Horner–Wadsworth–Emmons reaction
| Horner–Wadsworth–Emmons reaction | |
|---|---|
| Named after | Leopold Horner William S. Wadsworth William D. Emmons |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | wittig-horner-reaction |
| RSC ontology ID | RXNO:0000056 |
The Horner–Wadsworth–Emmons (HWE) reaction is a chemical reaction used in organic chemistry of stabilized phosphonate carbanions with aldehydes (or ketones) to produce predominantly E-alkenes.[1]
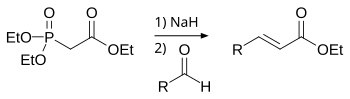
In 1958, Leopold Horner published a modified Wittig reaction using phosphonate-stabilized carbanions.[2][3] William S. Wadsworth and William D. Emmons further defined the reaction.[4][5]
In contrast to
Several reviews have been published.[6][7][8][9][10][11]
Reaction mechanism

The Horner–Wadsworth–Emmons reaction begins with the

The ratio of alkene
The
Stereoselectivity
The Horner–Wadsworth–Emmons reaction favours the formation of (E)-alkenes. In general, the more equilibration amongst intermediates, the higher the selectivity for (E)-alkene formation.
Disubstituted alkenes
Thompson and Heathcock have performed a systematic study of the reaction of methyl 2-(dimethylphosphono)acetate with various aldehydes.[16] While each effect was small, they had a cumulative effect making it possible to modify the stereochemical outcome without modifying the structure of the phosphonate. They found greater (E)-stereoselectivity with the following conditions:
- Increasing steric bulk of the aldehyde
- Higher reaction temperatures (23 °C over −78 °C)
- Li > Na > K salts
In a separate study, it was found that bulky phosphonate and bulky electron-withdrawing groups enhance E-alkene selectivity.
Trisubstituted alkenes
The steric bulk of the phosphonate and electron-withdrawing groups plays a critical role in the reaction of α-branched phosphonates with aliphatic aldehydes.[17]
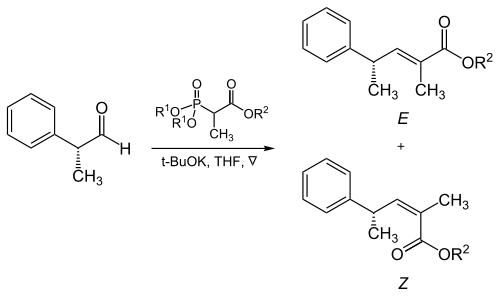
| R1 | R2 | Ratio of alkenes ( E : Z ) |
|---|---|---|
Methyl
|
Methyl | 5 : 95 |
| Methyl | Ethyl | 10 : 90 |
| Ethyl | Ethyl | 40 : 60 |
Isopropyl
|
Ethyl | 90 : 10 |
| Isopropyl | Isopropyl | 95 : 5 |
Olefination of ketones
The stereoselectivity of the Horner–Wadsworth–Emmons reaction of ketones is poor to modest.
Variations
Base sensitive substrates
Since many substrates are not stable to
Still modification
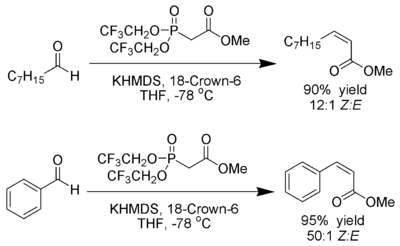
Ando has suggested that the use of electron-deficient phosphonates accelerates the elimination of the oxaphosphetane intermediates.[26]
See also
- Wittig reaction
- Michaelis–Arbuzov reaction
- Michaelis–Becker reaction
- Peterson reaction
- Tebbe olefination
References
- ^ Leopold Horner; Hoffmann, H. M. R.; Wippel, H. G. Ber. 1958, 91, 61–63.
- ^ Horner, L.; Hoffmann, H. M. R.; Wippel, H. G.; Klahre, G. Ber. 1959, 92, 2499–2505.
- )
- ^ Wadsworth, W. S., Jr.; Emmons, W. D. Organic Syntheses, Coll. Vol. 5, p. 547 (1973); Vol. 45, p. 44 (1965). (Article)
- ^ Wadsworth, W. S., Jr. Org. React. 1977, 25, 73–253. (Review)
- )
- ^ Kelly, S. E. Compr. Org. Synth. 1991, 1, 729–817. (Review)
- )
- Curr. Org. Chem.2012, 16, 2206–2230 (Review)
- ^ Bisceglia, J. A., Orelli, L. R. Curr. Org. Chem. 2015, 19, 744–775 (Review)
- ^ Larsen, R. O.; Aksnes, G. Phosphorus Sulfur 1983, 15, 218–219.
- J. Chem Soc., Chem. Commun.1970, 1308–09.
- )
- )
- )
- ^ Nagaoka, H.; Kishi, Y. Tetrahedron 1981, 37, 3873–3888.
- ^ Blanchette, M. A.; Choy, W.; Davis, J. T.; Essenfeld, A. P.; Masamune, S.; Roush, W. R.; Sakai, T. Tetrahedron Letters 1984, 25, 2183–2186.
- )
- ^ Paterson, I.; Yeung, K.-S.; Smaill, J. B. Synlett 1993, 774.
- ^ Simoni, D.; Rossi, M.; Rondanin, R.; Mazzali, A.; Baruchello, R.; Malagutti, C.; Roberti, M.; Invidiata, F. P. Org. Letters 2000, 2, 3765–3768.
- ^ Blasdel, L. K.; Myers, A. G. Org. Letters 2005, 7, 4281–4283.
- ^ Still, W. C.; Gennari, C. Tetrahedron Letters 1983, 24, 4405–4408.
- S2CID 216228029.
- ^ Patois, C.; Savignac, P.; About-Jaudet, E.; Collignon, N. Organic Syntheses, Coll. Vol. 9, p. 88 (1998); Vol. 73, p. 152 (1996). (Article)
- )
