Group 14 hydride
Group 14 hydrides are chemical compounds composed of hydrogen atoms and group 14 atoms (the elements of group 14 are carbon, silicon, germanium, tin, lead and flerovium).
Tetrahydrides
The tetrahydride series has the chemical formula XH4, with X representing any of the carbon family. Methane is commonly the result of the decomposition of organic matter and is a
They take on a pyramidal structure, and as such are not polar molecules like the other
Unlike other light hydrides such as
| Compound | Chemical formula | Molecular geometry | Space-filling model |
|---|---|---|---|
| carbon tetrahydride hydrogen carbide methane (carbane) |
CH4 | 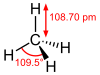 |

|
| silicon tetrahydride hydrogen silicide (silane) |
SiH4 | 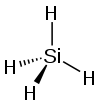 |

|
| germanium tetrahydride hydrogen germanide (germane) |
GeH4 |  |

|
| tin tetrahydride hydrogen stannide (stannane) |
SnH4 |  |

|
| lead tetrahydride hydrogen plumbide (plumbane) |
PbH4 |  |

|
| flerovium tetrahydride hydrogen flerovide (flerovane) |
FlH4 |
Hexahydrides
This series has the chemical formula X2H6. Ethane is commonly found alongside methane in natural gas. The other hydrides of the chemical formula X2H6 are less stable than the corresponding tetrahydrides XH4, and they are more and more less stable as X goes from carbon (ethane C2H6 is stable) down to lead (or flerovium) in the periodic table (diplumbane Pb2H6 is unknown[1]).
| Compound | Chemical formula | Molecular geometry | Space-filling model |
|---|---|---|---|
| Ethane (dicarbon hexahydride) (dicarbane) |
C2H6 |  |

|
| Disilane (disilicon hexahydride) |
Si2H6 |  |

|
| Digermane (digermanium hexahydride) |
Ge2H6 |  |

|
| Distannane (ditin hexahydride) |
Sn2H6 |  |

|
| Diplumbane (dilead hexahydride) |
Pb2H6 |  |

|
| Diflerovane (diflerovium hexahydride) |
Fl2H6 |
Higher group 14 hydrides
All straight-chain saturated group 14 hydrides follow the formula XnH2n+2, the same formula for the alkanes.

Many other group 14 hydrides are known.
Alongside
The other group 14 elements have a lower tendency to catenate. Hydrosilicons (
The first five hydrogermaniums GenH2n+2 are known and are fairly similar to the hydrosilicones,[5] e.g. germane GeH4 and digermane Ge2H6. They are germanium analogues of alkanes.
Stannane SnH4, a strong reducing agent slowly decomposes at room temperature to tin and hydrogen gas, and is decomposed by concentrated aqueous acids or alkalis; distannane, Sn2H6 is still more unstable, and longer hydrostannums (hydrotins) are unknown. Stannane and distannane are tin analogues of methane and ethane respectively.
Compounds containing hydrogen and multiple group 14 elements are known, one of the most famous of these being tetraethyllead Pb(CH2CH3)4 which contains carbon and lead. The other examples are methylsilane H3C−SiH3 which contains carbon and silicon, tris(trimethylsilyl)germanium hydride ((CH3)3Si)3GeH which contain carbon, silicon and germanium, silylgermane or germylsilane H3Si−GeH3 which contains silicon and germanium, and hexaphenyldiplumbane (C6H5)3Pb−Pb(C6H5)3 which contains carbon and lead.[6]
See also
- Methylene CH2
- MethylidyneCH
- Titanium(IV) hydride TiH4, a structural analog of the group 14 tetrahydrides
- Zirconium hydride ZrH4, ZrH2 and others
- Zirconium(II) hydride ZrH2
- Uranium(IV) hydride UH4
References
Bibliography
- ISBN 978-0-08-037941-8.