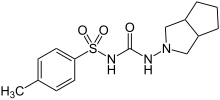
Gliclazide
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Diamicron, Diaprel, Azukon, others[1] |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 10.4 hours |
| Identifiers | |
| |
JSmol) | |
| Melting point | 180 to 182 °C (356 to 360 °F) |
| |
| |
| (verify) | |
Gliclazide, sold under the brand name Diamicron among others, is a sulfonylurea type of
Side effect may include
Gliclazide was patented in 1966 and approved for medical use in 1972.[8] It is on the World Health Organization's List of Essential Medicines.[9] It is not available for sale in the United States.[10]
Medical uses
Gliclazide is used for control of hyperglycemia in gliclazide-responsive
National Kidney Foundation (2012 Update) claims that Gliclazide does not require dosage up titration even in end stage kidney disease.
Contraindications
- Type 1 diabetes[12]
- Hypersensitivity to sulfonylureas[12]
- Severe renal or hepatic failure[12] (But relatively useful in mild renal impairment e.g. CKD stage 3)
- Pregnancy and lactation[12]
Adverse effects
Common adverse effects over 10%:[13]
- Hypoglycemia (11 - 12%) - while it was shown to have the same efficacy as glimepiride, one of the newer sulfonylureas, the European GUIDE study has shown that it has approximately 50% less hypoglycemic confirmed episodes in comparison with glimepiride.[14]
Uncommon adverse effect between 1 - 10%:[13]
- Hypertension (3 - 4% incidence)
- Dizziness (2% incidence)
- Hyperglycemia (2% incidence)
- Viral infection (6 - 8% incidence)
- Back pain (4 - 5% incidence)
Rare adverse effects (under 1%):[13]
- cystitis
- weight gain
- Vomiting
Interactions
Hyperglycemic action may be caused by
Overdose
Gliclazide overdose may cause severe hypoglycemia, requiring urgent administration of glucose by IV and Monitoring.[16]
Mechanism of action
Gliclazide selectively binds to sulfonylurea receptors (SUR-1) on the surface of the pancreatic beta-cells. It was shown to provide cardiovascular protection as it does not bind to sulfonylurea receptors (SUR-2A) in the heart.[17] This binding effectively closes these K+ ion channels. This decreases the efflux of potassium from the cell which leads to the depolarization of the cell. This causes voltage dependent Ca2+ ion channels to open increasing the Ca2+ influx. The calcium can then bind to and activate calmodulin which in turn leads to exocytosis of insulin vesicles leading to insulin release.[18]
The mouse model of Maturity-onset diabetes of the young (MODY) diabetes suggested that the reduced gliclazide clearance stands behind their therapeutic success in human MODY patients, but Urbanova et al. found that human MODY patients respond differently and that there was no consistent decrease in gliclazide clearance in randomly selected HNF1A-MODY and HNF4A-MODY patients.[19]
Its classification has been ambiguous, as literature uses it as both a first-generation[20] and second-generation[21] sulfonylurea.
Properties
According to the Biopharmaceutical Classification System (BCS), gliclazide falls under the BCS Class II drug, which is poorly soluble and highly permeable.
Water solubility = 0.027mg/L[citation needed]
- Hypoglycemic sulfonylurea, restoring first peak of insulin secretion, increasing insulin Sensitivity.[22]
- No active circulating Metabolites.[13]
Metabolism
Gliclazide undergoes extensive metabolism to several inactive metabolites in human beings, mainly methylhydroxygliclazide and carboxygliclazide. CYP2C9 is involved in the formation of hydroxygliclazide in human liver microsomes and in a panel of recombinant human P450s in vitro.[23][24] But the pharmacokinetics of gliclazide MR are affected mainly by CYP2C19 genetic polymorphism instead of CYP2C9 genetic polymorphism.[25][26]
References
- ^ "Gliclazide - Drugs.com". www.drugs.com. Archived from the original on 27 December 2016. Retrieved 27 December 2016.
- ^ "Gliclazide GH MR, Gliclazide LAPL MR, Gliclazide Lupin MR (Lupin Australia Pty Limited)". Therapeutic Goods Administration (TGA). 5 December 2022. Retrieved 7 April 2023.
- ^ "Product monograph brand safety updates". Health Canada. 7 July 2016. Retrieved 3 April 2024.
- ^ a b c d "Gliclazide Accord-UK 30mg Prolonged-release Tablets - Summary of Product Characteristics (SmPC)". (emc). 12 February 2021. Archived from the original on 22 September 2022. Retrieved 30 December 2021.
- ^ "Diamicron 30 mg MR Tablets - Summary of Product Characteristics (SmPC)". (emc). 11 May 2020. Retrieved 29 December 2021.
- ^ "Dacadis MR 30mg Modified Release Tablets - Summary of Product Characteristics (SmPC)". (emc). 15 July 2020. Retrieved 29 December 2021.
- ^ ISBN 9780857111562.
- ISBN 9783527607495. Archivedfrom the original on 27 December 2016.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Gliclazide Advanced Patient Information - Drugs.com". www.drugs.com. Archived from the original on 27 December 2016. Retrieved 27 December 2016.
- ^ "My Site - Special Article: Remission of Type 2 Diabetes". guidelines.diabetes.ca. Retrieved 1 June 2023.
- ^ a b c d "GLICLAZIDE 60 MG MR TABLETS DRUG LEAFLET". Drugs.com. Retrieved 23 March 2020.
- ^ a b c d "Gliclazide". Lexicomp. Wolters Kluwer N.V.
- S2CID 13636359.
- S2CID 21519151.
- S2CID 239887850.
- PMID 11484080.
- S2CID 239887850.
- PMID 26446475.
- PMID 2106423.
- PMID 16713429.
- S2CID 239887850.
- PMID 8719909.
- PMID 17517049.
- PMID 17298483.
- PMID 18204476.
