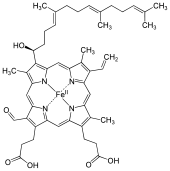
Heme A
| |||
| Names | |||
|---|---|---|---|
| Other names
Iron cytoporphyrin IX, formilporphyrin
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
| MeSH | Heme+a | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C49H56O6N4Fe | |||
| Molar mass | 852.837 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Heme A (or haem A) is a heme, a coordination complex consisting of a macrocyclic ligand called a porphyrin, chelating an iron atom. Heme A is a biomolecule and is produced naturally by many organisms. Heme A, often appears a dichroic green/red when in solution, is a structural relative of heme B, a component of hemoglobin, the red pigment in blood.
Relationship to other hemes
Heme A differs from
History
Heme A was first isolated by the German biochemist Otto Warburg in 1951 and shown by him to be the active component of the integral membrane metalloprotein cytochrome c oxidase.[3]
Stereochemistry
The final structural question of the exact geometric configuration about the first carbon at ring position 3 of ring I, the carbon bound to the hydroxyl group, has been shown to be the chiral S configuration.[4]
Like heme B, heme A is often attached to the apoprotein through a coordinate bond between the heme iron and a conserved amino acid side-chain. In the important respiratory protein
Heme A in the cytochrome a portion of cytochrome c oxidase, bound by two histidine residues (shown in pink)[6]
An example of a metalloprotein that contains heme A is cytochrome c oxidase. This very complicated protein contains heme A at two different sites, each with a different function. The iron of the heme A of
Both the
See also
- Heme
- Hemoprotein
- Cytochrome c oxidase (Complex IV of cellular respiration)
References
- ^
Caughey, W.S.; Smythe, G.A.; O'Keefe, D.H.; Maskasky, J.E.; Smith, M.L. (1975). "Heme A of Cytochrome c Oxidase". PMID 170266.
- .
- ^
Warburg, O; Gewitz H S. (1951). "Cytohämin aus Herzmuskel". PMID 14860765.
- PMID 16204889.
- PMID 14673090.
- ^ S2CID 37147458.
- ^
Shimokata K, Katayama Y, Murayama H, et al. (2007). "The proton pumping pathway of bovine heart cytochrome c oxidase". PMID 17360500.