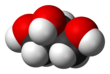
Glycerol

| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Propane-1,2,3-triol[1] | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.000.263 | ||
| E number | E422 (thickeners, ...) | ||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H8O3 | |||
| Molar mass | 92.094 g·mol−1 | ||
| Appearance | Colorless hygroscopic liquid
| ||
| Odor | Odorless | ||
| Density | 1.261 g/cm3 | ||
| Melting point | 17.8 °C (64.0 °F; 290.9 K) | ||
| Boiling point | 290 °C (554 °F; 563 K)[5] | ||
| miscible[2] | |||
| log P | −2.32[3] | ||
| Vapor pressure | 0.003 mmHg (0.40 Pa) at 50 °C[2] | ||
| −57.06×10−6 cm3/mol | |||
Refractive index (nD)
|
1.4746 | ||
| Viscosity | 1.412 Pa·s (20 °C)[4] | ||
| Pharmacology | |||
| A06AG04 (WHO) A06AX01 (WHO), QA16QA03 (WHO) | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 160 °C (320 °F; 433 K) (closed cup) 176 °C (349 °F; 449 K) (open cup) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 15 mg/m3 (total) TWA 5 mg/m3 (resp)[2] | ||
REL (Recommended)
|
None established[2] | ||
IDLH (Immediate danger) |
N.D.[2] | ||
| Safety data sheet (SDS) | JT Baker ver. 2008 archive | ||
| Supplementary data page | |||
| Glycerol (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Glycerol (
Structure
Although achiral, glycerol is prochiral with respect to reactions of one of the two primary alcohols. Thus, in substituted derivatives, the stereospecific numbering labels the molecule with a sn- prefix before the stem name of the molecule.[8][9][10]
Production
Glycerol is generally obtained from plant and animal sources where it occurs in
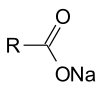
of these triglycerides produces glycerol as well as the fatty acid derivative:Triglycerides can be saponified with sodium hydroxide to give glycerol and fatty sodium salt or soap.
Typical plant sources include
Glycerol from triglycerides is produced on a large scale, but the crude product is of variable quality, with a low selling price of as low as US$0.02–0.05 per kilogram in 2011.[12] It can be purified, but the process is expensive. Some glycerol is burned for energy, but its heat value is low.[13]
Crude glycerol from the hydrolysis of triglycerides can be purified by treatment with activated carbon to remove organic impurities, alkali to remove unreacted glycerol esters, and ion exchange to remove salts. High purity glycerol (greater than 99.5%) is obtained by multi-step distillation; a vacuum chamber is necessary due to its high boiling point (290 °C).[7]
Synthetic glycerol
Because of the large-scale production of
Although usually not cost-effective because so much is produced from processing of fats, glycerol can be produced by various routes. During
Applications
Food industry
In food and beverages, glycerol serves as a
As used in foods, glycerol is categorized by the U.S.
It is also recommended as an additive when using polyol sweeteners such as erythritol and xylitol which have a cooling effect, due to its heating effect in the mouth, if the cooling effect is not wanted.[16]
Excessive consumption by children can lead to glycerol intoxication.[17] Symptoms of intoxication include hypoglycemia, nausea and a loss of consciousness (syncope). While intoxication as a result of excessive glycerol consumption is rare and its symptoms generally mild, occasional reports of hospitalization have occurred. In the United Kingdom in August 2023, manufacturers of syrup used in slush ice drinks were advised to reduce the amount of glycerol in their formulations by the Food Standards Agency to reduce the risk of intoxication.[18]
Medical, pharmaceutical and personal care applications




Glycerol is used in
Glycerol is a component of
Taken rectally, glycerol functions as a
Taken orally (often mixed with fruit juice to reduce its sweet taste), glycerol can cause a rapid, temporary decrease in the internal pressure of the eye. This can be useful for the initial emergency treatment of severely elevated eye pressure.[24]
In 2017, researchers showed that the probiotic Limosilactobacillus reuteri bacteria can be supplemented with glycerol to enhance its production of antimicrobial substances in the human gut. This was confirmed to be as effective as the antibiotic vancomycin at inhibiting Clostridioides difficile infection without having a significant effect on the overall microbial composition of the gut.[25]
Glycerol has also been incorporated as a component of bio-ink formulations in the field of bioprinting.[26] The glycerol content acts to add viscosity to the bio-ink without adding large protein, saccharide, or glycoprotein molecules.
Botanical extracts
When utilized in "tincture" method extractions, specifically as a 10% solution, glycerol prevents tannins from precipitating in ethanol extracts of plants (
When used as a primary "true" alcohol-free botanical extraction solvent in non-tincture based methodologies, glycerol has been shown to possess a high degree of extractive versatility for botanicals including removal of numerous constituents and complex compounds, with an extractive power that can rival that of alcohol and water–alcohol solutions.
Electronic cigarette liquid
Glycerin, along with
Antifreeze
Like
Glycerol was historically used as an anti-freeze for automotive applications before being replaced by ethylene glycol, which has a lower freezing point. While the minimum freezing point of a glycerol-water mixture is higher than an ethylene glycol-water mixture, glycerol is not toxic and is being re-examined for use in automotive applications.[34][35]
In the laboratory, glycerol is a common component of solvents for
Chemical intermediate
Glycerol is used to produce a variety of useful derivatives.
Trifunctional
Oxidation of glycerol affords mesoxalic acid.[37] Dehydrating glycerol affords hydroxyacetone.
Chlorination of glycerol gives the
- HOCH(CH2OH)2 + HCl → HOCH(CH2Cl)(CH2OH) + H2O
The same compound can be produced by hydrolysis of epichlorohydrin.[38]
Vibration damping
Glycerol is used as fill for
Niche uses
Entertainment industry
Glycerol is used by set decorators when filming scenes involving water to prevent an area meant to look wet from drying out too quickly.[40]
Glycerine is also used in the generation of theatrical smoke and fog as a component of the fluid used in fog machines as a replacement for glycol, which has been shown to be an irritant if exposure is prolonged.
Ultrasonic couplant
Glycerol can be sometimes used as replacement for water in ultrasonic testing, as it has favourably higher acoustic impedance (2.42 MRayl versus 1.483 MRayl for water) while being relatively safe, non-toxic, non-corrosive and relatively low cost.[41]
Internal combustion fuel
Glycerol is also used to power diesel generators supplying electricity for the
Research on additional uses
Research continues into potential
- Hydrogen gas production.[44]
- Glycerine acetate is a potential fuel additive.[45]
- Additive for starch thermoplastic.[46][47]
- Conversion to various other chemicals:
Metabolism
Glycerol is a precursor for synthesis of
Glycerol is mainly metabolized in the liver. Glycerol injections can be used as a simple test for liver damage, as its rate of absorption by the liver is considered an accurate measure of liver health. Glycerol metabolism is reduced in both cirrhosis and fatty liver disease.[55][56]
Blood glycerol levels are highly elevated during diabetes, and is believed to be the cause of reduced fertility in patients who suffer from diabetes and metabolic syndrome. Blood glycerol levels in diabetic patients average three times higher than healthy controls. Direct glycerol treatment of testes has been found to cause significant long-term reduction in sperm count. Further testing on this subject was abandoned due to the unexpected results, as this was not the goal of the experiment.[57]
Circulating glycerol does not
Before glycerol can enter the pathway of glycolysis or gluconeogenesis (depending on physiological conditions), it must be converted to their intermediate glyceraldehyde 3-phosphate in the following steps:
The enzyme glycerol kinase is present mainly in the liver and kidneys, but also in other body tissues, including muscle and brain.[58][59][60] In adipose tissue, glycerol 3-phosphate is obtained from dihydroxyacetone phosphate with the enzyme glycerol-3-phosphate dehydrogenase.
Glycerol has very low toxicity when ingested; its
Historical cases of contamination with diethylene glycol
On 4 May 2007, the FDA advised all U.S. makers of medicines to test all batches of glycerol for diethylene glycol contamination.[63] This followed an occurrence of hundreds of fatal poisonings in Panama resulting from a falsified import customs declaration by Panamanian import/export firm Aduanas Javier de Gracia Express, S. A. The cheaper diethylene glycol was relabeled as the more expensive glycerol.[64][65] Between 1990 and 1998, incidents of DEG poisoning reportedly occurred in Argentina, Bangladesh, India, and Nigeria, and resulted in hundreds of deaths. In 1937, more than one hundred people died in the United States after ingesting DEG-contaminated elixir sulfanilamide, a drug used to treat infections.[66]
Etymology
The origin of the gly- and glu- prefixes for glycols and sugars is from Ancient Greek γλυκύς glukus which means sweet.[67] Name glycérine was coined ca. 1811 by Michel Eugène Chevreul to denote what was previously called "sweet principle of fat" by its discoverer Carl Wilhelm Scheele. It was borrowed into English ca. 1838 and in the 20th c. displaced by 1872 term glycerol featuring an alcohols' suffix -ol.
Properties
Table of thermal and physical properties of saturated liquid glycerin:[68][69]
Temperature (°C) Density (kg/m3) Specific heat (kJ/kg·K) Kinematic viscosity(m2/s)Conductivity(W/m·K)Thermal diffusivity (m2/s) Prandtl number Bulk modulus (K−1) 0 1276.03 2.261 8.31×10−3 0.282 9.83×10−8 84700 4.7×10−4 10 1270.11 2.319 3.00×10−3 0.284 9.65×10−8 31000 4.7×10−4 20 1264.02 2.386 1.18×10−3 0.286 9.47×10−8 12500 4.8×10−4 30 1258.09 2.445 5.00×10−4 0.286 9.29×10−8 5380 4.8×10−4 40 1252.01 2.512 2.20×10−4 0.286 9.14×10−8 2450 4.9×10−4 50 1244.96 2.583 1.50×10−4 0.287 8.93×10−8 1630 5.0×10−4
See also
- Dioxalin
- Epichlorohydrin
- Nitroglycerin
- Oleochemicals
- Soapmaking
- Solketal
- Transesterification
References
- ISBN 978-0-85404-182-4.
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0302". National Institute for Occupational Safety and Health (NIOSH).
- ^ "glycerin_msds". Archived from the original on 8 March 2020. Retrieved 7 May 2018.
- .
- ^ Lide, D. R., ed. (1994). CRC Handbook of Data on Organic Compounds (3rd ed.). Boca Raton, FL: CRC Press. p. 4386.
- ^ "glycerol – Definition of glycerol in English by Oxford Dictionaries". Oxford Dictionaries – English. Archived from the original on 21 June 2016. Retrieved 21 February 2022.
- ^ ISBN 3527306730.
- PMID 13714619.
- PMID 6078528.
- PMID 29301208.
- ^ Nilles, Dave (2005). "A Glycerin Factor". Biodiesel Magazine. Archived from the original on 8 November 2007. Retrieved 21 February 2022.
- .
- ^ Sims, Bryan (25 October 2011). "Clearing the way for byproduct quality: why quality for glycerin is just as important for biodiesel". Biodiesel Magazine. Archived from the original on 29 April 2021. Retrieved 21 February 2022.
- .
- ^ Gouin, Francis R. (1994). "Preserving flowers and leaves" (PDF). Maryland Cooperative Extension Fact Sheet. 556: 1–6. Archived (PDF) from the original on 21 January 2018. Retrieved 20 January 2018.
- ^ Nikolov, Ivan (20 April 2014). "Functional Food Design Rules". Archived from the original on 30 April 2021. Retrieved 21 February 2022.
- ^ Burrell, Chloe (2 June 2023). "Perth and Kinross parents warned as 'intoxicated' kids hospitalised by slushy drinks". The Courier. Retrieved 3 June 2023.
- ^ "'Not suitable for under-4s': New industry guidance issued on glycerol in slush-ice drinks". Food Standards Agency. Retrieved 11 August 2023.
- ISBN 9781481659666.
- ISBN 9780702069130.
- ^ "Glycerin Enema". Drugs.com. Archived from the original on 6 November 2012. Retrieved 17 November 2012.
- ^ "glycerin enema". NCI Drug Dictionary. National Cancer Institute. 2 February 2011. Archived from the original on 2 May 2019. Retrieved 2 May 2019.
- S2CID 32872781
- ^ "Glycerin (Oral Route)". Mayo Foundation for Medical Education and Research. Archived from the original on 27 November 2012. Retrieved 17 November 2012.
- PMID 28760934.
- S2CID 9073831.
- JSTOR 3624347.
- ^ Does alcohol belong in herbal tinctures? Archived 12 October 2007 at the Wayback Machine newhope.com
- ^ "Glycerine: An Overview" (PDF). aciscience.org. The Soap and Detergent Association. 1990. Archived (PDF) from the original on 1 June 2019.
- ^ Lawrie, James W. (1928) Glycerol and the glycols – production, properties and analysis. The Chemical Catalog Company, Inc., New York, NY.
- ^ Leffingwell, Georgia and Lesser, Miton (1945) Glycerin – its industrial and commercial applications. Chemical Publishing Co., Brooklyn, NY.[page needed]
- ^ The manufacture of glycerol – Vol. III (1956). The Technical Press, LTD., London.[page needed]
- ISBN 9780124059177. Archivedfrom the original on 16 April 2021. Retrieved 16 August 2017.
- .
- ^ Proposed ASTM Engine Coolant Standards Focus on Glycerin Archived 14 September 2017 at the Wayback Machine. Astmnewsroom.org. Retrieved on 15 August 2012
- PMID 35625132.
- .
- PMID 26196761.
- ^ Pneumatic Systems: Principles and Maintenance by S. R. Majumdar. McGraw-Hill, 2006, p. 74 [ISBN missing]
- ^ Chemicals in Film reagent.co.uk
- ^ Acoustic Properties for Liquids Archived 27 October 2016 at the Wayback Machine nde-ed.org
- ^ Formula E uses pollution-free glycerine to charge cars. fiaformulae.com. 13 September 2014
- doi:10.1002/ep.10225.
- .
- .
- ISSN 0264-1275.
- .
- ^ "Dow achieves another major milestone in its quest for sustainable chemistries" (Press release). Dow Chemical Company. 15 March 2007. Archived from the original on 16 September 2009. Retrieved 13 July 2007.
- doi:10.1039/b506285c.
- PMID 16797980.
- S2CID 246853148.
- PMID 17532205.
- ^ "Engineers Find Way To Make Ethanol, Valuable Chemicals From Waste Glycerin". ScienceDaily (Press release). 27 June 2007.
- ^ "Dow Epoxy advances glycerine-to-epichlorohydrin and liquid epoxy resins projects by choosing Shanghai site" (Press release). Dow Chemical Company. 26 March 2007. Archived from the original on 8 December 2011. Retrieved 21 February 2022.
- ^ Glycerol clearance in alcoholic liver disease. Gut (British Society of Gastroenterology). 1982 Apr; 23(4): 257–264. D G Johnston, K G Alberti, R Wright, P G Blain
- ^ "Fatty liver disrupts glycerol metabolism in gluconeogenic and lipogenic pathways in humans". September 2018 The Journal of Lipid Research, 59, 1685–1694. Jeffrey D. Browning, Eunsook S. Jin1, Rebecca E. Murphy, and Craig R. Malloy
- ^ Molecular Human Reproduction, Volume 23, Issue 11, November 2017, pp. 725–737
- PMID 183753.
- PMID 5801671.
- from the original on 21 February 2022. Retrieved 27 August 2019.
- PMID 28882640.
- .
- ^ "FDA Advises Manufacturers to Test Glycerin for Possible Contamination". U.S. Food and Drug Administration. 4 May 2007. Archived from the original on 7 May 2007. Retrieved 8 May 2007.
- ^ Walt Bogdanich (6 May 2007). "From China to Panama, a Trail of Poisoned Medicine". The New York Times. Archived from the original on 26 September 2015. Retrieved 8 May 2007.
- ^ "10 Biggest Medical Scandals in History". 20 February 2013. Archived from the original on 8 January 2022. Retrieved 21 February 2022.
- from the original on 21 February 2022. Retrieved 25 December 2020.
- ^ glyco- Archived 30 April 2021 at the Wayback Machine, dictionary.com
- ISBN 9780072406559.
- ISBN 9780471457282.)
{{cite book}}: CS1 maint: numeric names: authors list (link