Gliosis
| Gliosis | |
|---|---|
| Specialty | Neurology |
Gliosis is a nonspecific reactive change of
The process of gliosis involves a series of cellular and molecular events that occur over several days.
Gliosis has historically been given a negative connotation due to its appearance in many CNS diseases and the inhibition of axonal regeneration caused by glial scar formation. However, gliosis has been shown to have both beneficial and detrimental effects, and the balance between these is due to a complex array of factors and molecular signaling mechanisms, which affect the reaction of all glial cell types.[citation needed]
Astrogliosis
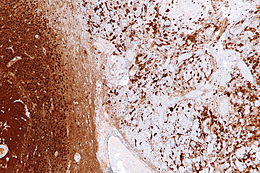
Reactive astrogliosis is the most common form of gliosis and involves the
Modulation of astrogliosis
Changes in astrogliosis are regulated in a context-dependent fashion, and the signaling events which dictate these changes may modify both their nature and severity.
Effects of astrogliosis
Although astrogliosis has traditionally been viewed as a negative response inhibitory to axonal regeneration, the process is highly conserved, suggesting it has important benefits beyond its detrimental effects.[4] Generally, the effects of astrogliosis vary with the context of the initial CNS insult and also with time after the injury. A few of the most important effects of astrogliosis are listed below.
Beneficial effects
- Neuroprotective effects – Reactive astrocytes release neurotrophic factors, such as glial cell-derived neurotrophic factor (GDNF), which protects against programmed cell death
- Maintenance of the extracellular environment – Astrocytes are responsible for the uptake of glutamate, which restricts excitotoxicity to neurons and other cell types, as well as the elimination of free radicals[4][7]
- Release of anti-inflammatory molecules
- Restoration of blood brain barrier function
- Seclusion of the injury site and containment of infection from healthy tissue[5]
Detrimental effects
- Restriction of axon regeneration – In cases of glial scar formation, reactive astrocytes enmesh the lesion site and deposit an inhibitory extracellular matrix consisting of chondroitin sulfate proteoglycans. The dense structure of these proteins is a physically and chemically inhibitory barrier to axon regeneration and the reestablishment of axon connections.[4][5]
- Secretion of TNF-α.[4]
- Release of excitotoxic glutamate[5]
- Hindrance of functional recovery and worsening of
Microgliosis
Microglia, another type of glial cell, act as macrophage-like cells in the CNS when activated. Unlike other glial cell types, microglia are extremely sensitive to even small changes in the cellular environment, allowing for a rapid response to inflammatory signals and prompt destruction of infectious agents before sensitive neural tissue can be damaged.[4] Due to their fast response time, microgliosis, or the activation of microglia, is commonly the first observed stage of gliosis.
Microgliosis following a CNS insult most commonly involves the development of an altered cellular morphology, specifically the enlargement of cellular processes.[2] The microglial immunological surface receptor CR3 is also upregulated within 24 hours after the initial injury. Within the first week following the injury, microglia begin to proliferate abnormally and while doing so exhibit several immunophenotypic changes, particularly an increased expression of MHC antigens.[2] The population of activated microglia at the site of a CNS injury includes not only endogenous microglia of the CNS but also exogeneous perivascular cells originating in the bone marrow that migrate to the area and transform into microglia to supplement the microgliosis response.[8]
While in their activated state, microglia may serve a variety of beneficial functions. For example, active microglia are the primary effectors of
Neural modulation of microgliosis
Microglial regulation of astrogliosis
Although the molecular triggers of gliosis, including both astrogliosis and microgliosis, are not fully understood, in vitro studies have indicated that activated microglia have an important role in initiating and modulating astrogliosis.
Response of oligodendrocytes
Triggers of gliosis
In general after any CNS insult, gliosis begins after the blood brain barrier is disrupted, allowing non-CNS molecules, such as blood and
In CNS injury and disease
Gliosis is the universal response of the CNS to tissue injury and occurs as a result of many acute conditions such as trauma,
CNS trauma
Acute trauma to the brain or spinal cord results in gliosis, most often in its severe form with the development of a glial scar. Different locations around the lesion site may exhibit different severities of gliosis; for example, a glial scar at the location of damaged tissue may be surrounded by areas with less severe astrocyte proliferation or hypertrophy. Diffuse traumatic injury can result in diffuse or more moderate gliosis without scar formation. In such cases, gliosis may also be reversible. In all instances of gliosis resulting from CNS trauma, the long-term clinical outcome is highly dependent on the degree of astrogliosis and scar formation.[14]
Multiple sclerosis and autoimmune inflammatory disorders
Gliosis is a prominent feature of many autoimmune inflammatory disorders, notably multiple sclerosis, in which demyelinated plaques are surrounded by reactive astrocytes. These astrocytes often exhibit extreme hypertrophy and multiple distinct nuclei, and their production of pro-inflammatory molecules has been implicated in several inflammatory disorders.[14] Cytokines produced by both active astrocytes and microglia in inflammatory conditions may contribute to myelin damage and may alter blood-brain barrier permeability, allowing the migration of lymphocytes into the CNS and heightening the autoimmune attack.[10]
Retinal gliosis
In vertebrates, the retina contains Müller cells, a type of glia not found elsewhere in the CNS. Upon retinal injury, gliosis of these cells occurs, functioning to repair damage, but often having harmful consequences in the process, worsening some of the diseases or problems that initially trigger it.[16] Reactive gliosis in the retina can have detrimental effects on vision; in particular, the production of proteases by astrocytes causes widespread death of retinal ganglion cells. A 2011 study compared the effects of two glial toxins, AAA and Neurostatin, on retinal gliosis in mice. AAA did not inhibit the production of protease by astrocytes, and so did not prevent ganglion cell apoptosis. However, Neurostatin successfully inhibited activation of astrocytes, in turn decreasing retinal ganglion cell death significantly. Neurostatin is also effective in the inhibition of other glial cells, and may be an area of interest in the treatment of degenerative diseases such as glaucoma.[17]
Massive retinal gliosis (MRG) is a phenomenon in which the retina is completely replaced by proliferation of glial cells, causing deterioration of vision and even blindness in some cases. Sometimes mistaken for an intraocular tumor, MRG can arise from a neurodegenerative disease, congenital defect, or from trauma to the eyeball, sometimes appearing years after such an incident.[18][19]
Alzheimer's disease
Gliosis has long been known as a characteristic of Alzheimer's Disease (AD), although its exact role in the disease remains unknown. Gliosis and glial scarring occur in areas surrounding the
Amyotrophic lateral sclerosis
Amyotrophic lateral sclerosis (ALS) is a debilitating disease involving the degeneration of motor neurons in the CNS. Reactive astrocytes have been implicated in this condition through either a loss of their neuroprotective ability or through the gain of neurotoxic effects. Late stages of ALS are also characterized by significant astrogliosis and astrocyte proliferation around areas of degeneration.[7]
Potential therapeutic targets in gliosis
The implications of gliosis in various neuropathologies and injury conditions has led to the investigation of various therapeutic routes which would regulate specific aspects of gliosis in order to improve clinical outcomes for both CNS trauma and a wide range of neurological disorders. Because gliosis is a dynamic process which involves a spectrum of changes depending on the type and severity of the initial insult, to date, no single molecular target has been identified which could improve healing in all injury contexts. Rather, therapeutic strategies for minimizing the contribution of astrogliosis to CNS pathologies must be designed to target specific molecular pathways and responses. One promising therapeutic mechanism is the use of
See also
- Bergmann gliosis
References
- ^ S2CID 20878075.
- ^ S2CID 44441542.
- ^ S2CID 12210165.
- ^ PMID 20148316.
- ^ PMID 19782411.
- ^ PMID 14735117.
- ^ PMID 20880504.
- S2CID 37387247.
- S2CID 21457903.
- ^ S2CID 1292442.
- PMID 19847447.
- S2CID 11800813.
- S2CID 4041686.
- ^ PMID 20012068.
- ^ PMID 20880511.
- S2CID 8803821.
- PMID 21483783.
- PMID 21586853.
- ^ Ali, Zafar; Atique, Muhammed (2012). "Massive Retinal Gliosis: A Rare Entity". The Annals of Pakistan Institute of Medical Sciences. 8 (2): 152–3.
- PMID 19765548.
