Glioma
This article may require cleanup to meet Wikipedia's quality standards. The specific problem is: Article is full of grammatical and tone errors. (January 2022) |
| Glioma | |
|---|---|
 | |
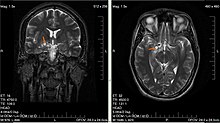
| Glioma in the left parietal lobe (brain CT scan), WHO grade 2 | |
| Specialty | Oncology, Neurology |
A glioma is a type of
Signs and symptoms
Symptoms of gliomas depend on which part of the central nervous system is affected. A brain glioma can cause
A child who has a subacute disorder of the central nervous system that produces cranial nerve abnormalities (especially of cranial nerve VII and the lower bulbar nerves), long-tract signs, unsteady gait secondary to spasticity, and some behavioral changes is most likely to have a pontine glioma.[4]
Causes
Hereditary disorders
The exact causes of gliomas are not known.
Radiation
The best-known risk factor is exposure to ionizing radiation, and CT scan radiation is an important cause.
Infection with cytomegalovirus
Some studies have reported that glioblastomas are infected with cytomegalovirus, with suggestions that this may speed the development of tumors.[18][19][20] However, this is a controversial opinion, with recent in-depth studies failing to find an association between viral infection and glioma growth.[21] There is also evidence that previous studies may have been impacted by false-positive antibody staining artifacts.[22]
Farming
Studies have shown that farmers have higher rates of gliomas compared to the general population. In a 2021 meta-analysis, 40 of 52 studies since 1998 reported positive associations between farming and brain cancer with effect estimates ranging from 1.03 to 6.53, of which 80% are gliomas. Livestock farming was associated with a greater risk compared with crop farming. Farmers with documented exposure to pesticides had greater than a 20% elevated risk of brain cancer[23][unreliable source?] The TRACTOR project study, including 1017 brain tumors among 1 036 069 farm managers, published in 2022, showed an increased risk of glioma in pig farming (HR = 2.28), crop farming (HR = 1.28) and fruit arboriculture (HR = 1.72)[24][medical citation needed]
Other causes
Data show that architects, surveyors, retail workers, butchers, and engineers have higher rates of gliomas.[25]
Inherited polymorphisms of the DNA repair genes
Germ-line (inherited) polymorphisms of the DNA repair genes ERCC1, ERCC2 (XPD) and XRCC1 increase the risk of glioma.[26] This indicates that altered or deficient repair of DNA damage contributes to the formation of gliomas. DNA damages are a likely major primary cause of progression to cancer in general.[27] Excess DNA damages can give rise to mutations through translesion synthesis. Furthermore, incomplete DNA repair can give rise to epigenetic alterations or epimutations.[28][29] Such mutations and epimutations may provide a cell with a proliferative advantage which can then, by a process of natural selection, lead to progression to cancer.[27]
Epigenetic reductions in expression of another DNA repair protein, ERCC1, were found in an assortment of 32 gliomas.[34] For 17 of the 32 (53%) of the gliomas tested, ERCC1 protein expression was reduced or absent. In the case of 12 gliomas (37.5%) this reduction was due to methylation of the ERCC1 promoter. For the other 5 gliomas with reduced ERCC1 protein expression, the reduction could have been due to epigenetic alterations in microRNAs that affect ERCC1 expression.[35]
When expression of DNA repair genes is reduced, DNA damages accumulate in cells at a higher than normal level, and such excess damages cause increased frequencies of mutation.[36][37][38] Mutations in gliomas frequently occur in either isocitrate dehydrogenase (IDH) 1 or 2 genes.[39] One of these mutations (mostly in IDH1) occurs in about 80% of low grade gliomas and secondary high-grade gliomas.[40] Wang et al.[41] pointed out that IDH1 and IDH2 mutant cells produce an excess metabolic intermediate, 2-hydroxyglutarate, which binds to catalytic sites in key enzymes that are important in altering histone and DNA promoter methylation. Thus, mutations in IDH1 and IDH2 generate a "DNA CpG island methylator phenotype or CIMP"[42][43] that causes promoter hypermethylation and concomitant silencing of tumor suppressor genes such as DNA repair genes MGMT and ERCC1. On the other hand, Cohen et al.[40] and Molenaar et al.[39] pointed out that mutations in IDH1 or IDH2 can cause increased oxidative stress. Increased oxidative damage to DNA could be mutagenic. This is supported by an increased number of DNA double-strand breaks in IDH1-mutated glioma cells.[44] Thus, IDH1 or IDH2 mutations act as driver mutations in glioma carcinogenesis, though it is not clear by which role they are primarily acting. A study, involving 51 patients with brain gliomas who had two or more biopsies over time, showed that mutation in the IDH1 gene occurred prior to the occurrence of a p53 mutation or a 1p/19q loss of heterozygosity, indicating that an IDH1 mutation is an early driver mutation.[45]
Pathophysiology
High-grade gliomas are highly
Conversely, low-grade gliomas grow slowly, often over many years, and can be followed without treatment unless they grow and cause symptoms.[medical citation needed]
Several acquired (not inherited) genetic mutations have been found in gliomas.
Diagnosis
Classification
By type of cell
Gliomas are named according to the specific type of cell with which they share histological features, but not necessarily from which they originate. The main types of glioma are:[50]
- ependymal cells
- glioblastoma multiformeis a malignant astrocytoma and the most common primary brain tumor among adults).
- Oligodendrogliomas: oligodendrocytes
- brain stem
- Optic nerve glioma: develop in or around the optic nerve
- Mixed gliomas, such as oligoastrocytomas, contain cells from different types of glia
By grade
Gliomas are further categorised according to their grade, which is determined by pathologic evaluation of the tumor. The neuropathological evaluation and diagnostics of brain tumor specimens is performed according to WHO Classification of Tumours of the Central Nervous System.[51][52]
- Biologically benign gliomas [WHO grade I] are comparatively low risk and can be removed surgically depending on their location[46]
- Low-grade gliomas [WHO grade II] are well-differentiated (not anaplastic); these tend to exhibit benign tendencies and portend a better prognosis for the patient. However, they have a uniform rate of recurrence and increase in grade over time so should be classified as malignant.
- High-grade [WHO grades III–IV] gliomas are undifferentiated or anaplastic; these are malignant and carry a worse prognosis. Despite being classified as a high-grade glioma, infant-type hemispheric gliomas have relatively good clinical outcomes, yet they endure significant deficits, making them good candidates for therapy de-escalation and trials of molecular targeted therapy.[53]
Of numerous grading systems in use, the most common is the World Health Organization (WHO) grading system for astrocytoma, under which tumors are graded from I (least advanced disease—best prognosis) to IV (most advanced disease—worst prognosis).
By location
Gliomas can be classified according to whether they are above or below a membrane in the brain called the
(below).- The supratentorial is above the tentorium, in the cerebrum, and mostly found in adults (70%).[55]
- The infratentorial is below the tentorium, in the cerebellum, and mostly found in children (70%).[55]
- The pontine tumors are located in the pons of the brainstem. The brainstem has three parts (pons, midbrain, and medulla); the pons controls critical functions such as breathing, making surgery on these extremely dangerous.[56]
Integrated diagnosis
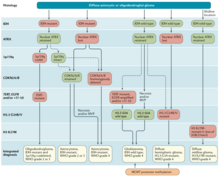
The modern approach to the diagnosis of diffuse gliomas takes mainly the histopathology and molecular profile into account.[57] Tissue specimens obtained through biopsy sampling in patients with diffuse gliomas are routinely assessed by immunohistochemistry for the presence of R132H-mutant IDH1 and loss of nuclear ATRX.[57] In patients aged >55 years with a histologically typical glioblastoma, without a pre-existing lower grade glioma, with a non-midline tumor location and with retained nuclear ATRX expression, immunohistochemical negativity for IDH1 R132H suffices for the classification as IDH-wild-type glioblastoma.[57] In all other instances of diffuse gliomas, a lack of IDH1 R132H immunopositivity should be followed by IDH1 and IDH2 DNA sequencing to detect or exclude the presence of non-canonical mutations.[57] IDH-wild-type diffuse astrocytic gliomas without microvascular proliferation or necrosis should be tested for EGFR amplification, TERT promoter mutation and a +7/–10 cytogenetic signature as molecular characteristics of IDH-wild-type glioblastomas.[57] In addition, the presence of histone H3.3 G34R/V mutations should be assessed by immunohistochemistry or DNA sequencing to identify H3.3 G34-mutant diffuse hemispheric gliomas, in particular in young patients with IDH-wild-type gliomas (such as those <50 years of age with nuclear ATRX loss in tumour cells).[57] Diffuse gliomas of the thalamus, brainstem or spinal cord should be evaluated for histone H3 K27M mutations and loss of nuclear K27-trimethylated histone H3 (H3K27me3) to identify H3 K27M-mutant diffuse midline gliomas.[57]
Treatment


Treatment for brain gliomas depends on the location, the cell type, and the grade of malignancy. Current treatment options include surgical removal, radiation (radiation therapy), and chemotherapy. In some cases, tumour treating fields (alternating electric field therapy), a recently developed technology, may be used.[58] Often, treatment is a combined approach, using surgery, radiation therapy, and chemotherapy. For many, treatment consists of just surgery, or even "watchful waiting" (waiting to see when an intervention is justified due to tumour progression). Doctors carefully balance the specifics of the patient's tumour and the downsides of intervention, since there can be significant side effects from medical intervention, despite recent attempts to predict outcomes have been proposed. [59]
Radiation and chemotherapy remain the mainstays of treatment beyond surgery. Radiation therapy is delivered in the form of external beam radiation or the stereotactic approach using radiosurgery. Temozolomide is a common chemotherapy drug which can be administered easily in an outpatient setting and is able to cross the blood–brain barrier effectively.
There are a wide variety of novel treatments currently being tested in clinical trials, ranging from IDH inhibitors like Ivosidenib, to the recently approved Dendritic cell-based cancer vaccine approach.[60] Treatment using immunotherapy is another promising research path that may help treat glioma in the near future.[61][62] Experimental therapies like oncolytic viruses have shown potential therapeutic benefits in clinical trials (but have not been approved for use in non-experimental settings).[63]
Refractory disease
For recurrent high-grade glioblastoma, recent studies have taken advantage of
Relative effectiveness
A 2017 meta-analysis compared surgical resection versus biopsy as the initial surgical management option for a person with a low-grade glioma.[65] Results show the evidence is insufficient to make a reliable decision.[65] The relative effectiveness of surgical resection compared to biopsy for people with malignant glioma (high-grade) is unknown.[66]
For high-grade gliomas, a 2003 meta-analysis compared radiotherapy with radiotherapy and chemotherapy. It showed a small but clear improvement from using chemotherapy with radiotherapy.[67] A 2019 meta-analysis suggested that for people with less aggressive gliomas, radiotherapy may increase the risk of long term neurocognitive side effects.[68] Whilst, evidence is uncertain on whether there are long term neurocognitive side effects associated with chemoradiotherapy.[68]
Temozolomide is effective for treating Glioblastoma Multiforme (GBM) compared to radiotherapy alone.
A mutational analysis of 23 initial low-grade gliomas and recurrent tumors from the same patients has challenged the benefits and usage of Temozolomide. The study showed that when lower-grade brain tumors of patients are removed and patients are further treated with Temozolomide, 6 out of 10 times the recurrent tumors were more aggressive and acquired alternative and more mutations.[70] As one of the last authors, Costello, stated "They had a 20- to 50-fold increase in the number of mutations. A patient who received surgery alone who might have had 50 mutations in the initial tumor and 60 in the recurrence. But patients who received TMZ might have 2,000 mutations in the recurrence."[71] Further, new mutations were verified to carry known signatures of Temozolomide induced mutations. The research suggests that Temozolomide for the treatment of certain brain tumors should be thoroughly thought. Unjudicious usage of Temozolomide might lower the prognosis of the patients further, or increase their burden. Further understanding of the mechanisms of Temozolomide induced mutations and novel combination approaches could be promising.[medical citation needed]
Prognosis
This section needs to be updated. (September 2020) |
Prognosis of gliomas is given in relation to what grade (as scored by the World Health Organization system) of tumour the patient presents with. Typically, any tumour presenting as above WHO grade I (i.e. a malignant tumour as opposed to a benign tumour) will have a prognosis resulting in eventual death, varying from years (WHO grade II/III) to months (WHO grade IV).[46][72] Prognosis can also be given based on cellular subtype, which may also impact prognosis.
Low grade
For low-grade tumors, the prognosis is somewhat more optimistic. Patients diagnosed with a low-grade glioma are 17 times as likely to die as matched patients in the general population.[73] The age-standardized 10-year relative survival rate was 47% according to research in 2014.[73] One study reported that low-grade oligodendroglioma patients have a median survival of 11.6 years;[74] another reported a median survival of 16.7 years.[75] Unfortunately, approximately 70% of low-grade (WHO grade-II) will progress to high-grade tumours within 5–10 years[46] Grade II gliomas, despite often being labeled as benign, are considered a uniformly fatal illness.[76]
High grade
This group comprises
Postoperative conventional daily radiotherapy improves survival for adults with good functional well‐being and high grade glioma compared to no postoperative radiotherapy. Hypofractionated radiation therapy has similar efficacy for survival as compared to conventional radiotherapy, particularly for individuals aged 60 and older with glioblastoma.[78]
Diffuse midline glioma
Diffuse midline glioma (DMG), also known as diffuse intrinsic pontine glioma (DIPG), primarily affects children, usually between the ages of 5 and 7.[79] The median survival time with DIPG is under 12 months.[80] Surgery to attempt tumour removal is usually not possible or advisable for pontine gliomas. By their very nature, these tumours invade diffusely throughout the brain stem, growing between normal nerve cells. Aggressive surgery would cause severe damage to neural structures vital for arm and leg movement, eye movement, swallowing, breathing, and even consciousness.[81][82][unreliable medical source?] Trials of drug candidates have been unsuccessful.[83] The disease is primarily treated with radiation therapy alone.[medical citation needed]
IDH1 and IDH2-mutated glioma
Patients with glioma carrying mutations in either IDH1 or IDH2 have a relatively favorable survival, compared with patients with glioma with wild-type IDH1/2 genes. In WHO grade III glioma, IDH1/2-mutated glioma have a median prognosis of ~3.5 years, whereas IDH1/2 wild-type glioma perform poor with a median overall survival of c. 1.5 years.[39][84]
References
- S2CID 20356267.
- PMID 23238284.
- S2CID 34392868.
- ^ PRETEST pediatrics p. 224
- )
- S2CID 2972931.
- ^ a b Smoll NR, Brady Z, Scurrah KJ, Lee C, Berrington de González A, Mathews JD. Computed tomography scan radiation and brain cancer incidence. Neuro-Oncology. 2023 Jan 14;https://doi.org/10.1093/neuonc/noad012
- ^ Smoll NR, Brady Z, Scurrah K, Mathews JD. Exposure to ionizing radiation and brain cancer incidence: The Life Span Study cohort. Cancer Epidemiology. 2016 Jun;42:60–5.
- PMID 22182218.
- .
- ^ "IARC classifies radiofrequency electromagnetic fields as possibly carcinogenic to humans" (PDF) (Press release). IARC. 31 May 2011.
- ^ "Cell Phones and Cancer Risk". National Cancer Institute. Retrieved 29 May 2016.
- ^ "Cell Phones and Cancer Risk (References)". National Cancer Institute. Retrieved 29 May 2016.
- ^ "Wireless Devices and Health Concerns". Federal Communications Commission (FCC). 26 May 2011. Retrieved 29 May 2016.
- ^ "Media Telebriefing: NTP Cell Phone Radiofrequency Radiation Study: Partial Release of Findings". niehs.nih.gov (Press release). Retrieved 29 May 2016.
- bioRxiv 10.1101/055699.
- ^ Storrs C (27 May 2016). "Cell phone radiation increases cancers in rats, but should we worry?". CNN. Retrieved 29 May 2016.
- S2CID 22444291.
- S2CID 6952978.
- PMID 22319219.
- PMID 27402152.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - PMID 28034905.
- PMID 34503287.
- PMID 35781883.
- PMID 24842956.
- PMID 24500421.
- ^ ISBN 978-953-51-1114-6, retrieved 3 July 2022
- PMID 17616978.
- PMID 18704159.
- PMID 22672670.
- ^ PMID 20150365.
- PMID 22570426.
- PMID 22570426.
- PMID 19626585.
- PMID 26967246.
- PMID 9096356.
- PMID 16728433.
- PMID 11850397.
- ^ PMID 24880135.
- ^ PMID 23532369.
- PMID 22824796.
- PMID 10411935.
- PMID 24834258.
- PMID 26363012.
- PMID 19246647.
- ^ PMID 11390353.
- S2CID 222413763.
- PMID 1349850.
- PMID 19228619.
- ^ "Gliomas". Johns Hopkins Medicine Health Library. Retrieved 19 April 2017.
- PMID 27157931.
- OCLC 951745876.[page needed]
- ^ Neuro-Oncology, Volume 25, Issue Supplement_1, June 2023, Pages i38–i39, https://doi.org/10.1093/neuonc/noad073.152
- PMID 33742279.
- ^ PMID 29099032.
- PMID 34195093.
- ^ PMID 33293629.)
{{cite journal}}: CS1 maint: multiple names: authors list (link
"This article is licensed under a Creative Commons Attribution 4.0 International License" - PMID 32478924.
- .
- ^ PMID 23633341.
- S2CID 22635893.
- PMID 26070552.
- PMID 34372501.
- S2CID 6164155.
- ^ PMID 28447767.
- PMID 31169915.
- PMID 12519620.
- ^ PMID 31425631.
- PMID 28744879.
- PMID 24336570.
- ^ "Recurrent Brain Cancers Follow Distinctive Genetic Paths". University of California Santa Cruz. University of California San Francisco. 17 December 2013. Retrieved 17 June 2015.
- PMID 22043865.
- ^ PMID 22773277.
- PMID 15977639.
- S2CID 26335770.
- PMID 25552286.
- PMID 22270850.
- PMID 32437039.
- ^ "Patients & Families: Basic Facts". DIPG Registry. Archived from the original on 2 May 2014. Retrieved 1 May 2014.
- S2CID 207491201.
- S2CID 25562391.
- PMID 16525181.
- PMID 21764221.
- PMID 24510240.
