayback Machine; Connecticut State Register & Manual; retrieved on December 20, 2008
- ^ New York State Gem Archived 2007-12-08 at the Wayback Machine; State Symbols USA; retrieved on October 12, 2007
- ^ Idaho state symbols. idaho.gov
- ^ Rapple, R. Randolph. "Selecting the right waterjet abrasive". The Fabricator. Retrieved 17 July 2023.
- ISBN 071344407X.
- ISBN 978-1-871677-52-2.
- ^ Industrial Mineral Opportunities in New South Wales
- ^ "Tips & Tools: Birthstones". The National Association of Goldsmiths. Archived from the original on 2007-05-28. Retrieved 2014-06-16.
- ^ Kunz, George F. (1913). The curious lore of precious stones. Lippincott. pp. 275–306, pp. 319-320
- ^ Knuth, Bruce G. (2007). Gems in Myth, Legend and Lore (Revised edition). Parachute: Jewelers Press. p. 294.
- ^ Kunz (1913), pp. 345–347
- ^ "Minerals of New York State". State of New York. Retrieved 2022-02-25.
- ^ "State of Connecticut – Sites, Seals and Symbols". State of Connecticut. Retrieved 2009-11-12.
- ^ "Idaho Symbols". State of Idaho. Archived from the original on 2010-06-30. Retrieved 2009-11-12.
- ^ "Vermont Emblems". State of Vermont. Archived from the original on 2009-10-29. Retrieved 2009-11-12.
- ^ "New York State Gem". Joseph L. Ferguson. Retrieved 2022-04-06.
- ^ "Minerals of New York State". State of New York.
- ^ a b "The Garnet Story". Lake George Guide. 21 October 2010.
- ^ .
Further reading
- Hurlbut, Cornelius S.; Klein, Cornelis, 1985, Manual of Mineralogy, 20th ed., Wiley, ISBN 0-471-80580-7
- Color Encyclopedia of Gemstones, ISBN 0-442-20333-0
External links
- http://www.gemstonemagnetism.com contains a comprehensive section about garnets and garnet magnetism.
- USGS Garnet locations – USA
- http://gemstone.org/education/gem-by-gem/154-garnet
- http://www.mindat.org/min-10272.html
- Blog post on garnets on the Law Library of Congress's blog
- https://www.birthstone.guide/garnet-birthstone-meaning Garnet birthstone stories