Grignard reagent
Grignard reagents or Grignard compounds are
Grignard compounds are popular reagents in organic synthesis for creating new carbon–carbon bonds. For example, when reacted with another halogenated compound R'−X' in the presence of a suitable catalyst, they typically yield R−R' and the magnesium halide MgXX' as a byproduct; and the latter is insoluble in the solvents normally used. In this aspect, they are similar to organolithium reagents.
Grignard reagents are rarely isolated as solids. Instead, they are normally handled as solutions in solvents such as diethyl ether or tetrahydrofuran using air-free techniques. Grignard reagents are complex with the magnesium atom bonded to two ether ligands as well as the halide and organyl ligands.
The discovery of the Grignard reaction in 1900 was recognized with the Nobel Prize awarded to Victor Grignard in 1912.
Synthesis
From Mg metal
Traditionally Grignard reagents are prepared by treating an organic halide (normally organobromine) with magnesium metal.
As is common for reactions involving solids and solution, the formation of Grignard reagents is often subject to an
Magnesium
Typically the reaction to form Grignard reagents involves the use of magnesium ribbon. All magnesium is coated with a
- Mg + BrC2H4Br → C2H4 + MgBr2
The amount of Mg consumed by these activating agents is usually insignificant. A small amount of
Specially activated magnesium, such as
"Rieke magnesium" is prepared by a reduction of an anhydrous magnesium chloride with an potassium:
- MgCl2 + 2 K → Mg + 2 KCl
Mechanism
In terms of mechanism, the reaction proceeds through
Mg transfer reaction (halogen–Mg exchange)
An alternative preparation of Grignard reagents involves transfer of Mg from a preformed Grignard reagent to an organic halide. Other organomagnesium reagents are used as well.[11] This method offers the advantage that the Mg transfer tolerates many functional groups. An illustrative reaction involves isopropylmagnesium chloride and aryl bromide or iodides:[12]
- i-PrMgCl + ArCl → i-PrCl + ArMgCl
From alkylzinc compounds (reductive transmetalation)
A further method to synthesize Grignard reagents involves reaction of Mg with an
- AdZnBr + Mg → AdMgBr + Zn
Testing Grignard reagents
Because Grignard reagents are so sensitive to moisture and oxygen, many methods have been developed to test the quality of a batch. Typical tests involve titrations with weighable, anhydrous protic reagents, e.g.
Reactions of Grignard reagents
| Grignard reagent reactions | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Named after | Victor Grignard | ||||||||||
| Reaction type | Coupling reaction | ||||||||||
| Reaction | |||||||||||
| |||||||||||
With carbonyl compounds
Grignard reagents react with a variety of

The most common application of Grignard reagents is the alkylation of aldehydes and ketones, i.e. the Grignard reaction:[16]

Note that the acetal functional group (a protected carbonyl) does not react.
Such reactions usually involve an aqueous acidic workup, though this step is rarely shown in reaction schemes. In cases where the Grignard reagent is adding to an aldehyde or a
Grignard reagents are
Grignard reagents also react with many "carbonyl-like" compounds and other electrophiles:

Reactions as a base
Grignard reagents serve as a base for non-protic substrates (this scheme does not show workup conditions, which typically includes water). Grignard reagents are basic and react with alcohols, phenols, etc. to give alkoxides (ROMgBr). The phenoxide derivative is susceptible to formylation by paraformaldehyde to give salicylaldehyde.[17]
Alkylation of metals and metalloids
Like
- 2 RMgX + CdCl2 → R2Cd + 2 Mg(X)Cl
Schlenk equilibrium
Most Grignard reactions are conducted in ethereal solvents, especially diethyl ether and THF. Grignard reagents react with 1,4-dioxane to give the diorganomagnesium compounds and insoluble coordination polymer MgX2(dioxane)2 and (R = organic group, X = halide):
- 2 RMgX + dioxane ⇌ R2Mg + MgX2(dioxane)2
This reaction exploits the Schlenk equilibrium, driving it toward the right.
Precursors to magnesiates
Grignard reagents react with organolithium compounds to give ate complexes (Bu = butyl):[19]
- BuMgBr + 3 BuLi → LiMgBu3 + BuBr
Coupling with organic halides
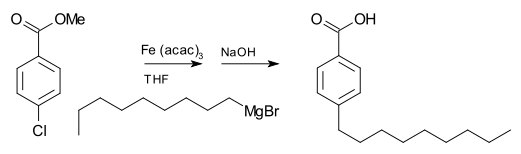
Grignard reagents do not typically react with organic halides, in contrast with their high reactivity with other main group halides. In the presence of metal catalysts, however, Grignard reagents participate in C-C coupling reactions. For example, nonylmagnesium bromide reacts with methyl p-chlorobenzoate to give p-nonylbenzoic acid, in the presence of Tris(acetylacetonato)iron(III) (Fe(acac)3), after workup with NaOH to hydrolyze the ester, shown as follows. Without the Fe(acac)3, the Grignard reagent would attack the ester group over the aryl halide.[20]
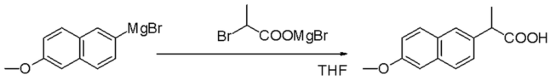
For the coupling of aryl halides with aryl Grignard reagents,
Oxidation
Treatment of a Grignard reagent with oxygen gives the magnesium organoperoxide. Hydrolysis of this material yields hydroperoxides or alcohol. These reactions involve radical intermediates.
The simple oxidation of Grignard reagents to give alcohols is of little practical importance as yields are generally poor. In contrast, two-step sequence via a borane (vide supra) that is subsequently oxidized to the alcohol with hydrogen peroxide is of synthetic utility.
The synthetic utility of Grignard oxidations can be increased by a reaction of Grignard reagents with oxygen in presence of an

Elimination
In the Boord olefin synthesis, the addition of magnesium to certain β-haloethers results in an elimination reaction to the alkene. This reaction can limit the utility of Grignard reactions.

Industrial use
An example of the Grignard reaction is a key step in the (non-stereoselective) industrial production of Tamoxifen[22] (currently used for the treatment of estrogen receptor positive breast cancer in women):[23]

See also
Gallery
-
Magnesium turnings are placed in a flask.
-
Tetrahydrofuran and a small piece of iodine are added.
-
A solution of alkyl bromide is added while heating.
-
After completion of the addition, the mixture is heated for a while.
-
Formation of the Grignard reagent is complete. A small amount of magnesium still remains in the flask.
-
The Grignard reagent thus prepared is cooled to 0°C before the addition of the carbonyl compound. The solution becomes cloudy as the Grignard reagent precipitates out.
-
A solution of carbonyl compound is added to the Grignard reagent.
-
The solution is warmed to room temperature. At this point the reaction is complete.
References
- .
- .
- ISBN 0-8247-9545-8.
- .
- S2CID 92794.
- ISBN 978-0-19-850346-0.
- ISBN 0080538177.
- ISBN 0-471-99908-3.
- ^ Advanced Organic chemistry Part B: Reactions and Synthesis F.A. Carey, R.J. Sundberg 2nd Ed. 1983. Page 435
- ^ Garst, J.F.; Soriaga, M.P. "Grignard reagent Formation", Coord. Chem. Rev. 2004, 248, 623 - 652. doi:10.1016/j.ccr.2004.02.018.
- .
- PMID 14502700.
- PMID 28443859.
- .
- ^ Henry Gilman and R. H. Kirby (1941). "Butyric acid, α-methyl-". Organic Syntheses; Collected Volumes, vol. 1, p. 361.
- . Retrieved November 26, 2009.
- .
- ISBN 978-81-7450-716-7. Archived from the original(PDF) on September 20, 2018. Retrieved March 9, 2019.
- .
- ^ A. Fürstner, A. Leitner, G. Seidel (2004). "4-Nonylbenzoic Acid". Organic Syntheses. 81: 33–42
{{cite journal}}: CS1 maint: multiple names: authors list (link). - PMID 16366543.
- ISBN 0471999083.
- PMID 8242225.
Further reading
- Rakita, Philip E.; Silverman, Gary S., eds. (1996). Handbook of Grignard Reagents. New York, N.Y: Marcel Dekker. ISBN 0-8247-9545-8.
- Mary McHale, "Grignard Reaction," Connexions, http://cnx.org/content/m15245/1.2/. 2007.
- Grignard knowledge: Alkyl coupling chemistry with inexpensive transition metals by Larry J. Westrum, Fine Chemistry November/December 2002, pp. 10–13 [1]
Specialized literature
- Rogers, H. R.; Hill, C. L.; Fujiwara, Y.; Rogers, R. J.; Mitchell, H. L.; Whitesides, G. M. (1980). "Mechanism of formation of Grignard reagents. Kinetics of reaction of alkyl halides in diethyl ether with magnesium". Journal of the American Chemical Society. 102 (1): 217. .
- De Boer, H.J.R.; Akkerman, O.S; Bickelhaupt, F. (1988). "Carbanions as intermediates in the synthesis of Grignard Reagents". Angew. Chem. Int. Ed. 27 (5): 687–89. .
- Van Klink, G.P.M.; de Boer, H.J.R; Schat, G.; Akkerman, O.S.; Bickelhaupt, F.; Spek, A. (2002). "Carbanions as Intermediates in the Formation of Grignard Reagents". Organometallics. 21 (10): 2119–35. S2CID 94556915.
- Shao, Y.; Liu, Z.; Huang, P.; Liu, B. (2018). "A unified model of Grignard reagent formation". Physical Chemistry Chemical Physics. 20 (16): 11100–08. PMID 29620768.

![{\displaystyle {\begin{aligned}{\ce {R-X{}+Mg}}&\longrightarrow {\ce {[R-X^{\bullet }]^{-}{}+[Mg^{\bullet }]+}}\\{\ce {[R-X^{\bullet }]-}}&\longrightarrow {\ce {R^{\bullet }{}+X-}}\\{\ce {R^{\bullet }{}+[Mg^{\bullet }]+}}&\longrightarrow {\ce {R-Mg+}}\\{\ce {R-Mg+{}+X-}}&\longrightarrow {\ce {R-MgX}}\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/e5ad1a3f77639428eb795568fed4835951e25400)
![Reactions of Grignard reagents with non carbon electrophiles {\displaystyle {\begin{matrix}{\ce {R4B-}}\\{\color {White}\scriptstyle {\ce {Et2O.BF3\ or\ NaBF4}}}{\Bigg \uparrow }\scriptstyle {\ce {Et2O.BF3\ or\ NaBF4}}\\{\ce {Ph2PR<-[{\ce {Ph2PCl}}]RMgX->[{\ce {Bu3SnCl}}]Bu3SnR}}\\{\color {White}\scriptstyle {\ce {B(OMe)3}}}{\Bigg \downarrow }\scriptstyle {\ce {B(OMe)3}}\\{\ce {RB(OMe)2}}\end{matrix}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/e437b60e37160b69a20e287dba0820ae949a6a4d)
![Grignard oxygen oxidation pathways {\displaystyle {\begin{array}{lcrll}{\ce {{R-MgX}+O2->}}\ {\color {Red}{\ce {{R^{\bullet }}+[O2^{\bullet }]-}}}+{\ce {MgX+->}}&{\ce {R-O-O-MgX}}&{\color {Gray}+\ {\ce {H3O+}}}&{\ce {->{R-O-O-H}}}&{\color {Gray}+\ {\ce {{HO-MgX}+H+}}}\\&{\Bigg \downarrow }{\ce {R-MgX}}\\&{\ce {R-O-MgX}}&{\color {Gray}+\ {\ce {H3O+}}}&{\ce {->{R-O-H}}}&{\color {Gray}+\ {\ce {{HO-MgX}+H+}}}\\\end{array}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/137122d243bd686bf1055500358b585b758a0c54)







