Human metapneumovirus
| Human metapneumovirus | |
|---|---|
| |
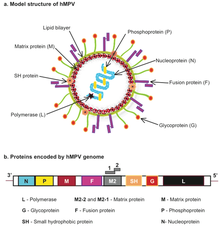
| Human metapneumovirus (hMPV) structure and genome | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Negarnaviricota
|
| Class: | Monjiviricetes |
| Order: | Mononegavirales |
| Family: | Pneumoviridae |
| Genus: | Metapneumovirus |
| Species: | Human metapneumovirus
|
Human metapneumovirus (HMPV or hMPV) is a negative-sense single-stranded
The peak age of hospitalization for infants with HMPV occurs between 6–12 months of age, slightly older than the peak of RSV, which is around 2–3 months. The clinical features and severity of HMPV are similar to those of RSV. HMPV is also an important cause of disease in older adults.
Taxonomy
| Genus | Species | Virus (abbreviation) | NCBI taxonomy ID |
|---|---|---|---|
| Metapneumovirus | Avian metapneumovirus | avian metapneumovirus (AMPV) | 38525 |
| Human metapneumovirus | human metapneumovirus (HMPV) | 162145 |
Discovery and naming
Human metapneumovirus was first discovered in 2001 in the Netherlands by Bernadette G. van den Hoogen and her colleagues.[5][6][7][8] hMPV was first detected in the respiratory secretions of 28 young children in the Netherlands and had initially stood out from other common respiratory viruses because the testing methods van den Hoogen et al. had tried using (immunological assays using virus-specific antibodies and PCR-based methods using virus genome-specific primers) were only able to test for known respiratory viruses and, therefore, were unable to identify the novel virus.[5] It was not until researchers began applying molecular biology techniques that the genetic characteristics and portions of the genomic sequences of the virus could be identified; these techniques included the randomly primed PCR technique which obtained the limited sequence data needed to reveal a clear relationship between this new virus and the avian pneumovirus.[5] It was this close relationship to AMPV that gave rise to this new virus being named human metapneumovirus[5] to reflect both its identity as a metapneumovirus and its use of humans as a host organism.
Epidemiology
HMPV was responsible for 12% of cases of acute
Genome
The
Life cycle and reproduction
hMPV is estimated to have a 3–6 day incubation period and is often most active during the later winter and spring seasons in temperate climates, overlapping with the RSV and influenza seasons and possibly allowing for repeated infection.[6] But because it is still a relatively new virus and has not yet been researched very heavily, hMPV and its replication cycle still have a lot of mystery surrounding them. However, researchers have been able to elucidate some principal steps of hMPV's replication cycle, basing their approach and experimentation on the current knowledge we have of the viral life cycles and reproductive measures of the rest of the Paramyxoviridae family.[38]
With that being said, it has been determined that the first step of the hMPV replication cycle is attachment to the host cell, specifically the
Next in the cycle is the fusion of the viral and host membranes which is likely mediated by the F protein.[8][38] Though the fusion mechanism is very similar to that of other Paramyxoviridae family members and involves conformational changes of the F protein, the mechanism for hMPV does not depend on the G protein for fusion like its family members, showing consistency with the previously mentioned idea that the G protein is not necessary for subsequent steps of the hMPV replication cycle.[8][38] Moreover, the fusion function of the F protein has been proven by its ability to bind to host cells via integrin αvβ1 using an Arginine-Glycine-Aspartate (RGD) motif, which is speculated to be the trigger for membrane fusion events.[8] One main difference between hMPV and other Paramyxoviridae viruses’ fusion mechanisms though is that hMPV's fusion events occur at acidic pH levels while other viruses’ fusion events occur at neutral pH levels; however, more research needs to be conducted in this area to get a better understanding of what is different about the hMPV fusion mechanism and why.[38] Although its specific function is uncertain, it is important to note the presence of the SH glycoprotein which seemingly does not have any effects on replication kinetics, cytopathic effects, or plaque formation of hMPV.[38]
After fusion, the viral
Virology
HMPV infects airway epithelial cells in the nose and lung. HMPV is thought to attach to the target cell via the glycoprotein (G) protein interactions with
Detection
The identification of HMPV has predominantly relied on
- detection of hMPV antigens in nasopharyngeal secretions by immunofluorescent-antibody test
- the use of immunofluorescence staining with monoclonal antibodiesto detect HMPV in nasopharyngeal secretions and shell vial cultures
- immunofluorescence assays for detection of hMPV-specific antibodies
- the use of polyclonal antibodies and direct isolation in cultured cells.
Distribution and habitat
Though hMPV was first discovered and identified in 2001,
So far, peak infection from hMPV in the northern hemisphere is in late winter and early spring, but it can be found globally across all continents[46] and its distribution is very complex and dynamic.[5] Researchers have found that hMPV is mostly localized and can differ significantly from community to community, allowing for the possibility of the strain in one location one year to be most similar to the strain in a different location the next year.[5] This phenomenon has actually been recorded with the virus strains in Australia in 2001; in France in 2000 and 2002; in Canada in 1999, 2000, 2001, and 2002; in Israel in 2002; and in the Netherlands in 2001 all being very closely related based on their F gene sequences.[5] There are at least two major genotypes of hMPV (A and B) that circulate during community outbreaks and each genotype has two of its own,[5] but as of now, it seems that no one strain is dominant over the others and none of them are known to cause varying levels of severity.[46]
hMPV is most likely spread from infected individuals to others through 1. secretions from coughing and sneezing, 2. close personal contact (ex. touching, shaking hands, etc), and 3. touching objects with viruses on them then touching your mouth, nose, or eyes.
Transmission
There are no conclusive studies to date; however, it is likely that transmission occurs by contact with contaminated secretions, via droplet, aerosol, or fomite vectors. Hospital-acquired infections with human metapneumovirus have been reported.[47] HMPV has been shown to circulate during fall and winter months with alternating predominance of a single subtype each year.[37]
Treatment
No treatment is yet known,[48] but ribavirin has shown effectiveness in an animal model.[49]
Evolution
Human metapneumovirus was first reported in 2001 and avian metapneumovirus in the 1970s. There are at least four lineages of human metapneumovirus—A1, A2, B1 and B2. Avian metapneumovirus has been divided into four subgroups—A, B, C and D. Bayesian estimates suggest that human metapneumovirus emerged 119–133 years ago and diverged from avian metapneumovirus around 1800.[52]
References
- ^ a b "ICTV Online (10th) Report".
- ^ PMID 11385510.
- ^ PMID 14749452.
- PMID 28389807.
- ^ PMID 16847085.
- ^ a b c d "Human Metapneumovirus". Centers for Disease Control and Prevention. 13 April 2023.
- ^ "Human Metapneumovirus (HMPV): Causes & Treatment". Cleveland Clinic.
- ^ PMID 21976607.
- PMID 25714161.
- PMID 16388486.
- PMID 15750086.
- ^ PMID 18680407.
- PMID 23945161.
- PMID 28369564.
- ^ PMID 12599052.
- PMID 27891060.
- PMID 21631655.
- PMID 25419459.
- PMID 27143659.
- PMID 27260872.
- ^ PMID 19064834.
- ^ PMID 22529314.
- PMID 24512531.
- PMID 16136455.
- PMID 15940152.
- S2CID 2395811.
- PMID 17140648.
- PMID 15338546.
- PMID 16412516.
- PMID 28379462.
- PMID 17407031.
- PMID 17674312.
- S2CID 2132679.
- ^ "Outbreaks of Human Metapneumovirus in Two Skilled Nursing Facilities — West Virginia and Idaho, 2011–2012". www.cdc.gov. Retrieved 2017-09-16.
- PMID 25056083.
- PMID 12033771.
- ^ PMID 33137178.
- ^ PMID 22074934.
- PMID 19164533.
- PMID 22238303.
- PMID 22933271.
- PMID 24478423.
- PMID 16971452.
- PMID 26629703.
- PMID 32809745, retrieved 2023-11-18
- ^ PMID 32809745.
- PMID 12781000.
- PMID 18516301.
- S2CID 21308968.
- ^ https://trials.modernatx.com/trials/NCT03392389 [dead link]
- ^ "Moderna to Present Data from Two of Its Prophylactic mRNA Vaccines at IDWeek 2019" (Press release). 2 October 2019.
- PMID 19008378.
External links
- ICTV Virus Taxonomy Profile: Pneumoviridae (December 2017)
- hMPV EIA kit (Biotrin, archived 13 February 2007)
- Human Metapneumovirus, hMPV (Biotrin, archived 11 September 2007)
- Virus Pathogen Database and Analysis Resource (ViPR): Paramyxoviridae[dead link]
