Glycoside

In
In formal terms, a glycoside is any molecule in which a sugar group is bonded through its
Many authors require in addition that the sugar be bonded to a non-sugar for the molecule to qualify as a glycoside, thus excluding polysaccharides. The sugar group is then known as the glycone and the non-sugar group as the aglycone or genin part of the glycoside. The glycone can consist of a single sugar group (monosaccharide), two sugar groups (disaccharide), or several sugar groups (oligosaccharide).
The first glycoside ever identified was
Related compounds
Molecules containing an N-glycosidic bond are known as
Chemistry
Much of the chemistry of glycosides is explained in the article on
There are many ways to chemically synthesize glycosidic bonds.
Classification
This section needs additional citations for verification. (January 2021) |
Glycosides can be classified by the glycone, by the type of glycosidic bond, and by the aglycone.
By glycone/presence of sugar
If the glycone group of a glycoside is glucose, then the molecule is a glucoside; if it is fructose, then the molecule is a fructoside; if it is glucuronic acid, then the molecule is a glucuronide; etc. In the body, toxic substances are often bonded to glucuronic acid to increase their water solubility; the resulting glucuronides are then excreted. Compounds can also be generally defined based on the class of glycone; for example, biosides are glycosides with a disaccharide (biose) glycone.
By type of glycosidic bond
Depending on whether the glycosidic bond lies "below" or "above" the plane of the cyclic sugar molecule, glycosides are classified as α-glycosides or β-glycosides. Some enzymes such as
There are four type of linkages present between glycone and aglycone:
- C-linkage/glycosidic bond, "nonhydrolysable by acids or enzymes"
- O-linkage/glycosidic bond
- N-linkage/glycosidic bond
- S-linkage/glycosidic bond
By aglycone
Glycosides are also classified according to the chemical nature of the aglycone. For purposes of biochemistry and pharmacology, this is the most useful classification.
Alcoholic glycosides
An example of an alcoholic glycoside is salicin, which is found in the genus Salix. Salicin is converted in the body into salicylic acid, which is closely related to aspirin and has analgesic, antipyretic, and anti-inflammatory effects.
Anthraquinone glycosides
These glycosides contain an aglycone group that is a derivative of
species. Anthron and anthranol are reduced forms of anthraquinone.Coumarin glycosides
Here, the aglycone is
Chromone glycosides
In this case, the aglycone is called benzo-gamma-pyrone.
Cyanogenic glycosides

In this case, the aglycone contains a cyanohydrin group. Plants that make cyanogenic glycosides store them in the vacuole, but, if the plant is attacked, they are released and become activated by enzymes in the cytoplasm. These remove the sugar part of the molecule, allowing the cyanohydrin structure to collapse and release toxic hydrogen cyanide. Storing them in inactive forms in the vacuole prevents them from damaging the plant under normal conditions.[6]
Along with playing a role in deterring herbivores, in some plants they control germination,
Examples include
Amygdalin and a synthetic derivative,
Some butterfly species, such as the Dryas iulia and Parnassius smintheus, have evolved to use the cyanogenic glycosides found in their host plants as a form of protection against predators through their unpalatability.[8][9]
Flavonoid glycosides
Here, the aglycone is a flavonoid. Examples of this large group of glycosides include:
- Hesperidin (aglycone: hesperetin, glycone: rutinose)
- Naringin (aglycone: naringenin, glycone: rutinose)
- Rutin (aglycone: quercetin, glycone: rutinose)
- Quercitrin (aglycone: quercetin, glycone: rhamnose)
Among the important effects of flavonoids are their antioxidant effect. They are also known to decrease capillary fragility.
Phenolic glycosides
Here, the aglycone is a simple
Saponins
These compounds give a permanent froth when shaken with water. They also cause
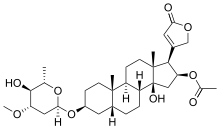
Steroid glycosides (cardiac glycosides)
In these glycosides, the aglycone part is a
Steviol glycosides
These sweet glycosides found in the stevia plant Stevia rebaudiana Bertoni have 40–300 times the sweetness of sucrose. The two primary glycosides, stevioside and rebaudioside A, are used as natural sweeteners in many countries. These glycosides have steviol as the aglycone part. Glucose or rhamnose-glucose combinations are bound to the ends of the aglycone to form the different compounds.
Iridoid glycosides
These contain an iridoid group; e.g. aucubin, geniposidic acid, theviridoside, loganin, catalpol.
Thioglycosides
As the name contains the prefix
See also
- Carbohydrate
- Carbohydrate chemistry
- Chemical glycosylation
- Glycorandomization
- Glycosylation
- Natural products
References
- ISBN 978-0-387-26251-2.
- .
- ISBN 978-0-9678550-9-7.
- ISBN 978-3-527-31528-4.
- ^ Robiquet; Boutron-Charlard (1830). "Nouvelles expériences sur les amandes amères et sur l'huile volatile qu'elles fournissent" [New experiments on bitter almonds and the volatile oil that they provide]. Annales de Chimie et de Physique. 2nd series (in French). 44: 352–382.
- ^ PMID 24579992.
- PMID 25918920.
- S2CID 84261089.
- doi:10.7939/R3VX32. Retrieved 13 November 2017.
- PMID 19208455.
- PMID 20003589.
External links
- Definition of glycosides, from the Gold Book"
- IUPAC naming rules for glycosides
