Hepatitis D
| Hepatitis D | |
|---|---|
| Other names | Hepatitis delta |
infectious disease | |
| Symptoms | Feeling tired, nausea and vomiting[1] |
| Complications | Cirrhosis[1] |
| Causes | Hepatitis D virus[1] |
| Diagnostic method | Immunoglobulin G[2] |
| Treatment | Antivirals, pegylated interferon alpha[2] |
| Medication | Bulevirtide |
Hepatitis D is a type of
HDV infecting a person with chronic hepatitis B (superinfection) is considered the most serious type of viral hepatitis due to its severity of complications.[7] These complications include a greater likelihood of experiencing liver failure in acute infections and a rapid progression to liver cirrhosis, with an increased risk of developing liver cancer in chronic infections.[8] In combination with hepatitis B virus, hepatitis D has the highest fatality rate of all the hepatitis infections, at 20%. A recent estimate from 2020 suggests that currently 48 million people are infected with this virus.[9]
| Hepatitis D | |
|---|---|
| Synonym | Delta hepatitis |
| Type of Virus | ssRNA |
| Incubation Period | 2–12 weeks |
| Transmission | Parenteral |
| Carrier State | Yes |
| Immunity
Passive immunization Active immunization |
Hyperimmune globulin
Vaccine (hepatitis B) |
Virology
| Hepatitis delta virus | |
|---|---|
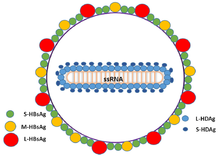
| |
| Schematic representation of the Hepatitis delta virus virion | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Ribozyviria |
| Family: | Kolmioviridae
|
| Genus: | Deltavirus |
| Species[10] | |
| |
Structure and genome
The hepatitis delta viruses, or HDV, are eight species of
The HDV genome is
Life cycle
Like hepatitis B, HDV gains entry into liver cells via the
The RNA polymerases treat the RNA genome as double-stranded DNA due to the folded rod-like structure it is in. Three forms of RNA are made; circular genomic RNA, circular complementary antigenomic RNA, and a linear polyadenylated antigenomic RNA, which is the mRNA containing the open reading frame for the HDAg. Synthesis of antigenomic RNA occurs in the nucleolus, mediated by RNA polymerase I, whereas synthesis of genomic RNA takes place in the nucleoplasm, mediated by RNA polymerase II.[26] HDV RNA is synthesized first as linear RNA that contains many copies of the genome. The genomic and antigenomic RNA contain a sequence of 85 nucleotides, the hepatitis delta virus ribozyme, that acts as a ribozyme, which self-cleaves the linear RNA into monomers. These monomers are then ligated to form circular RNA.[27][28]
Delta antigens
| Hepatitis delta virus delta antigen | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
A significant difference between
Antigenic loop infectivity
The HDV envelope protein has three of the HBV surface proteins anchored to it. The S region of the genome is most commonly expressed and its main function is to assemble subviral particles. HDV antigen proteins combine with the viral genome to form a ribonucleoprotein (RNP) which when enveloped with the subviral particles can form viral-like particles that are almost identical to mature HDV, but they are not infectious. Researchers had concluded that the determinant of infectivity of HDV was within the N-terminal pre-S1 domain of the large protein (L). It was found to be a mediator in binding to the cellular receptor. Researchers Georges Abou Jaoudé and Camille Sureau published an article in 2005 that studied the role of the antigenic loop, found in HDV envelope proteins, in the infectivity of the virus. The antigenic loop, like the N-terminal pre-S1 domain of the large protein, is exposed at the virion surface. Jaoudé and Sureau's study provided evidence that the antigenic loop may be an important factor in HDV entry into the host cell and by mutating parts of the antigenic loop, the infectivity of HDV may be minimized.[34]
Transmission
The routes of transmission of hepatitis D are similar to those for hepatitis B. Infection is largely restricted to persons at high risk of hepatitis B infection, particularly injecting drug users and persons receiving clotting factor concentrates. Worldwide more than 15 million people are co-infected. HDV is rare in most developed countries, and is mostly associated with intravenous drug use. However, HDV is much more common in the immediate Mediterranean region, sub-Saharan Africa, the Middle East, and the northern part of South America.[35] In all, about 20 million people may be infected with HDV.[36]
People at risk
As previously stated, patients previously diagnosed with hepatitis B are at risk for hepatitis D infection. Hepatitis D infection risk increases if a person uses injecting drugs, is a hemophiliac, if they are a hemodialysis patient, or through sexual contact with other infected persons.
Prevention
Vaccination against hepatitis B protects against hepatitis D viral infection as hepatitis D requires hepatitis B viral infection to be present in order to infect and replicate in people.[37][38] Universal vaccination against hepatitis B virus is recommended by the World Health Organization. The hepatitis B vaccine is routinely given soon after birth (usually within 24 hours) to protect against hepatitis B and D viral infection.[39]
Latex or polyurethane
Women who are pregnant or trying to become pregnant should undergo testing for HBV to know if they carry the virus, this will allow prevention strategies to be implemented during the birth of the child. The CDC recommends that all women who are pregnant be tested for hepatitis B viral infection and that all infants of women with HBV infection be given hepatitis B immune globulin (HBIG) and the hepatitis B vaccine within 12 hours of birth to prevent transmission of the virus from mother to child.[41]
Those who get
Diagnosis
Screening for hepatits D requires testing for anti-HDV antibodies, which indicate past exposure to the virus or current infection. If anti-HDV antibodies are present, then active HDV infection is confirmed by measuring hepatitis D RNA levels.
Treatment
Current established treatments for chronic hepatitis D include conventional or
In May 2020, the Committee for Medicinal Products for Human Use of the European Medicines Agency approved the antiviral Hepcludex (bulevirtide) to treat hepatitis D.[47] Bulevirtide binds and inactivates the sodium/bile acid cotransporter, blocking hepatitis D virus (as well as hepatitis B virus) from entering hepatocytes.[48][49] Bulevirtide may be given with pegylated interferon alpha as the two are thought to have a synergistic effect, leading to greater treatment response rates.[42]
In patients with HDV-related compensated Cirrhosis and clinically significant portal hypertension, the treatment with (bulevirtide) was safe, well tolerated and has led to a significant improvement in biochemical variables and an increase in liver function parameters.[50]
Other treatments for hepatitis D which are currently under development include pegylated interferon lambda (λ), which binds to receptors on the hepatocyte surface leading to an intracellular signaling cascade via the
Prognosis
Superinfections, in which hepatitis D viral infection occurs in someone who has chronic hepatitis B (as opposed to co-infection, in which a person is infected with hepatitis B and D simultaneously), are more likely to progress to chronic hepatitis D and are associated with a worse prognosis.[42] 90% of cases of chronic hepatitis D infection are thought to be due to superinfection in those already with hepatitis B.[42] Hepatitis B and D co-infection is likely to lead to acute hepatitis, but is usually self limited with regards to the hepatitis D infection.[42] Chronic hepatitis B and D is associated with a worse prognosis than chronic hepatitis B alone.[42] Infection with both viruses is characterized by a poor prognosis with 75% of those with chronic hepatitis D developing liver cirrhosis within 15 years and a much higher risk of developing liver cancer.[42] Persistent HDV viremia is the most important risk factor for disease progression in those with co-infection or superinfection.[42] Other factors that are responsible for a poor prognosis in chronic hepatitis D include male sex, older age at time of infection, alcohol use, diabetes, obesity and immunodeficiency.[42]
Epidemiology

HDV is prevalent worldwide. However, the prevalence is decreasing in many higher income countries due to hepatitis B vaccination programs (although rates remain high in some groups such as those who inject drugs or immigrants from HDV endemic reigions).[42][55] Infection with HDV is a major medical scourge in low income regions of the globe in which HBV prevalence remains high.[55] Currently the Amazon basin and low income regions of Asia and Africa have high rates of HDV, owing to concurrently high rates of HBV. Globally, five percent of those with chronic hepatitis B infection also have hepatitis D and 12.5% of people with HIV are also co-infected with hepatitis D.[56][42]
History
Hepatitis D virus was first reported in 1977 as a nuclear antigen in patients infected with HBV who had severe liver disease.[57] This nuclear antigen was then thought to be a hepatitis B antigen and was called the delta antigen. Subsequent experiments in chimpanzees showed that the hepatitis delta antigen (HDAg) was a structural part of a pathogen that required HBV infection to produce a complete viral particle.[58] The entire genome was cloned and sequenced in 1986. It was subsequently placed in its own genus: Deltavirus.[59][60]
Lábrea fever
| Lábrea fever | |
|---|---|
| Other names | Lábrea's black fever, Lábrea hepatitis, Santa Marta fever |
Infectious disease | |
| Usual onset | sudden |
| Duration | approx. 1 week |
| Prevention | HBV vaccination |
| Prognosis | death |
Lábrea fever is a lethal
Lábrea fever has a sudden onset, with
The main discovery of delta virus and HBV association was done by
Infected patients show extensive destruction of liver tissue, with steatosis of a particular type (microsteatosis, characterized by small fat droplets inside the cells), and infiltration of large numbers of inflammatory cells called morula cells, comprised mainly by macrophages containing delta virus antigens.[63]
In the 1987 Boca do Acre study, scientists did an epidemiological survey and reported delta virus infection in 24% of asymptomatic HBV carriers, 29% of acute nonfulminant hepatitis B cases, 74% of fulminant hepatitis B cases, and 100% of chronic hepatitis B cases.[61] The delta virus seems to be endemic in the Amazon region.[64]
Evolution
Three genotypes (I–III) were originally described. Genotype I has been isolated in Europe, North America, Africa and some Asia. Genotype II has been found in Japan, Taiwan, and Yakutia (Russia). Genotype III has been found exclusively in South America (Peru, Colombia, and Venezuela). Some genomes from Taiwan and the Okinawa islands have been difficult to type but have been placed in genotype 2. However it is now known that there are at least 8 genotypes of this virus (HDV-1 to HDV-8).[65] Phylogenetic studies suggest an African origin for this pathogen.[35]
An analysis of 36 strains of genotype 3 estimated that the most recent common ancestor of these strains originated around 1930.[66] This genotype spread exponentially from early 1950s to the 1970s in South America. The substitution rate was estimated to be 1.07×10−3 substitutions per site per year. Another study[67] found an overall evolution rate of 3.18×10−3 substitutions per site per year. The mutation rate varied with position : the hypervariable region evolved faster (4.55×10−3 substitutions per site per year) than the hepatitis delta antigen coding region (2.60×10−3 substitutions per site per year) and the autocatalytic region (1.11×10−3 substitutions per site per year). A third study suggested a mutation rate between 9.5×10−3 to 1.2×10−3 substitutions/site/year.[68]
Genotypes, with the exception of type 1, appear to be restricted to certain geographical areas: HDV-2 (previously HDV-IIa) is found in Japan, Taiwan and Yakutia; HDV-4 (previously HDV-IIb) in Japan and Taiwan; HDV-3 in the Amazonian region; HDV-5, HDV-6, HDV-7 and HDV-8 in Africa.[69] Genotype 8 has also been isolated from South America. This genotype is usually only found in Africa and may have been imported into South America during the slave trade.[70]
HDV-specific
Related species
A few other viruses with similarity to HDV have been described in species other than humans. Unlike HDV, none of them depend on a Hepadnaviridae (HBV family) virus to replicate. These agents have rod-like structure, a delta antigen, and a ribozyme.[72] HDV and all such relatives are classified in their own realm, Ribozyviria, by the International Committee on Taxonomy of Viruses.[11]
References
- ^ a b c "Hepatitis D | NIDDK". National Institute of Diabetes and Digestive and Kidney Diseases. Retrieved 10 September 2019.
- ^ a b "Hepatitis D". www.who.int. Retrieved 10 September 2019.
- ^ "Hepatitis (Viral) NIDDK". The National Institute of Diabetes and Digestive and Kidney Diseases. Retrieved 2020-06-19.
- PMID 14708706.
- PMID 30311870.
- S2CID 4368061.
- ^ "Hepatitis D". www.who.int. Retrieved 2020-09-20.
- PMID 10673308.
- PMID 31778167.
- ^ "ICTV 9th Report (2011) Deltavirus". International Committee on Taxonomy of Viruses (ICTV). Archived from the original on 30 January 2019. Retrieved 30 January 2019.
- ^ a b "Virus Taxonomy: 2020 Release". International Committee on Taxonomy of Viruses (ICTV). March 2021. Retrieved 5 August 2021.
- PMID 8245865.
- PMID 9687364.
- ^
- PMID 1712103.
- PMID 16903223.
- PMID 2374010.
- PMID 30260538.
- PMID 23150796.
- S2CID 23549907.
- PMID 20007265.
- PMID 1731113.
- S2CID 4393153.
- PMID 10668797.
- PMID 19246067.
- PMID 16775335.
- PMID 2492676.
- PMID 2648383.
- ^ PMID 2447291.
- PMID 12414985.
- PMID 15254184.
- PMID 16903218.
- PMID 8289368.
- PMID 16051838.
- ^ PMID 14963156.
- PMID 16364738.
- ^ "U.S. National Library of Medicine "Delta Agent (hepatitis D)"".
- ISBN 978-0-12-375147-8.
- ^ "Hepatitis B". www.who.int.
- ^ a b c "Hepatitis D - American Liver Foundation". liverfoundation.org. 23 May 2022.
- PMID 29939980.
- ^ S2CID 259354401.
- PMID 26253093.
- PMID 22161394.
- S2CID 205892640.
- S2CID 29142477.
- ^ "Hepcludex". European Medicines Agency. 26 May 2020.
- ^ Francisco EM (2020-05-29). "Hepcludex". European Medicines Agency. Archived from the original on 2020-06-15. Retrieved 2020-08-06.
- ^ "Bulevirtide - MYR Pharma". AdisInsight. Springer Nature Switzerland AG. Retrieved 2020-08-06.
MYR Pharmaceuticals receives Conditional Marketing Authorisation by the European Commission for bulevirtide in the European Union for Hepatitis B and D
- ^ Degasperi E, Anolli MP, Uceda Renteria SC, et al. Bulevirtide monotherapy for 48 weeks in patients with HDV-related compensated cirrhosis and clinically significant portal hypertension. J Hepatol. 2022;77(6):1525-1531. doi:10.1016/j.jhep.2022.07.016
- PMID 33889032.
- PMID 26189433.
- S2CID 52183556.
- .
- ^ PMID 26134842.
- ^ "Hepatitis D". www.who.int.
- PMID 75123.
- S2CID 83892580.
- S2CID 4265339.
- ^ Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA (2005). "Deltavirus". Eight Report of the International Committee on Taxonomy of Viruses. London: 735–8.
- ^ PMID 3599343.
- PMID 19605587.
- ^ Tommolino E, Piper MH, Sears D (2021-04-03). Anand BS (ed.). "Fatty Liver: Overview, Etiology, Epidemiology". Medscape.
- PMID 33005393.
- S2CID 31356.
- PMID 21645647.
- S2CID 22539505.
- PMID 27362848.
- PMID 17073101.
- PMID 21798297.
- PMID 30768983.
- PMID 32651267.
Bibliography
- Specter SC, ed. (1999). Viral Hepatitis: Diagnosis, Therapy, and Prevention. Humana Press. ISBN 0-89603-424-0.
- da Fonseca JC (2004). "[Hepatitis fulminant in Brazilian Amazon]". Revista da Sociedade Brasileira de Medicina Tropical. 37. 37 (Suppl 2): 93–5. PMID 15586904.
- Bensabath G, Soares M (2004). "[The evolution of knowledge about viral hepatitis in Amazon region: from epidemiology and etiology to the prophilaxy]". Revista da Sociedade Brasileira de Medicina Tropical. 37 (Suppl 2): 14–26. PMID 15586892.
- Fonseca JC, Souza RA, Brasil LM, Araújo JR, Ferreira LC (2004). "Fulminant hepatic failure in children and adolescents in Northern Brazil". Revista da Sociedade Brasileira de Medicina Tropical. 37 (1): 67–9. PMID 15042190.
External links
- World Health Organization Fact sheet on Hepatitis D
- ICTV Report: Deltavirus
- Viralzone: Deltavirus
- "Hepatitis delta virus". NCBI Taxonomy Browser. 12475.
