Genetic recombination
Genetic recombination (also known as genetic reshuffling) is the exchange of genetic material between different organisms which leads to production of offspring with combinations of traits that differ from those found in either parent. In eukaryotes, genetic recombination during meiosis can lead to a novel set of genetic information that can be further passed on from parents to offspring. Most recombination occurs naturally and can be classified into two types: (1) interchromosomal recombination, occurring through independent assortment of alleles whose loci are on different but homologous chromosomes (random orientation of pairs of homologous chromosomes in meiosis I); & (2) intrachromosomal recombination, occurring through crossing over.[1]
During
Recombination may also occur during
Gene conversion – the process during which homologous sequences are made identical also falls under genetic recombination.
Genetic recombination and recombinational DNA repair also occurs in bacteria and archaea, which use asexual reproduction.
Recombination can be artificially induced in laboratory (in vitro) settings, producing recombinant DNA for purposes including vaccine development.
V(D)J recombination in organisms with an adaptive immune system is a type of site-specific genetic recombination that helps immune cells rapidly diversify to recognize and adapt to new pathogens.
Synapsis
During meiosis, synapsis (the pairing of homologous chromosomes) ordinarily precedes genetic recombination.
Mechanism
Genetic recombination is catalyzed by many different enzymes. Recombinases are key enzymes that catalyse the strand transfer step during recombination. RecA, the chief recombinase found in Escherichia coli, is responsible for the repair of DNA double strand breaks (DSBs). In yeast and other eukaryotic organisms there are two recombinases required for repairing DSBs. The RAD51 protein is required for mitotic and meiotic recombination, whereas the DNA repair protein, DMC1, is specific to meiotic recombination. In the archaea, the ortholog of the bacterial RecA protein is RadA.
- Bacterial recombination
In
Chromosomal crossover
In
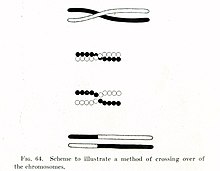
Chromosomal crossover involves recombination between the paired chromosomes inherited from each of one's parents, generally occurring during meiosis.[citation needed] During prophase I (pachytene stage) the four available chromatids are in tight formation with one another.[citation needed] While in this formation, homologous sites on two chromatids can closely pair with one another, and may exchange genetic information.[5]
Because there is a small probability of recombination at any location along a chromosome, the
Tracking the movement of genes resulting from crossovers has proven quite useful to geneticists. Because two genes that are close together are less likely to become separated than genes that are farther apart, geneticists can deduce roughly how far apart two genes are on a chromosome if they know the frequency of the crossovers.[citation needed] Geneticists can also use this method to infer the presence of certain genes. Genes that typically stay together during recombination are said to be linked. One gene in a linked pair can sometimes be used as a marker to deduce the presence of the other gene. This is typically used to detect the presence of a disease-causing gene.[6]
The recombination frequency between two loci observed is the
Gene conversion
In gene conversion, a section of genetic material is copied from one chromosome to another, without the donating chromosome being changed. Gene conversion occurs at high frequency at the actual site of the recombination event during meiosis. It is a process by which a DNA sequence is copied from one DNA helix (which remains unchanged) to another DNA helix, whose sequence is altered. Gene conversion has often been studied in fungal crosses[8] where the 4 products of individual meioses can be conveniently observed. Gene conversion events can be distinguished as deviations in an individual meiosis from the normal 2:2 segregation pattern (e.g. a 3:1 pattern).
Nonhomologous recombination
Recombination can occur between DNA sequences that contain no sequence homology. This can cause chromosomal translocations, sometimes leading to cancer.
In B cells
Genetic engineering
In
Examples include Restriction enzyme mediated integration, Gibson assembly and Golden Gate Cloning.
Recombinational repair
DNA damages caused by a variety of exogenous agents (e.g. UV light, X-rays, chemical cross-linking agents) can be repaired by homologous recombinational repair (HRR).[9][10] These findings suggest that DNA damages arising from natural processes, such as exposure to reactive oxygen species that are byproducts of normal metabolism, are also repaired by HRR. In humans, deficiencies in the gene products necessary for HRR during meiosis likely cause infertility[11] In humans, deficiencies in gene products necessary for HRR, such as BRCA1 and BRCA2, increase the risk of cancer (see DNA repair-deficiency disorder).
In bacteria, transformation is a process of gene transfer that ordinarily occurs between individual cells of the same bacterial species. Transformation involves integration of donor DNA into the recipient chromosome by recombination. This process appears to be an adaptation for repairing DNA damages in the recipient chromosome by HRR.[12] Transformation may provide a benefit to pathogenic bacteria by allowing repair of DNA damage, particularly damages that occur in the inflammatory, oxidizing environment associated with infection of a host.
When two or more viruses, each containing lethal genomic damages, infect the same host cell, the virus genomes can often pair with each other and undergo HRR to produce viable progeny. This process, referred to as multiplicity reactivation, has been studied in lambda and
Meiotic recombination
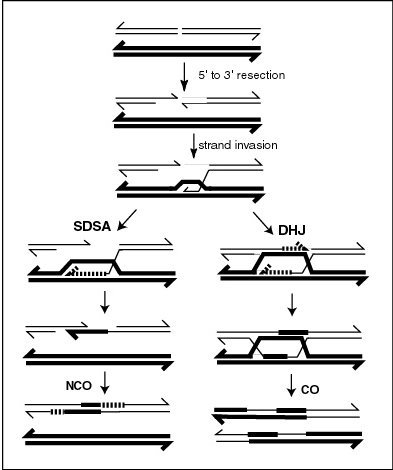
A molecular model for the mechanism of meiotic recombination presented by Anderson and Sekelsky[14] is outlined in the first figure in this article. Two of the four chromatids present early in meiosis (prophase I) are paired with each other and able to interact. Recombination, in this model, is initiated by a double-strand break (or gap) shown in the DNA molecule (chromatid) at the top of the figure. Other types of DNA damage may also initiate recombination. For instance, an inter-strand cross-link (caused by exposure to a cross-linking agent such as mitomycin C) can be repaired by HRR.
Two types of recombinant product are produced. Indicated on the right side is a "crossover" (CO) type, where the flanking regions of the chromosomes are exchanged, and on the left side, a "non-crossover" (NCO) type where the flanking regions are not exchanged. The CO type of recombination involves the intermediate formation of two "Holliday junctions" indicated in the lower right of the figure by two X-shaped structures in each of which there is an exchange of single strands between the two participating chromatids. This pathway is labeled in the figure as the DHJ (double-Holliday junction) pathway.
The NCO recombinants (illustrated on the left in the figure) are produced by a process referred to as "synthesis dependent strand annealing" (SDSA). Recombination events of the NCO/SDSA type appear to be more common than the CO/DHJ type.[15] The NCO/SDSA pathway contributes little to genetic variation, since the arms of the chromosomes flanking the recombination event remain in the parental configuration. Thus, explanations for the adaptive function of meiosis that focus exclusively on crossing-over are inadequate to explain the majority of recombination events.
Achiasmy and heterochiasmy
Achiasmy is the phenomenon where autosomal recombination is completely absent in one sex of a species. Achiasmatic chromosomal segregation is well documented in male Drosophila melanogaster. Heterochiasmy occurs when recombination rates differ between the sexes of a species.[16] This sexual dimorphic pattern in recombination rate has been observed in many species. In mammals, females most often have higher rates of recombination. The "Haldane-Huxley rule" states that achiasmy usually occurs in the heterogametic sex.[16]
RNA virus recombination
Numerous RNA viruses are capable of genetic recombination when at least two viral
Recombination also occurs in the
Recombination in RNA viruses appears to be an adaptation for coping with genome damage.[17] Switching between template strands during genome replication, referred to as copy-choice recombination, was originally proposed to explain the positive correlation of recombination events over short distances in organisms with a DNA genome (see first Figure, SDSA pathway).[25]
Recombination can occur infrequently between animal viruses of the same species but of divergent lineages. The resulting recombinant viruses may sometimes cause an outbreak of infection in humans.[23]
Especially in coronaviruses, recombination may also occur even among distantly related evolutionary groups (subgenera), due to their characteristic transcription mechanism, that involves subgenomic mRNAs that are formed by template switching.[26][24]
When replicating its
Recombination appears to be a major driving force in determining genetic variability within coronaviruses, as well as the ability of coronavirus species to jump from one host to another and, infrequently, for the emergence of novel species, although the mechanism of recombination in is unclear.[23]
In early 2020, many genomic sequences of Australian SARS‐CoV‐2 isolates have deletions or mutations (29742G>A or 29742G>U; "G19A" or "G19U") in the s2m, suggesting that RNA recombination may have occurred in this RNA element. 29742G("G19"), 29744G("G21"), and 29751G("G28") were predicted as recombination hotspots.[29] During the first months of the COVID-19 pandemic, such a recombination event was suggested to have been a critical step in the evolution of SARS-CoV-2's ability to infect humans.[30] Linkage disequilibrium analysis confirmed that RNA recombination with the 11083G > T mutation also contributed to the increase of mutations among the viral progeny. The findings indicate that the 11083G > T mutation of SARS-CoV-2 spread during Diamond Princess shipboard quarantine and arose through de novo RNA recombination under positive selection pressure. In three patients on the Diamond Princess cruise, two mutations, 29736G > T and 29751G > T (G13 and G28) were located in Coronavirus 3′ stem-loop II-like motif (s2m) of SARS-CoV-2. Although s2m is considered an RNA motif highly conserved in 3' untranslated region among many coronavirus species, this result also suggests that s2m of SARS-CoV-2 is RNA recombination/mutation hotspot.[31]
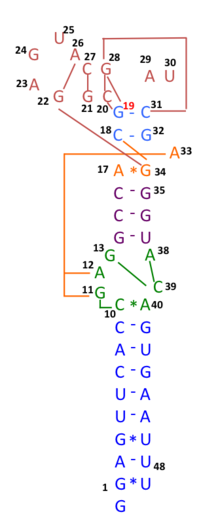
SARS-CoV-2's entire receptor binding motif appeared, based on preliminary observations, to have been introduced through recombination from coronaviruses of pangolins.[32] However, more comprehensive analyses later refuted this suggestion and showed that SARS-CoV-2 likely evolved solely within bats and with little or no recombination.[33][34]
Role of recombination in the origin of life
Nowak and Ohtsuki[35] noted that the origin of life (abiogenesis) is also the origin of biological evolution. They pointed out that all known life on earth is based on biopolymers and proposed that any theory for the origin of life must involve biological polymers that act as information carriers and catalysts. Lehman[36] argued that recombination was an evolutionary development as ancient as the origins of life. Smail et al.[37] proposed that in the primordial Earth, recombination played a key role in the expansion of the initially short informational polymers (presumed to be RNA) that were the precursors to life.
See also
- Eukaryote hybrid genome
- Four-gamete test
- Homologous recombination
- Independent assortment
- Recombination frequency
- Recombination hotspot
- Site-specific recombinase technology
- Site-specific recombination
- Reassortment
- V(D)J recombination
References
- PMID 7559335.
- ^ ISBN 978-3-540-07668-1.
- ISBN 978-0-19-50944-1-1.
- ISBN 978-9958-9344-1-4.
- ISBN 978-0-8153-3218-3.
- ^ "Access Excellence". Crossing-over: Genetic Recombination. The National Health Museum Resource Center. Retrieved February 23, 2011.
- ISBN 0-19-509442-5.
- ^ Stacey KA (1994). "Recombination". In Kendrew J, Lawrence E (eds.). The Encyclopedia of Molecular Biology. Oxford: Blackwell Science. pp. 945–950.
- PMID 825857.
- ^ Boyd JB (1978). "DNA repair in Drosophila". In Hanawalt PC, Friedberg EC, Fox CF (eds.). DNA Repair Mechanisms. New York: Academic Press. pp. 449–452.
- S2CID 23743474.
- ^ PMID 18295550.
- PMID 6261109.
- PMID 20967781.
- PMID 17166055.
- ^ PMID 12618416.
- ^ PMID 20335491.
- PMID 21725337.
- PMID 30307534.
- PMID 31540135.
- PMID 1700865.
- ^ PMID 29111273.
- ^ PMID 27012512.
- ^ PMID 34638137.
- .
- PMID 30393776.
- PMID 3021340.
- PMID 14581540.
- PMID 32506536.
- bioRxiv 10.1101/2020.04.20.052019.
- PMID 34248221.
- PMID 32937441.
- PMID 32724171.
- PMID 32958891.
- PMID 18791073.
- S2CID 33130898.
- PMID 30670484.
External links
- Animations – homologous recombination: Animations showing several models of homologous recombination
- The Holliday Model of Genetic Recombination
- Genetic+recombination at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Animated guide to homologous recombination.
![]() This article incorporates
This article incorporates
