Helium-3
| General | |
|---|---|
Parent isotopes | 3H (beta decay of tritium) |
| Isotopes of helium Complete table of nuclides | |
Helium-3 (3He[1][2] see also helion) is a light, stable isotope of helium with two protons and one neutron (in contrast, the most common isotope, helium-4 has two protons and two neutrons). Other than protium (ordinary hydrogen), helium-3 is the only stable isotope of any element with more protons than neutrons. Helium-3 was discovered in 1939.
Helium-3 occurs as a primordial nuclide, escaping from Earth's crust into its atmosphere and into outer space over millions of years. Helium-3 is also thought to be a natural nucleogenic and cosmogenic nuclide, one produced when lithium is bombarded by natural neutrons, which can be released by spontaneous fission and by nuclear reactions with cosmic rays. Some of the helium-3 found in the terrestrial atmosphere is also an artifact of atmospheric and underwater nuclear weapons testing.
Much speculation has been made over the possibility of helium-3 as a future
The abundance of helium-3 is thought to be greater on the Moon than on Earth, having been created in the upper layer of regolith by the solar wind over billions of years,[5] though still lower in abundance than in the Solar System's gas giants.[6][7]
History
The existence of helium-3 was first proposed in 1934 by the Australian
Physical properties
Due to its low atomic mass of 3.016
The
Pure helium-3 gas boils at 3.19
Superfluidity

An important property of helium-3, which distinguishes it from the more common helium-4, is that its nucleus is a
In a zero magnetic field, there are two distinct superfluid phases of 3He, the A-phase and the B-phase. The B-phase is the low-temperature, low-pressure phase which has an isotropic energy gap. The A-phase is the higher temperature, higher pressure phase that is further stabilized by a magnetic field and has two point nodes in its gap. The presence of two phases is a clear indication that 3He is an unconventional superfluid (superconductor), since the presence of two phases requires an additional symmetry, other than gauge symmetry, to be broken. In fact, it is a p-wave superfluid, with spin one, S=1, and angular momentum one, L=1. The ground state corresponds to total angular momentum zero, J=S+L=0 (vector addition). Excited states are possible with non-zero total angular momentum, J>0, which are excited pair collective modes. Because of the extreme purity of superfluid 3He (since all materials except 4He have solidified and sunk to the bottom of the liquid 3He and any 4He has phase separated entirely, this is the most pure condensed matter state), these collective modes have been studied with much greater precision than in any other unconventional pairing system.
Natural abundance
Terrestrial abundance
3He is a primordial substance in the Earth's mantle, thought to have become entrapped in the Earth during planetary formation. The ratio of 3He to 4He within the Earth's crust and mantle is less than that of estimates of solar disk composition as obtained from meteorite and lunar samples, with terrestrial materials generally containing lower 3He/4He ratios due to production of 4He from radioactive decay.
3He has a cosmological ratio of 300 atoms per million atoms of 4He (at. ppm),[17] leading to the assumption that the original ratio of these primordial gases in the mantle was around 200-300 ppm when Earth was formed. Over Earth's history alpha-particle decay of uranium, thorium and other radioactive isotopes has generated significant amounts of 4He, such that only around 7% of the helium now in the mantle is primordial helium,[17] lowering the total 3He/4He ratio to around 20 ppm. Ratios of 3He/4He in excess of atmospheric are indicative of a contribution of 3He from the mantle. Crustal sources are dominated by the 4He produced by radioactive decay.
The ratio of helium-3 to helium-4 in natural Earth-bound sources varies greatly.[18][19] Samples of the lithium ore spodumene from Edison Mine, South Dakota were found to contain 12 parts of helium-3 to a million parts of helium-4. Samples from other mines showed 2 parts per million.[18]
Helium is also present as up to 7% of some natural gas sources,[20] and large sources have over 0.5% (above 0.2% makes it viable to extract).[21] The fraction of 3He in helium separated from natural gas in the U.S. was found to range from 70 to 242 parts per billion.[22][23] Hence the US 2002 stockpile of 1 billion normal m3[21] would have contained about 12 to 43 kilograms (26 to 95 lb) of helium-3. According to American physicist Richard Garwin, about 26 cubic metres (920 cu ft) or almost 5 kilograms (11 lb) of 3He is available annually for separation from the US natural gas stream. If the process of separating out the 3He could employ as feedstock the liquefied helium typically used to transport and store bulk quantities, estimates for the incremental energy cost range from $34 to $300 per litre ($150 to $1,360/imp gal) NTP, excluding the cost of infrastructure and equipment.[22] Algeria's annual gas production is assumed to contain 100 million normal cubic metres[21] and this would contain between 7 and 24 cubic metres (250 and 850 cu ft) of helium-3 (about 1 to 4 kilograms (2.2 to 8.8 lb)) assuming a similar 3He fraction.
3He is also present in the
3He is produced on Earth from three sources: lithium
The total amount of helium-3 in the mantle may be in the range of 0.1–1 megatonne (98,000–984,000 long tons; 110,000–1,100,000 short tons). However, most of the mantle is not directly accessible. Some helium-3 leaks up through deep-sourced
Lunar surface
See Extraterrestrial mining or Lunar resources
Solar nebula (primordial) abundance
One early estimate of the primordial ratio of 3He to 4He in the solar nebula has been the measurement of their ratio in the atmosphere of Jupiter, measured by the mass spectrometer of the Galileo atmospheric entry probe. This ratio is about 1:10,000,
Human production
Tritium decay
Virtually all helium-3 used in industry today is produced from the radioactive decay of tritium, given its very low natural abundance and its very high cost.
Production, sales and distribution of helium-3 in the United States are managed by the
While tritium has several different experimentally determined values of its half-life, NIST lists 4,500±8 d (12.32±0.02 years).[30] It decays into helium-3 by beta decay as in this nuclear equation:
3
1H
→ 3
2He1+
+
e−
+
ν
e
Among the total released energy of 18.6 keV, the part taken by
The low energy of tritium's radiation makes it difficult to detect tritium-labeled compounds except by using liquid scintillation counting.
Tritium is a radioactive isotope of hydrogen and is typically produced by bombarding lithium-6 with neutrons in a nuclear reactor. The lithium nucleus absorbs a neutron and splits into helium-4 and tritium. Tritium decays into helium-3 with a half-life of 12.3 years, so helium-3 can be produced by simply storing the tritium until it undergoes radioactive decay. As tritium forms a stable compound with oxygen (
Tritium is a critical component of
For decades this has been, and remains, the principal source of the world's helium-3.[32] However, since the signing of the START I Treaty in 1991 the number of nuclear warheads that are kept ready for use has decreased.[33][34] This has reduced the quantity of helium-3 available from this source. Helium-3 stockpiles have been further diminished by increased demand,[22] primarily for use in neutron radiation detectors and medical diagnostic procedures. US industrial demand for helium-3 reached a peak of 70,000 litres (15,000 imp gal; 18,000 US gal) (approximately 8 kilograms (18 lb)) per year in 2008. Price at auction, historically about $100 per litre ($450/imp gal), reached as high as $2,000 per litre ($9,100/imp gal).[35] Since then, demand for helium-3 has declined to about 6,000 litres (1,300 imp gal; 1,600 US gal) per year due to the high cost and efforts by the DOE to recycle it and find substitutes. Assuming a density of 114 grams per cubic metre (0.192 lb/cu yd) at $100/l helium-3 would be about a thirtieth as expensive as tritium (roughly $880 per gram ($25,000/oz) vs roughly $30,000 per gram ($850,000/oz)) while at $2000/l helium-3 would be about half as expensive as tritium ($17,540 per gram ($497,000/oz) vs $30,000 per gram ($850,000/oz)).
The DOE recognized the developing shortage of both tritium and helium-3, and began producing tritium by lithium irradiation at the
Currently only two commercial nuclear reactors (Watts Bar Nuclear Plant Units 1 and 2) are being used for tritium production but the process could, if necessary, be vastly scaled up to meet any conceivable demand simply by utilizing more of the nation's power reactors[citation needed]. Substantial quantities of tritium and helium-3 could also be extracted from the heavy water moderator in
As tritium is also produced inadvertently in various processes in
Uses
Helium-3 spin echo
Helium-3 can be used to do spin echo experiments of surface dynamics, which are underway at the Surface Physics Group at the Cavendish Laboratory in Cambridge and in the Chemistry Department at Swansea University.
Neutron detection
Helium-3 is an important isotope in instrumentation for neutron detection. It has a high absorption cross section for thermal neutron beams and is used as a converter gas in neutron detectors. The neutron is converted through the nuclear reaction
- n + 3He → 3H + 1H + 0.764 MeV
into charged particles
Furthermore, the absorption process is strongly spin-dependent, which allows a spin-polarized helium-3 volume to transmit neutrons with one spin component while absorbing the other. This effect is employed in neutron polarization analysis, a technique which probes for magnetic properties of matter.[41][42][43][44]
The United States
Cryogenics
A helium-3 refrigerator uses helium-3 to achieve temperatures of 0.2 to 0.3 kelvin. A dilution refrigerator uses a mixture of helium-3 and helium-4 to reach cryogenic temperatures as low as a few thousandths of a kelvin.[47]
Medical imaging
Helium-3 nuclei have an intrinsic
Radio energy absorber for tokamak plasma experiments
Both MIT's Alcator C-Mod tokamak and the Joint European Torus (JET) have experimented with adding a little helium-3 to a H–D plasma to increase the absorption of radio-frequency (RF) energy to heat the hydrogen and deuterium ions, a "three-ion" effect.[50][51]
Nuclear fuel
This section contains weasel words: vague phrasing that often accompanies biased or unverifiable information. (March 2013) |
| Reactants | Products | Q | n/MeV | |
|---|---|---|---|---|
| First-generation fusion fuels | ||||
| 2D + 2D | → | 3He + 1 0n |
3.268 MeV
|
0.306 |
| 2D + 2D | → | 3T + 1 1p |
4.032 MeV
|
0 |
| 2D + 3T | → | 4He + 1 0n |
17.571 MeV
|
0.057 |
| Second-generation fusion fuel | ||||
| 2D + 3He | → | 4He + 1 1p |
18.354 MeV
|
0 |
| Third-generation fusion fuels | ||||
| 3He + 3He | → | 4He + 2 1 1p |
12.86 MeV
|
0 |
11B + 11p |
→ | 3 4He | 8.68 MeV
|
0 |
| Net result of 2D burning (sum of first 4 rows) | ||||
| 6 2D | → | 2(4He + n + p) | 43.225 MeV
|
0.046 |
| Current nuclear fuel | ||||
235U + n
|
→ | 2 FP + 2.5n
|
~200 MeV
|
0.0075 |
3He can be produced by the low temperature fusion of → 3He + γ + 4.98 MeV. If the fusion temperature is below that for the helium nuclei to fuse, the reaction produces a high energy alpha particle which quickly acquires an electron producing a stable light helium ion which can be utilized directly as a source of electricity without producing dangerous neutrons.
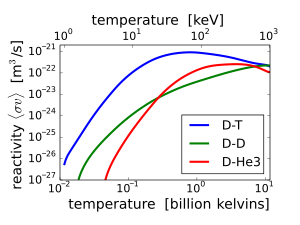
3He can be used in fusion reactions by either of the reactions 2H + 3He → 4He + 1p + 18.3 MeV, or 3He + 3He → 4He + 2 1p + 12.86 MeV.
The conventional
Because of the higher
The second possibility, fusing 3He with itself (3He + 3He), requires even higher temperatures (since now both reactants have a +2 charge), and thus is even more difficult than the D-3He reaction. However, it does offer a possible reaction that produces no neutrons; the charged protons produced can be contained using electric and magnetic fields, which in turn results in direct electricity generation. 3He + 3He fusion is feasible as demonstrated in the laboratory and has immense advantages, but commercial viability is many years in the future.[58]
The amounts of helium-3 needed as a replacement for
A second-generation approach to controlled
He-3 power plants
There have been many claims about the capabilities of helium-3 power plants. According to proponents, fusion power plants operating on
The reality is not so clear-cut. The most advanced fusion programs in the world are inertial confinement fusion (such as National Ignition Facility) and magnetic confinement fusion (such as ITER and Wendelstein 7-X). In the case of the former, there is no solid roadmap to power generation. In the case of the latter, commercial power generation is not expected until around 2050.[61] In both cases, the type of fusion discussed is the simplest: D-T fusion. The reason for this is the very low Coulomb barrier for this reaction; for D+3He, the barrier is much higher, and it is even higher for 3He–3He. The immense cost of reactors like ITER and National Ignition Facility are largely due to their immense size, yet to scale up to higher plasma temperatures would require reactors far larger still. The 14.7 MeV proton and 3.6 MeV alpha particle from D–3He fusion, plus the higher conversion efficiency, means that more electricity is obtained per kilogram than with D-T fusion (17.6 MeV), but not that much more. As a further downside, the rates of reaction for helium-3 fusion reactions are not particularly high, requiring a reactor that is larger still or more reactors to produce the same amount of electricity.
Alternatives to He-3
To attempt to work around this problem of massively large power plants that may not even be economical with D-T fusion, let alone the far more challenging D–3He fusion, a number of other reactors have been proposed – the
Extraterrestrial
Moon
Materials on the
The primary objective of
In January 2006, the Russian space company
Not all writers feel the extraction of lunar helium-3 is feasible, or even that there will be a demand for it for fusion. Dwayne Day, writing in The Space Review in 2015, characterises helium-3 extraction from the Moon for use in fusion as magical thinking about an unproven technology, and questions the feasibility of lunar extraction, as compared to production on Earth.[74]
Gas giants
Mining gas giants for helium-3 has also been proposed.[75] The British Interplanetary Society's hypothetical Project Daedalus interstellar probe design was fueled by helium-3 mines in the atmosphere of Jupiter, for example.
See also
- List of elements facing shortage
Notes and references
- arXiv:astro-ph/0412380v1.
- ^ Ley, Willy (October 1966). "The Delayed Discovery". For Your Information. Galaxy Science Fiction. pp. 116–127.
- ^ Matson, John (12 Jun 2009). "Is MOON's Sci-Fi Vision of Lunar Helium 3 Mining Based in Reality?". Scientific American – News Blog. Retrieved 29 Aug 2017.
- ^ Close, Frank (August 2007). "Fears Over Factoids" (PDF). CERN Document Server. Physicsworld.com. Retrieved 8 July 2018.
- ^ Fa WenZhe; Jin YaQiu (December 2010). "Global inventory of Helium-3 in lunar regoliths estimated by a multi-channel microwave radiometer on the Chang-E 1 lunar satellite".
- ^ Slyuta, E. N.; Abdrakhimov, A. M.; Galimov, E. M. (March 12–16, 2007). The Estimation of Helium-3 Probable Reserves in Lunar Regolith (PDF). 38th Lunar and Planetary Science Conference. p. 2175.
- ^ .
- JSTOR 2935553.
- .
- ISBN 978-0-226-81304-2.
- ^ "Lawrence and His Laboratory: Episode: A Productive Error". Newsmagazine Publication. 1981. Archived from the original on 2017-05-10. Retrieved 2009-09-01.
- ^ Teragon's Summary of Cryogen Properties Teragon Research, 2005
- S2CID 123038029.
- .
- .
- .
- ^ a b Wittenberg 1994
- ^ a b Aldrich, L.T.; Nier, Alfred O. Phys. Rev. 74, 1590 – 1594 (1948). The Occurrence of He3 in Natural Sources of Helium. Page 1592, Tables I and II.
- S2CID 4358031.)
- ^ WebElements Periodic Table: Professional Edition: Helium: key information. Webelements.com. Retrieved on 2011-11-08.
- ^ NTP)
- ^ a b c d e Shea, Dana A.; Morgan, Daniel (22 December 2010). The Helium-3 Shortage: Supply, Demand, and Options for Congress (PDF) (Report). Congressional Research Service. 7-5700.
- US Department of the Interior. Report of Investigations 9302.
- S2CID 16754900.
- ^ Wittenberg 1994 p. 3, Table 1; p. 9.
- ^ Wittenberg 1994 Page A-1 citing Anderson 1993, "1200 metric tonne"
- ^ Wittenberg 1994 Page A-4 "1 kg (3He), pumping power would be 1.13×106 MWyr ... fusion power derived ... 19 MWyr"
- S2CID 3242002.
- ^ "Isotope Development & Production for Research and Applications (IDPRA)". US Department of Energy Office of Science. 18 October 2018. Retrieved 11 January 2019.
- ^
Lucas, L. L. & Unterweger, M. P. (2000). "Comprehensive Review and Critical Evaluation of the Half-Life of Tritium". PMID 27551621.
- ^ Nuclide safety data sheet: Hydrogen-3. ehso.emory.edu
- ^ Savannah River Tritium Enterprise: Fact Sheet
- ^ Charmian Schaller Accelerator Production of Tritium – That Could Mean 40 Years of Work. Los Alamos Monitor. March 1, 1998
- ^ Science for Democratic Action Vol. 5 No. 1. IEER. Retrieved on 2011-11-08;
- ^ Physics Projects Deflate for Lack of Helium-3. Spectrum.ieee.org. Retrieved on 2011-11-08.
- ^ Tritium Production Nuclear Regulatory Commission, 2005.
- ^ CA 2810716, Sur, Bhaskar; Rodrigo, Lakshman & Didsbury, Richard, "System and method for collecting 3He gas from heavy water nuclear reactors", published 30 September 2013, issued 2013 Archived 23 December 2015 at the Wayback Machine
- ^ "Basic policy on handling of the ALPS treated water" (PDF). Ministry of Economy, Trade and Industry. 13 April 2021.
- ^ "2020년도 원전주변 환경방사능 조사 및 평가보고서" [2020 Environmental Radiation Survey and Evaluation Report Around Nuclear Power Plant]. Korea Hydro & Nuclear Power. 26 April 2021. p. 25. (table 8)
- ^ A Modular Neutron Detector | Summer 2003| Los Alamos National Laboratory Archived 2008-05-03 at the Wayback Machine. Lanl.gov. Retrieved on 2011-11-08.
- ^ NCNR Neutron Spin Filters. Ncnr.nist.gov (2004-04-28). Retrieved on 2011-11-08.
- ^ 3He-spin-filters/ ILL 3He spin filters[permanent dead link]. Ill.eu (2010-10-22). Retrieved on 2011-11-08.
- .
- ^ Neutron Spin Filters: Polarized 3He. NIST.gov
- ^ Wald, Matthew L.. (2009-11-22) 3Helium.html?partner=rss&emc=rss Nuclear Bomb Detectors Stopped by Material Shortage. Nytimes.com. Retrieved on 2011-11-08.
- ^ "Office of Science" (PDF). Archived from the original (PDF) on 2014-07-26. Retrieved 2014-07-18.
- ^ Dilution Refrigeration. cern.ch
- doi:10.1002/cmr.1014.
- PMID 15502612.
- ^ MIT Achieves Breakthrough in Nuclear Fusion Aug 2017
- S2CID 106402331.
- ^ "Inertial Electrostatic Confinement Fusion". Retrieved 2007-05-06.
- ^ "Nuclear Fission and Fusion". Archived from the original on 2007-04-04. Retrieved 2007-05-06.
- ^ "The Fusion Reaction". Archived from the original on 2013-07-31. Retrieved 2007-05-06.
- ^ John Santarius (June 2006). "A Strategy for D – 3
He
Development" (PDF). Archived from the original (PDF) on 2007-07-03. Retrieved 2007-05-06. - ^ "Nuclear Reactions". Retrieved 2007-05-06.
- ^ John Santarius (September 28, 2004). "Lunar 3
He
and Fusion Power" (PDF). Archived from the original (PDF) on 2007-07-03. Retrieved 2007-05-06. - ^ Mark Williams (August 23, 2007). "Mining the Moon: Lab experiments suggest that future fusion reactors could use helium-3 gathered from the moon". MIT Technology Review. Retrieved 2011-01-25.
- ^ Date from the US Energy Information Administration
- ^ Paul DiMare (October 2004). "Mining The Moon". Popular Mechanics. Archived from the original on 2007-08-14. Retrieved 2007-05-06.
- ^ "ITER & Beyond". Archived from the original on 2009-05-20. Retrieved 2009-08-04.
- hdl:1721.1/29869.
- ^ FTI Research Projects :: 3He Lunar Mining Archived 2006-09-04 at the Wayback Machine. Fti.neep.wisc.edu. Retrieved on 2011-11-08.
- Bibcode:2007LPI....38.2175S.
- ^ Eric R. Hedman (January 16, 2006). "A fascinating hour with Gerald Kulcinski". The Space Review.
- ^ I.N. Sviatoslavsky (November 1993). "The challenge of mining He-3 on the lunar surface: how all the parts fit together" (PDF). Archived from the original (PDF) on 2019-01-20. Retrieved 2008-03-04. Wisconsin Center for Space Automation and Robotics Technical Report WCSAR-TR-AR3-9311-2.
- ^ "With He-3 on mind, India gets ready for lunar mission". The Times Of India. 2008-09-19.
- ^ Scientific Archived 2009-10-12 at the Wayback Machine. Isro.org (2008-11-11). Retrieved on 2011-11-08.
- ^ Luna C/I:: Chandrayaan-1 Payload Feature #2: Sub KeV Atom Reflecting Analyser (SARA). Luna-ci.blogspot.com (2008-11-12). Retrieved on 2011-11-08.
- ^ He asked for the moon-and got it. Chinadaily.com.cn (2006-07-26). Retrieved on 2011-11-08.
- ^ Russian Rocket Builder Aims for Moon Base by 2015, Reports Say. Associated Press (via space.com). 26 January 2006
- ^ James Oberg (February 6, 2006). "Moonscam: Russians try to sell the Moon for foreign cash".
- ^ Dwayne A. Day (March 5, 2007). "Death throes and grand delusions". The Space Review.
- ^ Day, Dwayne (September 28, 2015). "The helium-3 incantation". The Space Review. Retrieved 11 January 2019.
The belief in helium-3 mining is a great example of a myth that has been incorporated into the larger enthusiasm for human spaceflight, a magical incantation that is murmured, but rarely actually discussed.
- ^ Bryan Palaszewski. "Atmospheric Mining in the Outer Solar System" (PDF). Archived from the original (PDF) on 2009-03-27. NASA Technical Memorandum 2006-214122. AIAA–2005–4319. Prepared for the 41st Joint Propulsion Conference and Exhibit cosponsored by AIAA, ASME, SAE, and ASEE, Tucson, Arizona, July 10–13, 2005.
Bibliography
- D.M. Smith, T.W. Goodwin, J.A.Schiller (26 September 2003). "Challenges to the worldwide supply of helium in the next decade" (PDF). American Institute of Physics Conference Series. 49: 119–138. doi:10.1063/1.1774674. Retrieved 2015-03-08.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - L.J. Wittenberg (July 1994). "Non-Lunar 3He Resources" (PDF). Retrieved 2008-07-01.
{{cite journal}}: Cite journal requires|journal=(help) - H.H. Schmitt (2005). Return to the Moon: Exploration, Enterprise, and Energy in the Human Settlement of Space. Springer. ISBN 978-0-387-24285-9.
- J. Wilks (1967). The properties of liquid and solid helium. Oxford University Press.
- E. R. Dobbs (2000). Helium three. Oxford University Press.
- G. E. Volovik (1992). Exotic properties of superfluid 3He. World Scientific.
- W. P. Halperin, ed. (1990). Helium three. North-Holland.
- J. G. Daunt, ed. (1960). Helium three: proceedings of the Second Symposium on Liquid and Solid Helium Three, held at the Ohio State University, August 23--25, 1960. Ohio State University Press.
External links
- The Nobel Prize in Physics 2003, presentation speech
- Moon for Sale: A BBC Horizon documentary on the possibility of lunar mining of Helium-3
