Hemozoin

Haemozoin is a disposal product formed from the digestion of
Since the formation of hemozoin is essential to the survival of these parasites, it is an attractive target for
Discovery
Black-brown pigment was observed by
Later, in 1891, T. Carbone and W.H. Brown (1911) published papers linking the hemoglobin degradation with pigment production, describing the malaria pigment as a form of hematin and disproving the widely held idea that it is related to melanin. Brown observed that all melanins were bleaching rapidly with potassium permanganate, while with this reagent malarial pigment manifests not the slightest sign of a true bleach reaction.[5][6] The name "hemozoin" was proposed by Louis Westenra Sambon.[7] In the 1930s several authors identified hemozoin as a pure crystalline form of α-hematin and showed that the substance did not contain proteins within the crystals,[4] but no explanation for the solubility differences between malaria pigment and α-hematin crystals was given.[citation needed]
Formation
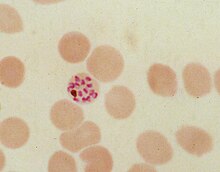
During its intraerythrocytic asexual reproduction cycle Plasmodium falciparum consumes up to 80% of the host cell
The malaria parasite, therefore, detoxifies the hematin, which it does by
Several mechanisms have been proposed for the production of hemozoin in Plasmodium, and the area is highly controversial, with membrane lipids,[17][18] histidine-rich proteins,[19] or even a combination of the two,[20] being proposed to catalyse the formation of hemozoin. Other authors have described a heme detoxification protein, which is claimed to be more potent than either lipids or histidine-rich proteins.[12] It is possible that many processes contribute to the formation of hemozoin.[21] The formation of hemozoin in other blood-feeding organisms is not as well-studied as in Plasmodium.[22] However, studies on Schistosoma mansoni have revealed that this parasitic worm produces large amounts of hemozoin during its growth in the human bloodstream. Although the shapes of the crystals are different from those produced by malaria parasites,[23] chemical analysis of the pigment showed that it is made of hemozoin.[24][25] In a similar manner, the crystals formed in the gut of the kissing bug Rhodnius prolixus during digestion of the blood meal also have a unique shape, but are composed of hemozoin.[26] Hz formation in R. prolixus midgut occurs at physiologically relevant physico-chemical conditions and lipids play an important role in heme biocrystallization. Autocatalytic heme crystallization to Hz is revealed to be an inefficient process and this conversion is further reduced as the Hz concentration increases.[27]
Several other mechanisms have been developed to protect a large variety of
Although β-hematin can be produced in assays spontaneously at low pH, the development of a simple and reliable method to measure the production of hemozoin has been difficult. This is in part due to the continued uncertainty over what molecules are involved in producing hemozoin, and partly from the difficulty in measuring the difference between aggregated or precipitated heme, and genuine hemozoin.[29] Current assays are sensitive and accurate, but require multiple washing steps so are slow and not ideal for high-throughput screening.[29] However, some screens have been performed with these assays.[30]
Structure

β-Hematin crystals are made of
Hemozoin crystals have a distinct
Inhibitors
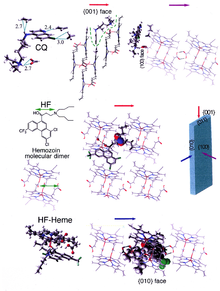
Hemozoin formation is an excellent drug target, since it is essential to malaria parasite survival and absent from the human host. The drug target hematin is host-derived and largely outside the genetic control of the parasite, which makes the development of drug resistance more difficult. Many clinically used drugs are thought to act by inhibiting the formation of hemozoin in the food vacuole.[37] This prevents the detoxification of the heme released in this compartment, and kills the parasite.[38]
The best-understood examples of such hematin biocrystallization inhibitors are quinoline drugs such as chloroquine and mefloquine. These drugs bind to both free heme and hemozoin crystals,[39] and therefore block the addition of new heme units onto the growing crystals. The small, most rapidly growing face is the face to which inhibitors are believed to bind.[40][41]
Role in pathophysiology
Hemozoin is released into the circulation during reinfection and phagocytosed in vivo and in vitro by host phagocytes and alters important functions in those cells. Most functional alterations were long-term postphagocytic effects,[42][43] including erythropoiesis inhibition shown in vitro.[44][45][46] In contrast, a powerful, short-term stimulation of oxidative burst by human monocytes was also shown to occur during phagocytosis of nHZ.[47] Lipid peroxidation non-enzymatically catalysed by hemozoin iron was described in immune cells.[48][49] Lipoperoxidation products, as hydroxyeicosatetraenoic acids (HETEs) and 4-hydroxynonenal (4-HNE), are functionally involved in immunomodulation.[43][46][49][50][51]
See also
References
- S2CID 207143167.
- ^ Meckel H (1847). "Ueber schwarzes Pigment in der Milz und dem Blute einer Geisteskranken". Zeitschrift für Psychiatrie. IV: 198–226.
- S2CID 40172296.
- ^ PMID 12435449.
- ^ Carbone T (1891). "Sulla natura chimica del pigmento malarico". G R Accad Med Torino. 39: 901–906.
- PMID 19867409.
- ^ Sinton JA, Ghosh BN (1934). "Studies of malarial pigment (haemozoin). Part I. Investigation of the action of solvents on haemozoin and the spectroscopical appearances observed in the solutions". Records of the Malaria Survey of India. 4: 15–42.
- PMID 9027746.
- PMID 19023444.
- PMID 6640106.
- PMID 2658089.
- ^ PMID 18437218.
- PMID 3119578.
- S2CID 4420567.
- S2CID 30446678. Archived from the originalon 2011-06-10.
- PMID 16115609.
- PMID 18373972.
- PMID 23894579.
- S2CID 3489880.
- PMID 12927780.
- PMID 23471987.
- PMID 18083247.
- PMID 1098591.
- PMID 11087932.
- PMID 19597543.
- PMID 16229843.
- PMID 20060043.
- PMID 11891128.
- ^ PMID 9586945.
- PMID 15720198.
- ^ Lemberg R, Legge JW (1949). "Hematin compounds and bile pigments". Interscience, New York.
- PMID 7833503.
- S2CID 42076.
- PMID 20074366.
- ]
- PMID 18390603. Archived from the original(PDF) on 2011-07-27. Retrieved 2009-12-19.
- PMID 11172673.
- PMID 24556123.
- PMID 8876229.
- PMID 18508124.
- PMID 19006402.
- PMID 9329987.
- ^ PMID 15356156.
- PMID 15075084.
- PMID 16804108.
- ^ PMID 20686121.
- PMID 21460246.
- S2CID 23594376.
- ^ PMID 25969702.
- PMID 34439507.
- PMID 25017964.
