Housane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Bicyclo[2.1.0]pentane | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| C5H8 | |||
| Molar mass | 68.119 g·mol−1 | ||
| Appearance | colorless liquid | ||
| Boiling point | 45.5 ′C | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
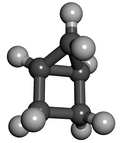
Housane or bicyclo[2.1.0]pentane is a saturated
Preparation
The first synthesis of housane was reported by
Housane can be prepared in several steps starting with cyclopentadiene. Other methods include photolysis of 2,3-diazabicyclo[2.2.1]hept-2-ene, pyrolysis of N-Phenyl-2-oxo-3-azabicyclo[2.2.1]heptane, and addition of methylene to cyclobutene.[2]
Structure and properties
The two rings are fused in a
Housane is easily attacked by bromine or iodine. In the presence of a platinum catalyst at room temperature, it is hydrogenated into cyclopentane. Reaction with hydrogen bromide at lower temperatures affords bromocyclopentane. Housane also reacts with lead tetraacetate, forming cis-1,3-diacetoxycyclopentane among other products.[1]
Housane is thermally quite stable, isomerizing to cyclopentene at 330 °C.[1]
References
- ^ ISSN 0009-2940.