Henipavirus
| Henipavirus | |
|---|---|

| |
| Colored transmission electron micrograph of a Hendra henipavirus virion (ca. 300 nm length)
| |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Negarnaviricota
|
| Class: | Monjiviricetes |
| Order: | Mononegavirales |
| Family: | Paramyxoviridae |
| Subfamily: | Orthoparamyxovirinae |
| Genus: | Henipavirus |
| Species | |
| |
Henipavirus is a
In 2009, RNA sequences of three novel viruses in phylogenetic relationship to known henipaviruses were detected in African straw-colored fruit bats (
Nipah and Hendra henipaviruses are both considered category C (USDA-HHS overlap) select agents.[11]
Structure


Henipavirions are
Embedded within the lipid membrane are spikes of F (fusion) protein trimers and G (attachment) protein tetramers. The function of the G protein (except in the case of MojV-G) is to attach the virus to the surface of a host cell via
Genome
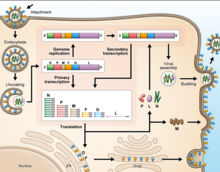
As all mononegaviral genomes, Hendra virus and Nipah virus genomes are non-segmented, single-stranded negative-sense RNA. Both genomes are 18.2 kb in length and contain six genes corresponding to six structural proteins.[17]
In common with other members of the Paramyxoviridae family, the number of nucleotides in the henipavirus genome is a multiple of six, consistent with what is known as the 'rule of six'.[18][19] Deviation from the rule of six, through mutation or incomplete genome synthesis, leads to inefficient viral replication, probably due to structural constraints imposed by the binding between the RNA and the N protein.
Three additional protein products are produced from the henipavirus P gene: V, W, and C. The V and W proteins are generated through an unusual process called
Life cycle
Cell receptor ephrin-B2, which is located on epithelial cells around smaller arteries, neurons, and smooth muscle cells, is targeted by the viral protein G.[25] Once the protein G binds to ephrin-B2, the viral protein F facilitates fusion with the host cell membrane and releases viral RNA into the host cell cytoplasm.[26] Upon entry, transcription of viral mRNA takes place using the viral RNA as a template. This process is started and stopped by the polymerase complex. Viral proteins are gathering in the cell as transcription occurs until the polymerase complex stops transcription and starts genome replication. Transcription of the viral RNA makes positive sense strands of RNA, which are then used as templates to make more negative sense viral RNA . Genome replication is halted before the viral particles can assemble to make a virion. Once the cell membrane is ready, new virions exit the host cell through budding.[27]
Vaccine
Henipaviruses have high mortality rates in mammalian hosts, both human and animal. Because of this, there is a need for immunization against HeV and NiV. The
The primary mechanism of protection against NiV and HeV induced by vaccination is thought to be neutralizing antibodies.[32] However, a number of preclinical vaccine studies in animal models of disease have identified that the cell-mediated immune response including CD8+ and CD4+ T-cells may play a role in protection.[33]
Causes of emergence
The emergence of henipaviruses parallels the emergence of other
There is evidence that habitat loss for flying foxes, both in South Asia and Australia (particularly along the east coast) as well as encroachment of human dwellings and agriculture into the remaining habitats, is creating greater overlap of human and flying fox distributions.[34]
Taxonomy
| Genus | Species | Virus (Abbreviation) |
| Henipavirus | Cedar henipavirus
|
Cedar virus (CedV) |
Ghanaian bat henipavirus
|
Kumasi virus (KV)
| |
Hendra henipavirus
|
Hendra virus (HeV) | |
Mojiang henipavirus
|
Mòjiāng virus (MojV)[3] | |
Nipah henipavirus
|
Nipah virus (NiV) | |
| Langya henipavirus |
See also
- Animal viruses
- Paramyxovirus
References
- PMID 31609197.
- ^ "ICTV Report Paramyxoviridae".
- ^ PMID 24865545.
- PMID 19046545.
- ^ Cheng, Amy (10 August 2022). "New Langya virus that may have spilled over from animals infects dozens". The Washington Post.
- ^ S2CID 251315935.
- ISBN 978-1-904455-22-6.
- ^ "Nipah yet to be confirmed, 86 under observation: Shailaja". OnManorama. Retrieved 4 June 2019.
- PMID 19636378.
- PMID 22531181.
- ^ "Federal Select Agent Program". www.selectagents.gov. 8 January 2021. Retrieved 15 January 2021.
- PMID 11334747.
- PMID 15998730.
- S2CID 4367038.
- PMID 20863312.
- PMID 18815311.
- PMID 11334745.
- PMID 14993656.
- PMID 9444980.
- PMID 21994589.
- PMID 35632678.
- PMID 15140960.
- PMID 23094089.
- PMID 27203423.
- PMID 15998730.
- .
- PMID 22552699
- ^ "Prioritizing diseases for research and development in emergency contexts". www.who.int. Retrieved 29 March 2023.
- ^ "Equivac® HeV". www.zoetis.com.au. Retrieved 29 March 2023.
- PMID 34358146.
- ^ Auro Vaccines LLC (16 November 2022). "A Phase 1 Randomized, Placebo-controlled, Observer-blind Trial to Assess the Safety and Immunogenicity of a Nipah Vaccine, HeV-sG-V (Hendra Virus Soluble Glycoprotein Vaccine), in Healthy Adults". ClinicalTrials.gov. PATH, Coalition for Epidemic Preparedness Innovations, Cincinnati Children's Hospital Medical Center (CCHMC).
- PMID 32991264.
- PMID 35744680.
- PMID 32226079.
- PMID 28389807.
- ^ "Zoonotic Langya virus found in China, CDC says - Taipei Times". 9 August 2022.
External links
- ICTV Report: Paramyxoviridae
- Disease card
- ViralZone: Henipavirus
- Henipavirus – Henipavirus Ecology Research Group (HERG) INFO
- Virus Pathogen Database and Analysis Resource (ViPR): Paramyxoviridae
