Gene doping
| Part of a series on |
| Doping in sport |
|---|
  |
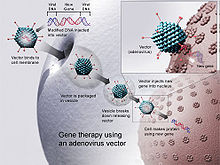
Gene doping is the hypothetical non-therapeutic use of
The historical development of interest in gene doping by athletes and concern about the risks of gene doping and how to detect it moved in parallel with the development of the field of gene therapy, especially with the publication in 1998 of work on a
Scientists themselves, as well as bodies including the World Anti-Doping Agency (WADA), the International Olympic Committee, and the American Association for the Advancement of Science, started discussing the risk of gene doping in 2001, and by 2003 WADA had added gene doping to the list of banned doping practices, and shortly thereafter began funding research on methods to detect gene doping.
Genetic enhancement includes manipulation of genes or
History of gene doping
The history of concern about the potential for gene doping follows the history of gene therapy, the medical use of genes to treat diseases, which was first clinically tested in the 1990s.[4] Interest by the athletic community was especially spurred by the creation in a university lab of a "mighty mouse", created by administering a virus carrying the gene expressing insulin-like growth factor 1 to mice; the mice were stronger and remained strong even as they aged, without exercise.[4] The lab had been seeking treatments for muscle wasting diseases, but when their work was made public, the lab was inundated with calls from athletes seeking the treatment, with one coach offering his whole team.[5] The scientist told The New York Times in 2007: "I was quite surprised, I must admit. People would try to entice me, saying things like, 'It'll help advance your research.' Some offered to pay me." He also told the Times that every time similar research is published he gets calls and that he explains that, even should the treatment became ready for use in people, which would take years, there would be serious risks, including death; he also said that even after he explains this, the athletes still want it.[5]
In 1999, the field of gene therapy was set back when Jesse Gelsinger died in a gene therapy clinical trial, suffering a massive inflammatory reaction to the drug.[4][6] This led regulatory authorities in the US and Europe to increase safety requirements in clinical trials even beyond the initial restrictions that had been put in place at the beginning of the biotechnology era to deal with the risks of recombinant DNA.[7]
In June 2001, Theodore Friedmann, one of the pioneers of gene therapy, and Johann Olav Koss an Olympic gold medallist in speed skating, published a paper that was the first public warning about gene doping.[7][8] Also in June 2001, a Gene Therapy Working Group, convened by the Medical Commission of the International Olympic Committee noted that "we are aware that there is the potential for abuse of gene therapy medicines and we shall begin to establish procedures and state-of-the-art testing methods for identifying athletes who might misuse such technology".[7]
Research was published in 2002 about a preclinical gene therapy called Repoxygen, which delivered the gene encoding erythropoietin (EPO) as a potential treatment for anemia.[4] The scientists from that company also received calls from athletes and coaches.[4] In that same year the World Anti-Doping Agency held its first meeting to discuss the risk of gene doping,[7][9] and the US President's Council on Bioethics discussed gene doping in the context of human enhancement at several sessions.[10][11][12]
In 2003, the field of gene therapy took a step forward and a step back; first gene therapy drug was approved, Gendicine, which was approved in China for the treatment of certain cancers,[13] but children in France who had seemingly been effective treated with gene therapy for severe combined immunodeficiency (non-human) began developing leukemia.[6] In 2003 the BALCO scandal became public, in which chemists, trainers and athletes conspired to evade doping controls with new and undetectable doping substances.[7] In 2003 the World Doping Agency proactively added gene doping to the list of banned doping practices.[4] Also in 2003, a symposium convened by the American Association for the Advancement of Science focused on the issue.[14]
Research published in 2004 showing that mice given gene therapy coding for a protein called PPAR gamma had about double the endurance of untreated mice and were dubbed "marathon mice"; those scientists received calls from athletes and coaches.[4] Also in 2004 the World Anti-Doping Agency began to fund research to detect gene doping, and formed a permanent expert panel to advise it on risks and to guide the funding.[4][9]
In 2006 interest from athletes in gene doping received widespread media coverage due its mention during the trial of a German coach who was accused and found guilty of giving his athletes
In 2011 the second gene therapy drug was approved;
In 2012
As the field of gene therapy has developed, the risk of gene doping becoming a reality has increased with it.[6]
Agents used in gene doping
There are numerous genes of interest as agents for gene doping.
The risks of gene doping would be similar to those of gene therapy: immune reaction to the native protein leading to the equivalent of a
Alpha-actinin-3
Myostatin
Erythropoietin (EPO)
Insulin-like growth factor 1
Insulin-like growth factor 1 is a protein involved in the mediation of the growth hormone. Administration of IGF-1 to mice has resulted in more muscle growth and quicker muscle and nerve regeneration.[19][6] If athletes were to use this the sustained production of IGF-1 could cause heart disease and cancer.[19]
Others
Modulating the levels of proteins that affect psychology are also potential goals for gene doping; for example pain perception depends on
Detection
The
Indirect methods are by nature more subjective, as it becomes very difficult to determine which anomalies are proof of gene doping, and which are simply natural, though unusual, biological properties.[6] For example, Eero Mäntyranta, an Olympic cross country skier, had a mutation which made his body produce abnormally high amounts of red blood cells. It would be very difficult to determine whether or not Mäntyranta's red blood cell levels were due to an innate genetic advantage, or an artificial one.[26]
First generation of gene doping detecting methods
Gene doping detection idea started in 2004 when WADA has put gene doping in the banned list and started investigating a new method that can detect the inserted transgenes.
The first generation of gene doping detection techniques used PCR tests that targets the transgenes’ sequences. It can be obtained from a blood sample which will contain endogenous and transgene DNA since a small amount of the transgene will leak into the bloodstream. It can be easily distinguished from endogenous DNA because it lacks introns since the transgene will most likely use cDNA that is obtained by reverse transcriptase from RNA, which has removed its intones though RNA splicing leaving only exon-exon junction that include only the coding sequences and some important sequences like promoters since the viral victors has a limited capacity. Therefore, PCR can target these exon-exon junctions as a unique sequence that is not present in gDNA[27]
Real time PCR
PCR has many applications in molecular biology field including
In gene doping detection, If the sequence started to amplify producing an exponential graph, then the test is positive and indicates the presence of the gene in the sample obtained from that person. But if the sequence didn't amplify and a linear graph was produced, then the test is said to be negative and the targeted DNA sequence was not present in that person's sample.[28]
Next generation sequencing
With the limitation of the first-generation detection methods, it was important to develop a new method that overcomes the previous failures with a high accuracy and can detect the manipulation in DNA sequences that could evade to be detected by PCR methods.
The solution was using
DNA sequencing was established in the 1970s with the two-dimensional chromatography and kept improving until 2001 with the completion of human genome project which costed about three billion dollars and required 15 years to finish sequencing the whole genome. However, with nowadays sequencing technology, whole genome sequencing (WGS) takes only a single day and costs around a thousand dollars. Moreover, a new sequencing technology is under development that will cost only 100 dollars for WGS.[30]
There are many NGS techniques that are used in DNA sequencing but the most used method is the one done by illumina[31]
Research
A 2016 review found that about 120 DNA polymorphisms had been identified in the literature related to some aspect of athletic performance, 77 related to endurance and 43 related to power. 11 had been replicated in three or more studies and six were identified in
The 11 replicated markers were:[20]
- Endurance
- power/strength markers
- ACE Alu I/D (rs4646994) (called ACE D)
- ACTN3Arg577
- AMPD1Gln12
- HIF1A 582Ser
- MTHFRrs1801131 C
- NOS3rs2070744 T
- PPARG12Ala
The six GWAS markers were:[20]
- CREM rs1531550 A,
- DMD rs939787 T
- GALNT13 rs10196189 G
- NFIA-AS1 rs1572312 C,
- RBFOX1 rs7191721 G
- TSHRrs7144481 C
Ethics of gene doping
The World Anti-Doping Agency (WADA) determined that any non-therapeutic form of genetic manipulation for enhancement of athletic performance is banned under its code. There are guidelines to determine if said technology should be prohibited in sport: if two of the three conditions are met, then the technology is prohibited in sport (harmful to one's health, performance enhancing, and/or against the "spirit of sport").[32]
Kayser et al. argue that gene doping could level the playing field if all athletes receive equal access. Critics claim that any therapeutic intervention for non-therapeutic/enhancement purposes compromises the ethical foundations of medicine and sports.[33]
The high risks associated with gene therapy can be outweighed by the potential of saving the lives of individuals with diseases: according to Alain Fischer, who was involved in clinical trials of gene therapy in children with severe combined immunodeficiency, "Only people who are dying would have reasonable grounds for using it. Using gene therapy for doping is ethically unacceptable and scientifically stupid."[34] As seen with past cases, including the steroid tetrahydrogestrinone (THG), athletes may choose to incorporate risky genetic technologies into their training regimes.[3]
The mainstream perspective is that gene doping is dangerous and unethical, as is any application of a therapeutic intervention for non-therapeutic or enhancing purposes, and that it compromises the ethical foundation of medicine and the spirit of sport.
See also
References
- ^ S2CID 45124293.
- ^ World Anti-Doping Agency The World Anti-Doping Code: The 2008 Prohibited List of International Standards pages 7-8
- ^ a b Wells DJ. 2008. "Gene doping: the hype and the reality". Br. J. Pharmacol. [Internet] 154:623–31.
- ^ JSTOR 20494726.
- ^ a b c Gretchen Reynolds for The New York Times. June 3, 2007. Outlaw DNA
- ^ PMID 23082866.
- ^ S2CID 23370410.
- PMID 11407894.
- ^ a b c World Anti Doping Agency. Gene Doping Page archived January 7, 2016
- ^ President's Council on Bioethics April 25-26, 2002 Agenda, Working Paper 7
- ^ President's Council on Bioethics July 11-12, 2002 Meeting Agenda Session 4
- ^ President's Council on Bioethics September 12-13, 2002 Meeting Agenda Session 7: Enhancement 5: Genetic Enhancement of Muscle, H. Lee Sweeney, Ph.D., Professor and Chairman of Physiology, University of Pennsylvania
- PMID 14704685.
- ^ UCSD Public Relations: February 18, 2003 News Release: Bigger, Faster, Stronger: Genetic Enhancement and Athletics
- ^ AdisInsight Vascular endothelial growth factor gene therapy - HSCI Page accessed June 5, 2016
- ^ "Gene Therapy for PAD Approved". 6 December 2011. Retrieved 5 August 2015.
- ^ Richards, Sabrina (6 November 2012). "Gene Therapy Arrives in Europe". The Scientist.
- ^ Gallagher, James. (2 November 2012) BBC News – Gene therapy: Glybera approved by European Commission. BBC. Retrieved 15 December 2012.
- ^ S2CID 4983625.
- ^ PMID 26231489. review.
- .
- ^ PMID 6572366.
- S2CID 9770266.
- PMID 22030863.
- S2CID 41151847.
- PMID 15255589. Archived from the originalon 13 November 2005.
- PMID 25435666.
- ^ "Laboratory Guidelines - Gene Doping Detection based on Polymerase Chain Reaction (PCR)". World Anti Doping Agency. Retrieved 2023-01-21.
- ^ "Gene doping detection by next generation sequencing". World Anti Doping Agency. Retrieved 2023-01-21.
- S2CID 195878410.
- S2CID 235370014.
- S2CID 840205.
- PMID 17394662.
- PMID 17471256.
- PMID 17394662.
- S2CID 206521864. Archived from the original(PDF) on 2016-08-06. Retrieved 2016-06-06.
- S2CID 40434985.
- ISBN 978-0-415-29880-3.
- S2CID 149132266.
attribution contains text from Human genetic enhancement as of 17:03, 26 October 2020
