Heritability

Heritability is a statistic used in the fields of breeding and genetics that estimates the degree of variation in a phenotypic trait in a population that is due to genetic variation between individuals in that population.[1] The concept of heritability can be expressed in the form of the following question: "What is the proportion of the variation in a given trait within a population that is not explained by the environment or random chance?"[2]
Other causes of measured variation in a trait are characterized as
Heritability is estimated by comparing individual phenotypic variation among related individuals in a population, by examining the association between individual phenotype and genotype data,
Overview
Heritability measures the fraction of phenotype variability that can be attributed to genetic variation. This is not the same as saying that this fraction of an individual phenotype is caused by genetics. For example, it is incorrect to say that since the heritability of personality traits is about 0.6, that means that 60% of your personality is inherited from your parents and 40% comes from the environment. In addition, heritability can change without any genetic change occurring, such as when the environment starts contributing to more variation. As a case in point, consider that both genes and environment have the potential to influence intelligence. Heritability could increase if genetic variation increases, causing individuals to show more phenotypic variation, like showing different levels of intelligence. On the other hand, heritability might also increase if the environmental variation decreases, causing individuals to show less phenotypic variation, like showing more similar levels of intelligence. Heritability increases when genetics are contributing more variation or because non-genetic factors are contributing less variation; what matters is the relative contribution. Heritability is specific to a particular population in a particular environment. High heritability of a trait, consequently, does not necessarily mean that the trait is not very susceptible to environmental influences.[8] Heritability can also change as a result of changes in the environment, migration, inbreeding, or the way in which heritability itself is measured in the population under study.[9] The heritability of a trait should not be interpreted as a measure of the extent to which said trait is genetically determined in an individual.[10][11]
The extent of dependence of phenotype on environment can also be a function of the genes involved. Matters of heritability are complicated because genes may
Estimates of heritability use
A prerequisite for heritability analyses is that there is some population variation to account for. This last point highlights the fact that heritability cannot take into account the effect of factors which are invariant in the population. Factors may be invariant if they are absent and do not exist in the population, such as no one having access to a particular antibiotic, or because they are omni-present, like if everyone is drinking coffee. In practice, all human behavioral traits vary and almost all traits show some heritability.[13]
Definition
Any particular phenotype can be modeled as the sum of genetic and environmental effects:[14]
- Phenotype (P) = Genotype (G) + Environment (E).
Likewise the phenotypic variance in the trait – Var (P) – is the sum of effects as follows:
- Var(P) = Var(G) + Var(E) + 2 Cov(G,E).
In a planned experiment Cov(G,E) can be controlled and held at 0. In this case, heritability, is defined as[15]
H2 is the broad-sense heritability. This reflects all the genetic contributions to a population's phenotypic variance including additive,
A particularly important component of the genetic variance is the additive variance, Var(A), which is the variance due to the average effects (additive effects) of the alleles. Since each parent passes a single allele per locus to each offspring, parent-offspring resemblance depends upon the average effect of single alleles. Additive variance represents, therefore, the genetic component of variance responsible for parent-offspring resemblance. The additive genetic portion of the phenotypic variance is known as Narrow-sense heritability and is defined as
An upper case H2 is used to denote broad sense, and lower case h2 for narrow sense.
For traits which are not continuous but dichotomous such as an additional toe or certain diseases, the contribution of the various alleles can be considered to be a sum, which past a threshold, manifests itself as the trait, giving the liability threshold model in which heritability can be estimated and selection modeled.
Additive variance is important for
Example
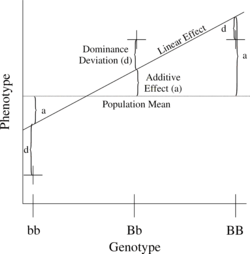
The simplest genetic model involves a single locus with two alleles (b and B) affecting one quantitative phenotype.
The number of B alleles can be 0, 1, or 2. For any genotype, (Bi,Bj), where Bi and Bj are either 0 or 1, the expected phenotype can then be written as the sum of the overall mean, a linear effect, and a dominance deviation (one can think of the dominance term as an interaction between Bi and Bj):
The additive genetic variance at this locus is the
where
There is a similar relationship for the variance of dominance deviations:
where
The linear regression of phenotype on genotype is shown in Figure 1.
Assumptions
Estimates of the total heritability of human traits assume the absence of epistasis, which has been called the "assumption of additivity". Although some researchers have cited such estimates in support of the existence of "
Estimating heritability
Since only P can be observed or measured directly, heritability must be estimated from the similarities observed in subjects varying in their level of genetic or environmental similarity. The
In non-human populations it is often possible to collect information in a controlled way. For example, among farm animals it is easy to arrange for a bull to produce offspring from a large number of cows and to control environments. Such
In classical quantitative genetics, there were two schools of thought regarding estimation of heritability.
One
The second was originally developed by R. A. Fisher and expanded at The University of Edinburgh, Iowa State University, and North Carolina State University, as well as other schools. It is based on the analysis of variance of breeding studies, using the intraclass correlation of relatives. Various methods of estimating components of variance (and, hence, heritability) from ANOVA are used in these analyses.
Today, heritability can be estimated from general pedigrees using linear mixed models and from genomic relatedness estimated from genetic markers.
Studies of human heritability often utilize adoption study designs, often with identical twins who have been separated early in life and raised in different environments. Such individuals have identical genotypes and can be used to separate the effects of genotype and environment. A limit of this design is the common prenatal environment and the relatively low numbers of twins reared apart. A second and more common design is the twin study in which the similarity of identical and fraternal twins is used to estimate heritability. These studies can be limited by the fact that identical twins are not completely genetically identical, potentially resulting in an underestimation of heritability.
In
Regression/correlation methods of estimation
The first school of estimation uses regression and correlation to estimate heritability.
Comparison of close relatives
In the comparison of relatives, we find that in general,
where r can be thought of as the coefficient of relatedness, b is the coefficient of regression and t is the coefficient of correlation.
Parent-offspring regression

Heritability may be estimated by comparing parent and offspring traits (as in Fig. 2). The slope of the line (0.57) approximates the heritability of the trait when offspring values are regressed against the average trait in the parents. If only one parent's value is used then heritability is twice the slope. (This is the source of the term "regression," since the offspring values always tend to regress to the mean value for the population, i.e., the slope is always less than one). This regression effect also underlies the DeFries–Fulker method for analyzing twins selected for one member being affected.[19]
Sibling comparison
A basic approach to heritability can be taken using full-Sib designs: comparing similarity between siblings who share both a biological mother and a father.[20] When there is only additive gene action, this sibling phenotypic correlation is an index of familiarity – the sum of half the additive genetic variance plus full effect of the common environment. It thus places an upper limit on additive heritability of twice the full-Sib phenotypic correlation. Half-Sib designs compare phenotypic traits of siblings that share one parent with other sibling groups.
Twin studies

Heritability for traits in humans is most frequently estimated by comparing resemblances between twins. "The advantage of twin studies, is that the total variance can be split up into genetic, shared or common environmental, and unique environmental components, enabling an accurate estimation of heritability".[21] Fraternal or dizygotic (DZ) twins on average share half their genes (assuming there is no assortative mating for the trait), and so identical or monozygotic (MZ) twins on average are twice as genetically similar as DZ twins. A crude estimate of heritability, then, is approximately twice the difference in correlation between MZ and DZ twins, i.e. Falconer's formula H2=2(r(MZ)-r(DZ)).
The effect of shared environment, c2, contributes to similarity between siblings due to the commonality of the environment they are raised in. Shared environment is approximated by the DZ correlation minus half heritability, which is the degree to which DZ twins share the same genes, c2=DZ-1/2h2. Unique environmental variance, e2, reflects the degree to which identical twins raised together are dissimilar, e2=1-r(MZ).
Analysis of variance methods of estimation
The second set of methods of estimation of heritability involves ANOVA and estimation of variance components.
Basic model
We use the basic discussion of Kempthorne.[14] Considering only the most basic of genetic models, we can look at the quantitative contribution of a single locus with genotype Gi as
where is the effect of genotype Gi and is the environmental effect.
Consider an experiment with a group of sires and their progeny from random dams. Since the progeny get half of their genes from the father and half from their (random) mother, the progeny equation is
Intraclass correlations
Consider the experiment above. We have two groups of progeny we can compare. The first is comparing the various progeny for an individual sire (called within sire group). The variance will include terms for genetic variance (since they did not all get the same genotype) and environmental variance. This is thought of as an error term.
The second group of progeny are comparisons of means of half sibs with each other (called among sire group). In addition to the
- ,
since environmental effects are independent of each other.
The ANOVA
In an experiment with sires and progeny per sire, we can calculate the following ANOVA, using as the genetic variance and as the environmental variance:
| Source | d.f. | Mean Square | Expected Mean Square |
|---|---|---|---|
| Between sire groups | |||
| Within sire groups |
The term is the intraclass correlation between half sibs. We can easily calculate . The expected mean square is calculated from the relationship of the individuals (progeny within a sire are all half-sibs, for example), and an understanding of intraclass correlations.
The use of ANOVA to calculate heritability often fails to account for the presence of
Model with additive and dominance terms
For a model with additive and dominance terms, but not others, the equation for a single locus is
where
is the additive effect of the ith allele, is the additive effect of the jth allele, is the dominance deviation for the ijth genotype, and is the environment.
Experiments can be run with a similar setup to the one given in Table 1. Using different relationship groups, we can evaluate different intraclass correlations. Using as the additive genetic variance and as the dominance deviation variance, intraclass correlations become linear functions of these parameters. In general,
- Intraclass correlation
where and are found as
P[
P[
Some common relationships and their coefficients are given in Table 2.
| Relationship | ||
|---|---|---|
| Identical Twins | ||
| Parent-Offspring | ||
| Half Siblings | ||
| Full Siblings | ||
| First Cousins | ||
| Double First Cousins |
Linear mixed models
A wide variety of approaches using linear mixed models have been reported in literature. Via these methods, phenotypic variance is partitioned into genetic, environmental and experimental design variances to estimate heritability. Environmental variance can be explicitly modeled by studying individuals across a broad range of environments, although inference of genetic variance from phenotypic and environmental variance may lead to underestimation of heritability due to the challenge of capturing the full range of environmental influence affecting a trait. Other methods for calculating heritability use data from
When a large, complex pedigree or another aforementioned type of data is available, heritability and other quantitative genetic parameters can be estimated by restricted maximum likelihood (REML) or Bayesian methods. The raw data will usually have three or more data points for each individual: a code for the sire, a code for the dam and one or several trait values. Different trait values may be for different traits or for different time points of measurement.
The currently popular methodology relies on high degrees of certainty over the identities of the sire and dam; it is not common to treat the sire identity probabilistically. This is not usually a problem, since the methodology is rarely applied to wild populations (although it has been used for several wild ungulate and bird populations), and sires are invariably known with a very high degree of certainty in breeding programmes. There are also algorithms that account for uncertain paternity.
The pedigrees can be viewed using programs such as Pedigree Viewer [1], and analyzed with programs such as ASReml, VCE [2], WOMBAT [3], MCMCglmm within the R environment [4] or the BLUPF90 family of programs [5].
Pedigree models are helpful for untangling confounds such as
Genomic heritability
When genome-wide genotype data and phenotypes from large population samples are available, one can estimate the relationships between individuals based on their genotypes and use a linear mixed model to estimate the variance explained by the genetic markers. This gives a genomic heritability estimate based on the variance captured by common genetic variants.[4] There are multiple methods that make different adjustments for allele frequency and linkage disequilibrium. Particularly, the method called High-Definition Likelihood (HDL) can estimate genomic heritability using only GWAS summary statistics,[5] making it easier to incorporate large sample size available in various GWAS meta-analysis.
Response to selection
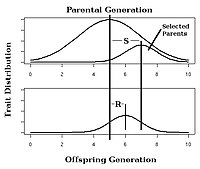
In selective breeding of plants and animals, the expected response to selection of a trait with known narrow-sense heritability can be estimated using the breeder's equation:[25]
In this equation, the Response to Selection (R) is defined as the realized average difference between the parent generation and the next generation, and the Selection Differential (S) is defined as the average difference between the parent generation and the selected parents.[14]: 1957 [26]
For example, imagine that a plant breeder is involved in a selective breeding project with the aim of increasing the number of kernels per ear of corn. For the sake of argument, let us assume that the average ear of corn in the parent generation has 100 kernels. Let us also assume that the selected parents produce corn with an average of 120 kernels per ear. If h2 equals 0.5, then the next generation will produce corn with an average of 0.5(120-100) = 10 additional kernels per ear. Therefore, the total number of kernels per ear of corn will equal, on average, 110.
Observing the response to selection in an artificial selection experiment will allow calculation of realized heritability as in Fig. 4.
Heritability in the above equation is equal to the ratio only if the genotype and the environmental noise follow
Controversies
This section may be unbalanced towards certain viewpoints. . (August 2016) |
Heritability estimates' prominent critics, such as
The controversy over heritability estimates is largely via their basis in
See also
- Behavioral genetics
- Heredity
- Heritability of IQ
References
- ^ Wray N, Visscher P (2008). "Estimating Trait Heritability". Nature Education. 1 (1): 29. Archived from the original on 2 August 2015. Retrieved 24 July 2015.
- )
- PMID 21167468.
- ^ (PDF) from the original on 2020-10-05. Retrieved 2020-09-06.
- ^ (PDF) from the original on 2021-04-15. Retrieved 2021-02-08.
- from the original on 2020-12-02. Retrieved 2020-12-24.
- (PDF) from the original on 2018-10-24. Retrieved 2019-12-05.
- S2CID 43435035.
- ^ (PDF) from the original on 2016-03-24. Retrieved 2015-08-28.
- PMID 29083200.
- S2CID 204981536.
- .
- review of: Hartl DL, Clark AG (2007). Principles of Population Genetics. Sunderland, MA: Sinauer and Associates. pp. xv + 652. ISBN 978-0-87893-308-2.
- review of: Hartl DL, Clark AG (2007). Principles of Population Genetics. Sunderland, MA: Sinauer and Associates. pp. xv + 652.
- (PDF) from the original on 19 October 2013. Retrieved 29 October 2013.
- ^ OCLC 422371269.
- ^ Stephen Downes and Lucas Matthews. "Heritability". Stanford Encyclopedia of Philosophy. Stanford University. Archived from the original on 2020-02-25. Retrieved 2020-02-20.
- PMID 22223662.
- PMID 26004471.
- PMID 13691636.
- S2CID 1172312.
- ISBN 978-0582243026.
- PMID 18157630.
- (PDF) from the original on 2020-10-05. Retrieved 2020-09-06.
- PMID 27382152.
- PMID 18454194.

- ISBN 978-0-7167-2056-0.
- ISBN 978-0-582-24302-6.
- PMID 16645027.
- ISBN 978-1-898059-47-9. Archivedfrom the original on 2017-07-19. Retrieved 2016-04-02.
- ISBN 978-0-8147-8723-6. Archivedfrom the original on 2020-10-05. Retrieved 2016-04-02.
- The Telegraph. Archivedfrom the original on 28 September 2011. Retrieved 4 April 2018.
- PMID 27906501.
- PMID 29440529.
...all complex human traits result from a combination of causes. If these causes interact, it is impossible to assign quantitative values to the fraction of a trait due to each, just as we cannot say how much of the area of a rectangle is due, separately, to each of its two dimensions. Thus, in the analyses of complex human phenotypes...we cannot actually find 'the relative importance of genes and environment in the determination of phenotype'.
- from the original on 20 May 2021. Retrieved 20 May 2021.
- ^ Tredoux, Gavan. "The Nature and Nurture of Rectangles." (2019).
- ^ S2CID 14737438.
- PMID 26413946.
- ISBN 978-1-317-60590-4. Archived(PDF) from the original on 2016-04-04. Retrieved 2016-04-02.
Further reading
- Lynch M, Walsh B (1998). Genetics and analysis of quantitative traits. Sunderland, Mass.: Sinauer Assoc. ISBN 978-0-87893-481-2.
- Johnson W, Penke L, Spinath FM (2011). "Understanding Heritability: What it is and What it is Not". European Journal of Personality. 25 (4): 287–294. S2CID 41842465.













































