Gastric acid
This article needs additional citations for verification. (March 2022) |
Gastric acid, gastric juice, or stomach acid is a digestive fluid formed within the
The primary active component of gastric acid is
The highly acidic environment in the stomach lumen degrades
Secretion
A typical adult human stomach will secrete about 1.5 liters of gastric acid daily.
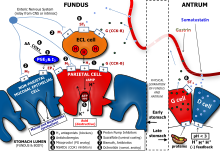
Chloride and sodium ions are secreted actively from the
The enzyme carbonic anhydrase catalyses the reaction between carbon dioxide and water to form carbonic acid. This acid immediately dissociates into hydrogen and bicarbonate ions. The hydrogen ions leave the cell through H+/K+ ATPase antiporter pumps.
At the same time, sodium ions are actively reabsorbed[
The highest concentration that gastric acid reaches in the stomach is 160
There is a small continuous basal secretion of gastric acid between meals of usually less than 10 mEq/hour.[4]
There are three phases in the secretion of gastric acid which increase the secretion rate in order to digest a meal:[2]
- The D cells.[5]
- The amino acidspresent in the food.
- The amino acids. The duodenal cells release entero-oxyntin which acts on parietal cells without affecting gastrin.[5]
Regulation of secretion

Gastric acid production is regulated by both the
all inhibit production.The production of gastric acid in the stomach is tightly regulated by positive regulators and
Nerve endings in the stomach secrete two stimulatory neurotransmitters: acetylcholine[6] and gastrin-releasing peptide. Their action is both direct on parietal cells and mediated through the secretion of gastrin from G cells and histamine from enterochromaffine-like cells. Gastrin acts on parietal cells directly and indirectly too, by stimulating the release of histamine.
The release of histamine is the most important positive regulation mechanism of the secretion of gastric acid in the stomach. Its release is stimulated by gastrin and acetylcholine and inhibited by somatostatin.[7]
Neutralization
In the
- HCl + NaHCO3 → NaCl + H2CO3
The
Role in disease
In
In
In diseases featuring excess vomiting, patients develop hypochloremic metabolic alkalosis (decreased blood acidity by H+ and chlorine depletion).
Gastroesophageal reflux disease occurs when stomach acid repeatedly flows back into the Esophagus, this backwash (acid reflux) can irritate the lining of the esophagus.
Many people experience acid reflux from time to time. However, when acid reflux happens repeatedly over time, it can cause GERD.
Most people are able to manage the discomfort of GERD with lifestyle changes and medications. While it is uncommon, some may need surgery to ease symptoms.[9]
Pharmacology
The proton pump enzyme is the target of
Comparison between humans and other animals
The pH of gastric acid in humans is 1.5-2.0. This is a much lower pH level than that of most animals and very close to scavengers, which eat carrion.[10] This suggests that carrion feeding could have been more important in human evolution than previously thought.[10]
History
This section needs expansion. You can help by adding to it. (November 2010) |
The role of gastric acid in digestion was established in the 1820s and 1830s by William Beaumont on Alexis St. Martin, who, as a result of an accident, had a fistula (hole) in his stomach, which allowed Beaumont to observe the process of digestion and to extract gastric acid, verifying that acid played a crucial role in digestion.[11]
See also
References
- ISBN 978-0-13-458099-9.
- ^ a b c Dworken HJ (2016). Human digestive system: gastric secretion. Encyclopædia Britannica Inc.
- ISBN 0-7216-0240-1.
- ISBN 978-0-7817-7153-5.
- ^ a b Lecture, "Function of the Stomach and Small Intestine" Deakin University School of Medicine. October 15, 2012
- ^ "acetylcholine | Definition, Function, & Facts | Britannica". www.britannica.com. Retrieved 2021-12-13.
- ^ "Somatostatin". www.hormone.org. Retrieved 2021-12-13.
- PMID 2506730.
- ^ "Gastroesophageal reflux disease (GERD) - Symptoms and causes". Mayo Clinic. Retrieved 2023-09-10.
- ^ PMID 26222383.
- ISBN 0-7148-2096-2.
