HeLa


HeLa (
The cells from Lacks's cancerous cervical tumor were taken without her knowledge, which was common practice in the United States at the time.
History
Origin
In 1951, a woman named Henrietta Lacks was admitted to the Johns Hopkins Hospital with symptoms of irregular vaginal bleeding; she was subsequently treated for cervical cancer.[9] Her first treatment was performed by Lawrence Wharton Jr., who at that time collected tissue samples from her cervix without her consent.[10] Her cervical biopsy supplied samples of tissue for clinical evaluation and research by George Otto Gey, head of the Tissue Culture Laboratory. Gey's lab assistant Mary Kubicek used the roller-tube technique to culture the cells.[9] It was observed that the cells grew robustly, doubling every 20–24 hours, unlike previous specimens, which died out.[11]
The cells were propagated by Gey shortly before Lacks died of her cancer in 1951. This was the first human cell line to prove successful in vitro, which was a scientific achievement with profound future benefit to medical research. Gey freely donated these cells, along with the tools and processes that his lab developed, to any scientist requesting them, simply for the benefit of science. Neither Lacks nor her family gave permission to harvest the cells.[12] The cells were later commercialized, although never patented in their original form. There was no requirement at that time to inform patients or their relatives about such matters, because discarded material or material obtained during surgery, diagnosis, or therapy was the property of the physician or the medical institution.[citation needed]
As was customary for Gey's lab assistant, the culture was named after the first two letters of Henrietta Lacks' first and last names, He + La.[9] Before a leak to the public in the 1970s, which revealed her true name, the "HeLa" cell line was mistakenly believed to have been named after a "Helen Lane" or "Helen Larson".[4][13]
When other cell cultures were being invaded by suspected HeLa cells, one research group contacted the Lacks family,[when?] seeking DNA samples to help identify contaminating cell lines. The family never understood the purpose of the visit, but they were distressed by their understanding of what the researchers told them.[13][14] These cells are treated as cancer cells, as they are descended from a biopsy taken from a visible lesion on the cervix as part of Lacks's diagnosis of cancer.[15]
HeLa cells, like other cell lines, are termed "immortal" because they can divide an unlimited number of times in a laboratory cell culture plate, as long as fundamental cell survival conditions are met (i.e. being maintained and sustained in a suitable environment). There are many strains of HeLa cells, because they mutate during division in cell cultures, but all HeLa cells are descended from the same tumor cells removed from Lacks. The total number of HeLa cells that have been propagated in cell culture far exceeds the total number of cells that were in Henrietta Lacks's body.[16]
Controversy

Lacks's case is one of many examples of the lack of informed consent in 20th-century medicine. Communication between tissue donors and doctors was virtually nonexistent—cells were taken without patient consent, and patients were not told what the cells would be used for. Johns Hopkins Hospital, where Lacks received treatment and had her tissue harvested, was the only hospital in the Baltimore area where African American patients could receive free care. The patients who received free care from this segregated sect of the hospital often became research subjects without their knowledge.[17] Lacks's family also had no access to her patient files and had no say in who received HeLa cells or what they would be used for. Additionally, as HeLa cells were popularized and used more frequently throughout the scientific community, Lacks's relatives received no financial benefit and continued to live with limited access to healthcare.[18][13]
This issue of who owns tissue samples taken for research was brought up in the Supreme Court of California case of Moore v. Regents of the University of California in 1990. The court ruled that a person's discarded tissue and cells are not his or her property and can be commercialized.[19]
Lacks's case influenced the establishment of the Common Rule in 1981. The Common Rule enforces informed consent by ensuring that doctors inform patients if they plan to use any details of the patient's case in research and give them the choice of disclosing the details or not. Tissues connected to their donors' names are also strictly regulated under this rule, and samples are no longer named using donors' initials, but rather by code numbers.[19] To further resolve the issue of patient privacy, Johns Hopkins established a joint committee with the NIH and several of Lacks's family members to determine who receives access to Henrietta Lacks's genome.[20]
In 2021, Henrietta Lacks's estate sued to get past and future payments for the alleged unauthorized and widely known sale of HeLa cells by Thermo Fisher Scientific.[21] Lacks's family hired an attorney to seek compensation from upwards of 100 pharmaceutical companies that have used and profited from HeLa cells.[22] Settlement of the suit with Thermo Fisher Scientific was announced in August 2023, with undisclosed terms.[23] Subsequent to the settlement it was announced that the Lacks family was suing the company Ultragenyx.[24]
Use in research
HeLa cells were the first human cells to be successfully cloned in 1953, by
Polio eradication
HeLa cells were used by
Virology
HeLa cells have been used in testing how
HeLa cells have also been instrumental in the development of human papilloma virus (HPV) vaccines. In the 1980s, Harald zur Hausen found that Lacks's cells from the original biopsy contained HPV-18, which was later found to be the cause of the aggressive cancer that had killed her. His work in linking HPV with cervical cancer won him a Nobel Prize and led to the development of HPV vaccines, which are predicted to reduce the number of deaths from cervical cancer by 70%.[34]
Over the years, HeLa cells have been infected with various types of viruses, including HIV, Zika, mumps, and herpesviruses to test and develop new vaccines and drugs. Dr. Richard Axel discovered that the addition of the CD4 protein to HeLa cells enabled them to be infected with HIV, allowing the virus to be studied.[35] In 1979, scientists learned that the measles virus constantly mutates when it infects HeLa cells,[36] and in 2019 they found that Zika cannot multiply in HeLa cells.[37]
Cancer
HeLa cells have been used in a number of cancer studies, including those involving sex steroid hormones, such as estradiol and estrogen, and estrogen receptors, along with estrogen-like compounds, such as quercetin, which has cancer-reducing properties.[38] There have also been studies on HeLa cells, involving the effects of flavonoids and antioxidants with estradiol on cancer cell proliferation.
In 2011, HeLa cells were used in tests of novel
In 2014, HeLa cells were shown to provide a viable cell line for tumor
Genetics
In 1953, a lab mistake involving mixing HeLa cells with the wrong liquid allowed researchers for the first time to see and count each chromosome clearly in the HeLa cells with which they were working. This accidental discovery led scientists Joe Hin Tjio and Albert Levan to develop better techniques for staining and counting chromosomes.[34] They were the first to show that humans have 23 pairs of chromosomes rather than 24, as was previously believed. This was important for the study of developmental disorders, such as Down syndrome, that involve the number of chromosomes.
In 1965, Henry Harris and John Watkins created the first human-animal hybrid by fusing HeLa cells with mouse embryo cells. This enabled advances in mapping genes to specific chromosomes, which would eventually lead to the Human Genome Project.[34]
Space microbiology
In the 1960s, HeLa cells were sent on the Soviet satellite Sputnik-6 and human space missions to determine the long term effects of space travel on living cells and tissues. Scientists discovered that HeLa cells divide more quickly in zero gravity.[44]
Analysis
Telomerase
The HeLa
Chromosome number
Complete genome sequence
The complete
Contamination
HeLa cells are sometimes difficult to control, because they adapt to growth in tissue culture plates and invade and outcompete other cell lines. Through improper maintenance, they have been known to contaminate other cell cultures in the same laboratory, interfering with biological research and forcing researchers to declare many results invalid. The degree of HeLa cell contamination among other cell types is unknown, because few researchers test the identity or purity of already established cell lines. It has been shown that a substantial fraction of
HeLa cell contamination has become a pervasive worldwide problem – affecting even the laboratories of many notable physicians, scientists, and researchers, including Jonas Salk. The HeLa contamination problem also contributed to Cold War tensions. The USSR and the USA had begun to cooperate in the war on cancer launched by President Richard Nixon, only to find that the exchanged cells were contaminated by HeLa.[57]
Rather than focus on how to resolve the problem of HeLa cell contamination, many scientists and science writers continue to document this problem as simply a contamination issue – caused not by human error or shortcomings but by the hardiness, proliferation, or overpowering nature of HeLa cells.[58] Recent data suggest that cross-contamination is still a major problem with modern cell cultures.[3][59] The International Cell Line Authentication Committee (ICLAC) notes that many cases of cell line misidentification are the result of cross-contamination of the culture by another, faster-growing cell line. This calls into question the validity of the research done using contaminated cell lines, as certain attributes of the contaminant, which may come from an entirely different species or tissue, may be misattributed to the cell line under investigation.[60]
New species proposal
HeLa cells were described by
- the chromosomal incompatibility of HeLa cells with human cells;
- the ecological niche of HeLa cells;
- their ability to persist and expand well beyond the desires of human cultivators;
- the possession by HeLa cells of their own clonal karyotype, defining it as a distinct species.[62]
Van Valen proposed the new family Helacytidae and the genus Helacyton, and in the same paper proposed a new species for HeLa cells.[63]
However, this proposal was not taken seriously by other prominent evolutionary biologists, nor by scientists in other disciplines. Van Valen's argument that HeLa are a new species does not fulfill the criteria for an independent unicellular asexually reproducing species, because of the notorious instability of HeLa's karyotype and their lack of a strict ancestral-descendant lineage.[64]
Gallery
-
Multiphoton fluorescence image of HeLa cells stained with the actin- binding toxin phalloidin (red), microtubules (cyan), and cell nuclei (blue). Nikon RTS2000MP custom laser scanning microscope.
-
Multiphoton fluorescence image of HeLa cells with cytoskeletal microtubules (magenta) and DNA (cyan). Nikon RTS2000MP custom laser scanning microscope.
-
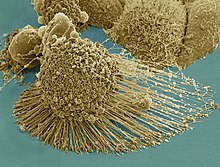
Scanning electron micrograph of just-divided HeLa cells. Zeiss Merlin HR-SEM.
-
HeLa cells stained with Hoechst 33258
-
HeLa cells grown in culture and stained with antibody tomicrotubules and the Ki-67 antibody is expressed in cells about to divide. Preparation, antibodies and image courtesy of EnCor Biotechnology.
-
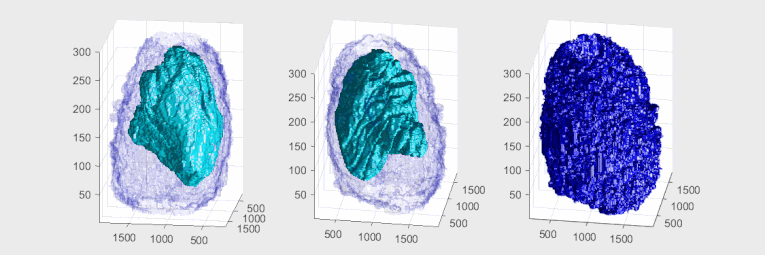
A volumetric surface render (red) of theelectron microscopyand the nuclear envelope was automatically segmented and rendered. One vertical and one horizontal slice are added for reference.
-
Plasma membrane and nuclear envelope of one Hela cell, displayed as a volumetric surface rendering. Left and centre: the plasma membrane in blue, with transparency, and the nuclear envelope in solid cyan. Right: the plasma membrane without transparency and the same angle of view as the centre picture. The membranes have been segmented from data acquired by electron microscopy.
In media
- The 1997 documentary The Way of All Flesh by Adam Curtis explained the history of HeLa cells and their implications for medicine and society.[65]
- A 2010 episode of Law & Order, "Immortal", was heavily based on the story of Henrietta Lacks and the HeLa cell line, using the fictional "NaRo" cells as a stand-in.[66]
- The story of how the HeLa cell line came to be was also the subject of a 2010 episode of the podcast Radiolab.[67]
- HeLa cells were the subject of a 2010 book by Rebecca Skloot, The Immortal Life of Henrietta Lacks, investigating the historical context of the cell line and how the Lacks family was involved in its use.[13]
- A 2017 HBO film, Renee Elise Goldsberry as Henrietta Lacks. Author Rebecca Skloot also appeared as a character in the film, portrayed by Rose Byrne.[68]
See also
- Clonally transmissible cancer
- Moore v. Regents of the University of California, case that set precedent for discarded tissue
- List of contaminated cell lines
- WI-38
References
- PMID 19450234.
- ^ Morris, Rhys Bowen (August 2, 2023). "What were the top 100 cell lines of 2022?". CiteAb Blog. Retrieved August 17, 2023.
- ^ S2CID 2929020.
- ^ a b Batts DW (May 10, 2010). "Cancer cells killed Henrietta Lacks – then made her immortal". The Virginian-Pilot. pp. 1, 12–14. Archived from the original on November 25, 2016. Retrieved May 8, 2020.
- ^ PMID 13052828.
- ^ "Johns Hopkins Magazine -- April 2000". pages.jh.edu.
- ABC World News. Retrieved August 19, 2012.
- ^ McKie, Robin (April 3, 2010). "Henrietta Lacks's cells were priceless, but her family can't afford a hospital". The Guardian. London. Retrieved July 18, 2017.
- ^ PMID 19722756.
- PMID 9215212.
- ^ Butanis, Benjamin. "The Legacy of Henrietta Lacks". www.hopkinsmedicine.org. Retrieved May 7, 2020.
- ^ Washington, Harriet "Henrietta Lacks: An Unsung Hero", Emerge Magazine, October 1994
- ^ a b c d Zielinski, Sarah (January 2, 2010). "Cracking the code of the human genome – Henrietta Lacks' 'immortal' cells". Smithsonian. Retrieved May 27, 2017.
- ^ White, Tracie (May 2, 2018), "Descendants of Henrietta Lacks discuss her famous cell line", Stanford Medicine News Center, retrieved December 9, 2021
- ^ del Carpio, Alexandra (April 27, 2014). "The Good, the Bad, and the HeLa". Berkeley Science Review. Archived from the original on May 23, 2017. Retrieved May 27, 2017.
- ^ Sharrer T (2006). ""HeLa" Herself". The Scientist. 20 (7): 22.
- JSTOR 43264385.
- ^ Day, Jo Ann. "Upholding the Highest Bioethical Standards | Johns Hopkins Medicine". www.hopkinsmedicine.org. Retrieved April 15, 2020.
- ^ ISBN 978-1-4000-5217-2.
- ^ "HeLa Cell Line Origins, Contamination, Controversy, and Cytogenetics". Retrieved April 15, 2020.
- ^ "Estate of Henrietta Lacks sues Thermo Fisher over the improper sale of her immortal cells".
- ISSN 0190-8286. Retrieved December 11, 2021.
- ^ Holpuch, Amanda (August 1, 2023). "Family of Henrietta Lacks Settles With Biotech Company That Used Her Cells". The New York Times.
- ^ "Ultragenyx Sued by Henrietta Lacks' Family in Second HeLa Cell Line Lawsuit". Retrieved August 11, 2023.
- PMID 16589695.
- ^ Smith, Van (April 17, 2002). "Wonder Woman: The Life, Death, and Life After Death of Henrietta Lacks, Unwitting Heroine of Modern Medical Science". Baltimore City Paper. Archived from the original on August 14, 2004. Retrieved March 2, 2017.
- ^ S2CID 991019.
- PMID 23124495.
- .
- PMID 11264378.
- PMID 20080135.
- S2CID 9203953.
- ^ PMID 21718481.
- ^ a b c MacDonald, Anna (June 13, 2018). "5 Contributions HeLa Cells Have Made to Science". Cell Science from Technology Networks. Retrieved March 25, 2020.
- PMID 9557643.
- PMID 513191.
- PMID 31434319.
- S2CID 24034074.
- PMID 22182749.
- S2CID 37043095.
- ^ Briiuner., Thomas; Dieter F. Hulser (1990). "Tumor Cell Invasion and Gap Junctional Communication" (PDF). Invasion Metastasis. 10: 31–34. Retrieved April 3, 2012.
- S2CID 13743291.
- S2CID 25652255. Archived from the original(PDF) on April 25, 2018.
- ^ Davies, Preston. "Significant Research Advances Enabled by HeLa Cells". Office of Science Policy. Archived from the original on February 2, 2020. Retrieved March 25, 2020.
- ^ The Nobel Prize in Physiology or Medicine 2009 on nobelprize.org
- S2CID 9390790.
- ^ PMID 9892199.
- PMID 23550136.
- PMID 5768876.
- PMID 23925245.
- PMID 23550136.
- S2CID 87549985. Retrieved August 8, 2013.
- ^ S2CID 88020977. Retrieved August 8, 2013.
- PMID 23925220.
- ^ a b "NIH, Lacks family reach understanding to share genomic data of HeLa cells". The National Institutes of Health. August 7, 2013. Archived from the original on August 11, 2013. Retrieved August 8, 2013.
- S2CID 190871214.
- OCLC 12805138.
- PMID 16817967.
- S2CID 21077969. Archived from the original(PDF) on December 17, 2008. Retrieved December 4, 2007.
- ^ "ATCC® Standards Development Organization: The International Cell Line Authentication Committee (ICLAC)". Standards.atcc.org. Archived from the original on May 8, 2013. Retrieved June 27, 2013.
- ^ Maiorana VC, Van Valen LM (February 7, 1991). "Hela, a new microbial species" (PDF). Evolutionary Theory. 10 (2): 71–4. Archived (PDF) from the original on March 29, 2018.
- PMID 21666415.
- ISSN 1528-2619.
- PMID 12142474.
- ^ "Modern Times: The Way of All Flesh - BBC Two England - 19 March 1997". The Radio Times (3815): 92. March 13, 1997.
- ^ Thomas, June (May 19, 2010). "Ripped From Which Headline? "Immortal"". Slate Magazine. Retrieved August 3, 2021.
- ^ "Henrietta's Tumor".
- ^ "About The Immortal Life of Henrietta Lacks". Rebecca Skloot. Retrieved April 24, 2018.
Further reading
- ISBN 978-0-253-21428-7.
- Rebecca Skloot (2010). The Immortal Life of Henrietta Lacks.
External links
- HeLa (CCL-2 Cells) in the ATCCdatabase
- HeLa Cells at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- HeLa Transfection and Selection Data for HeLa Cells
- Rebecca Skloot, The Immortal Life of Henrietta Lacks book website with additional features (photo/video/audio)
- The Henrietta Lacks Foundation, a foundation established to, among other things, help provide scholarship funds and health insurance to Henrietta Lacks's family.
- "Wonder Woman: The Life, Death, and Life After Death of Henrietta Lacks, Unwitting Heroine of Modern Medical Science" by Van Smith
- "What's Left of Henrietta Lacks?" by Anne Enright
- Cell Centered Database – HeLa cell
- Cellosaurus entry for HeLa
- The Legacy of Henrietta Lacks