Heck reaction
| Heck reaction | |
|---|---|
| Named after | Richard F. Heck |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | heck-reaction |
| RSC ontology ID | RXNO:0000024 |
The Heck reaction (also called the Mizoroki–Heck reaction)
 |
| The Heck reaction |
|---|
History
The original reaction by Tsutomu Mizoroki (1971) describes the coupling between
 |
| Mizoroki 1971 |
|---|
In 1972 Heck acknowledged the Mizoroki publication and detailed independently discovered work. Heck's reaction conditions differ in terms of the catalyst (palladium acetate), catalyst loading (0.01 eq.), base (hindered amine), and absence of solvent.[9][10]
 |
| Heck 1972 |
|---|
In 1974 Heck showed that phosphine ligands facilitated the reaction.[11]
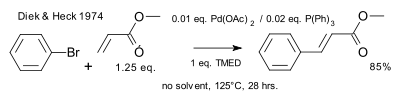 |
| Heck reaction 1974 phosphines |
|---|
Catalyst and substrates
The reaction is catalyzed by palladium complexes. Typical catalysts and precatalysts include
The aryl electrophile can be a halide (Br, Cl) or a triflate as well as
Reaction mechanism
The mechanism of this
For instance,
 |
| Heck Reaction Mechanism |
|---|
This cycle is not limited to vinyl compounds, in the
Stereoselectivity
This
 |
| The Heck reaction in Naproxen production |
|---|
Variations
Ionic liquid Heck reaction
In the presence of an
 |
| Siloxane application |
|---|
Heck oxyarylation
In the Heck oxyarylation modification the palladium substituent in the syn-addition intermediate is displaced by a hydroxyl group and the reaction product contains a dihydrofuran ring.[17]
 |
| Heck oxyarylation |
|---|
Amino-Heck reaction
In the amino-Heck reaction a
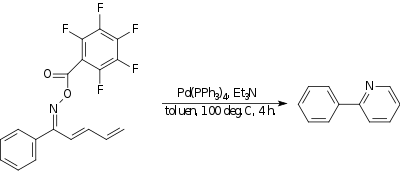 |
| Amino-Heck reaction |
|---|
See also
- Hiyama coupling
- Stille reaction
- Suzuki reaction
- Sonogashira coupling
- Intramolecular Heck reaction
- Negishi Coupling
References
- . Retrieved June 4, 2011.
- ISBN 978-0471264187.
- .
- PMID 11749313.
- PMID 21677934.
- .
- PMID 27462934.
- .
- .
- .
- .
- ^ Littke, A. F.; Fu, G. C. (2005). "Heck reactions of aryl chlorides catalyzed by palladium/tri-tert-butylphosphine: (E)-2-Methyl-3-phenylacryacid butyl ester and (E)-4-(2-phenylethenyl)benzonitrile". Organic Syntheses. 81: 63.
- .
- PMID 22011792.
- .
- PMID 15957033.
- ^ Lorand Kiss; Tibor Kurtan; Sandor Antus; Henri Brunner (2003). "Further insight into the mechanism of Heck oxyarylation in the presence of chiral ligands". Arkivoc: GB–653J.
- ^ Mitsuru Kitamura; Daisuke Kudo; Koichi Narasaka (2005). "Palladium(0)-catalyzed synthesis of pyridines from β-acetoxy-γ,δ-unsaturated ketone oximes". Arkivoc: JC–1563E.
External links
- The Heck reaction at organic-chemistry.org Article
