Hofmann rearrangement
| Hofmann rearrangement | |
|---|---|
| Named after | August Wilhelm von Hofmann |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000410 |
The Hofmann rearrangement (Hofmann degradation) is the
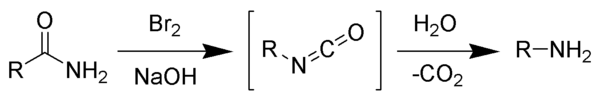
The reaction is named after its discoverer,
Mechanism
The reaction of bromine with sodium hydroxide forms sodium hypobromite in situ, which transforms the primary amide into an intermediate isocyanate. The formation of an intermediate nitrene is not possible because it implies also the formation of a hydroxamic acid as a byproduct, which has never been observed. The intermediate isocyanate is hydrolyzed to a primary amine, giving off carbon dioxide.[2]
- Base abstracts an acidic N-H proton, yielding an anion.
- The anion reacts with bromine in an α-substitution reaction to give an N-bromoamide.
- Base abstraction of the remaining amide proton gives a bromoamide anion.
- The bromoamide anion rearranges as the R group attached to the carbonyl carbon migrates to nitrogen at the same time the bromide ion leaves, giving an isocyanate.
- The isocyanate adds water in a nucleophilic addition step to yield a carbamic acid (aka urethane).
- The carbamic acid spontaneously loses CO2, yielding the amine product.
Variations
Several reagents can be substituted for bromine.
The intermediate isocyanate can be trapped with various nucleophiles to form stable carbamates or other products rather than undergoing decarboxylation. In the following example, the intermediate isocyanate is trapped by methanol.[7]
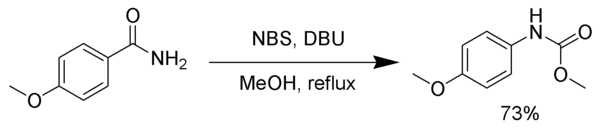
In a similar fashion, the intermediate isocyanate can be trapped by
The Hofmann Rearrangement also can be used to yield carbamates from
in good yields (≈70%).Applications
- In the preparation of anthranilic acid from phthalimide[10]
- Nicotinamide is converted into 3-Aminopyridine[11]
- The symmetrical structure[clarification needed] of α-phenyl propanamide does not change after Hofmann reaction.
- In the synthesis of gabapentin, beginning with the mono-amidation of 1,1-cyclohexane diacetic acid anhydride with ammonia to 1,1-cyclohexane diacetic acid mono-amide, followed by a Hofmann rearrangement[12]
See also
- Beckmann rearrangement
- Curtius rearrangement
- Iodoform reaction
- Lossen rearrangement
- Schmidt reaction
- Weerman degradation
References
- .
- ^ )
- ISBN 9780080359298.
- .
- .
- .
- .
- .
- .
- ISBN 3527306730..
- ; Collected Volumes, vol. 4, p. 45.
- ^ US 20080103334, "Process For Synthesis Of Gabapentin"
Bibliography
- Clayden, Jonathan (2007). Organic Chemistry. Oxford University Press Inc. pp. 1073. ISBN 978-0-19-850346-0.
- Fieser, Louis F. (1962). Advanced Organic Chemistry. Reinhold Publishing Corporation, Chapman & Hall, Ltd. pp. 499–501.

