Helicene


In
chiral molecules.[1] The chemistry of helicenes has attracted continuing attention because of their unique structural, spectral, and optical features.[2][3][4][5][6][7][8]
Structure and properties
The
steric collisions. For helicenes with six benzene units, a 360° turn is completed. In the helicene series the dihedral angles
between the extremities increases going from [4]helicene (26°) to [6]helicene (58°) and then decreases again for example in [7]helicene (30°).
Helicenes are notable for having chirality despite lacking both
dextrorotatory
.
The stability of the two complementary helical enantiomers with respect to interconversion and the mechanism by which they interconvert depend on n.[9]
Synthesis
The first helicene structure was reported by
2-nitronaphthalene.[10] [5]helicene was synthesized in 1918 by Weitzenböck & Klingler.[11] The first [6]helicene (also called hexahelicene) was synthesized by M. S. Newman and D. Lednicer in 1955 via a scheme that closed the two central rings by Friedel–Crafts cyclization of carboxylic acid compounds.[12][13] Since then, several methods for synthesizing helicenes with different lengths and substituents are used. The oxidative photocyclization of a stilbene-type precursor is used most often as the key step. The longest helicene prepared by this method is [16]helicene in 2015.[14]
In one study,Grubbs' second generation catalyst:
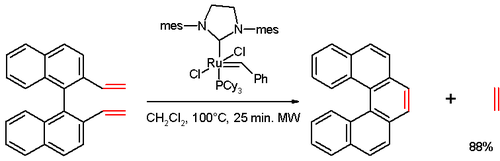
Other approach is also non-photochemical and is based on assembly of biphenylyl-naphthalenes and their platinum-catalyzed double cycloisomerization leading to various [6]helicenes:[16]
-
[4]Helicene
-
[5]Helicene
-
[6]Helicene
-
[6]Helicene, other chirality
-
[7]Helicene
-
[7]Helicene, other chirality
-
[8]Helicene
-
[9]Helicene
-
[10]Helicene
-
[11]Helicene
-
[12]Helicene
-
[13]Helicene
-
[14]Helicene
-
[15]Helicene
-
[16]Helicene
-
[18]Helicene
Applications
Helicenes have been studied with respect to
See also
- Other configurations of consecutively-fused benzene rings:
- Acenes, linear
- Phenacenes, zig-zag
- Circulenes, closed ring
References
- ^
- .
- .
- ^ Helquat Dyes: Helicene-like Push–Pull Systems with Large Second-Order Nonlinear Optical Responses Benjamin J. Coe, Daniela Rusanova, Vishwas D. Joshi, Sergio Sánchez, Jan Vávra, Dushant Khobragade, Lukáš Severa, Ivana Císařová, David Šaman, Radek Pohl, Koen Clays, Griet Depotter, Bruce S. Brunschwig, and Filip Teplý The Journal of Organic Chemistry 2016 81 (5), 1912-1920
- ^ Synthesis and Structural Features of Quadruple Helicenes: Highly Distorted π Systems Enabled by Accumulation of Helical Repulsions Takao Fujikawa, Yasutomo Segawa, and Kenichiro Itami Journal of the American Chemical Society 2016 138 (10), 3587-3595
- General references
- Chuan-Feng; Yun Shen (2017). Helicene Chemistry: From Synthesis to Applications. Springer. S2CID 199492403.
Wikimedia Commons has media related to Helicenes.


![[4]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/e/ee/Tetrahelicene.jpg/120px-Tetrahelicene.jpg)
![[5]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/7/7c/Pentahelicene.jpg/120px-Pentahelicene.jpg)
![[6]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/9/95/Hexahelicene.jpg/120px-Hexahelicene.jpg)
![[6]Helicene, other chirality](http://upload.wikimedia.org/wikipedia/commons/thumb/e/e2/Hexahelicene2.jpg/119px-Hexahelicene2.jpg)
![[7]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/2/29/Heptahelicene.jpg/120px-Heptahelicene.jpg)
![[7]Helicene, other chirality](http://upload.wikimedia.org/wikipedia/commons/thumb/9/9f/Heptahelicene2.jpg/120px-Heptahelicene2.jpg)
![[8]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/6/64/Octahelicene.jpg/120px-Octahelicene.jpg)
![[9]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/5/57/Nonahelicene.jpg/120px-Nonahelicene.jpg)
![[10]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Decahelicene.jpg/90px-Decahelicene.jpg)
![[11]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/2/2e/Undecahelicene.jpg/120px-Undecahelicene.jpg)
![[12]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/e/e5/Dodecahelicene.jpg/111px-Dodecahelicene.jpg)
![[13]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/0/08/Tridecahelicene.jpg/99px-Tridecahelicene.jpg)
![[14]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/8/86/Tetradecahelicene.jpg/120px-Tetradecahelicene.jpg)
![[15]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/c/cc/Pentadecahelicene.jpg/120px-Pentadecahelicene.jpg)
![[16]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/e/e6/Hexadecahelicene.jpg/112px-Hexadecahelicene.jpg)
![[18]Helicene](http://upload.wikimedia.org/wikipedia/commons/thumb/0/04/Octadecahelicene.jpg/97px-Octadecahelicene.jpg)