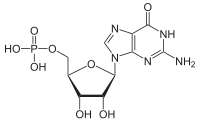
Guanosine monophosphate
| |

| |
| Names | |
|---|---|
| IUPAC name
5′-Guanylic acid
| |
| Systematic IUPAC name
[(2R,3S,4R,5R)-5-(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methyl dihydrogen phosphate | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| Abbreviations | GMP |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.001.453 |
| E number | E626 (flavour enhancer) |
IUPHAR/BPS |
|
| MeSH | Guanosine+monophosphate |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C10H14N5O8P | |
| Molar mass | 363.223 g·mol−1 |
| Acidity (pKa) | 0.7, 2.4, 6.1, 9.4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Guanosine monophosphate (GMP), also known as 5′-guanidylic acid or guanylic acid (
As an
De novo synthesis
GMP synthesis starts with D-ribose 5′-phosphate, a product of the pentose phosphate pathway. The synthesis proceeds by the gradual formation of the purine ring on carbon-1 of ribose, with CO2, glutamine, glycine, aspartate and one-carbon derivatives of tetrahydrofolate donating various elements towards the building of the ring.[2]
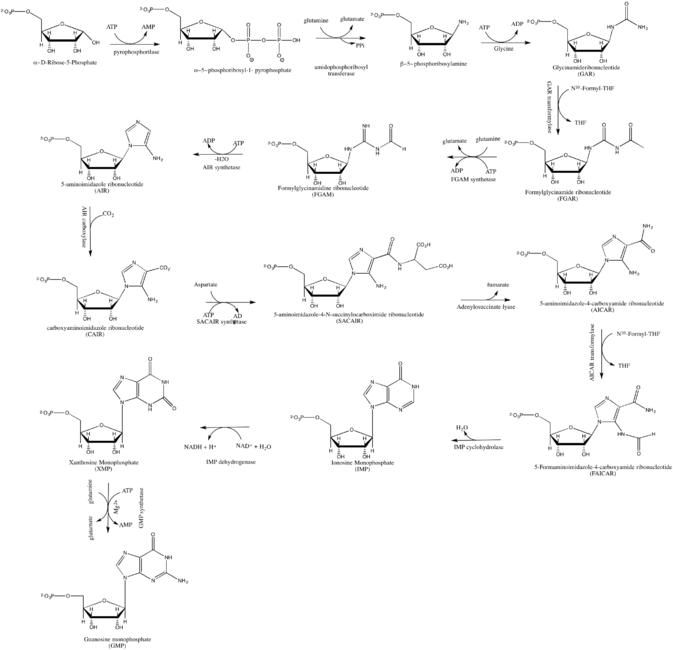
As inhibitor of guanosine monophosphate synthesis in experimental models, the glutamine analogue DON can be used.[3]
cGMP
GMP can also exist as a cyclic structure known as cyclic GMP. Within certain cells the enzyme guanylyl cyclase makes cGMP from GTP.
cGMP plays an important role in mediating hormonal signaling.[2]
Sources
GMP was originally identified as the umami substance in dried shiitake mushroom. The drying process significantly increases GMP content with the breakdown of RNA. It can be found in a number of other mushrooms.[4]
Industrial production is based on fermentation: a bacterium converts sugars into
Food additive
Guanosine monophosphate is known as
As it is a fairly expensive additive, it is usually not used independently of
See also
References
- ^ "The Vegetarian Resource Group Blog". www.vrg.org. Retrieved 25 April 2018.
- ^ ISBN 978-0-470-57095-1.
- ^ Ahluwalia GS et al. Metabolism and action of amino acid analog anti-cancer agents ”, in Pharmac. Ther. (1990) 46: 243-271
- PMID 26247011.
- S2CID 84216811.
- .
- ^ a b "Additive categories | CEFF". www.ceff.info. Retrieved 2021-11-30.
