Hunter syndrome
| Hunter syndrome | |
|---|---|
 | |
| Patient with Hunter syndrome | |
| Specialty | Endocrinology |
| Symptoms | Skeletal abnormalities, hearing loss, retinal degeneration, enlarged liver and spleen |
| Complications | Upper airway disease; cardiovascular failure |
| Causes | Defiency of the enzyme iduronate-2-sulfatase |
| Differential diagnosis | Mucopolysaccharidosis type I; other mucopolysaccharidoses |
| Prognosis | In severe cases, death usually occurs by age 15. In attenuated cases, patients may survive into their 50s |
| Frequency | 1 in 100,000 to 150,000 male births[1] |
Hunter syndrome, or
The symptoms of Hunter syndrome are comparable to those of MPS I. It causes abnormalities in many organs, including the skeleton, heart, and respiratory system. In severe cases, this leads to death during the teenaged years. Unlike MPS I, corneal clouding is not associated with this disease.[1]
Signs and symptoms
Hunter syndrome may present with a wide variety of phenotypes. It has traditionally been categorized as either "mild" or "severe" depending on the presence of central nervous system symptoms, but this is an oversimplification. Patients with "attenuated" or "mild" forms of the disease may still have significant health issues. For severely affected patients, the clinical course is relatively predictable; patients will normally die at an early age. For those with milder forms of the disease, a wider variety of outcomes exist. Many live into their 20s and 30s, but some may have near-normal life expectancies. Cardiac and respiratory abnormalities are the usual cause of death for patients with milder forms of the disease.[2]
The symptoms of Hunter syndrome (MPS II) are generally not apparent at birth. Often, the first symptoms may include
The continued storage of GAGs leads to abnormalities in multiple organ systems. After 18 months, children with severe MPS II may experience developmental decline and progressive loss of skills.
The age at onset of symptoms and the presence or absence of behavioral disturbances are predictive factors of ultimate disease severity in very young patients. Behavioral disturbances can often mimic combinations of symptoms of
Genetics
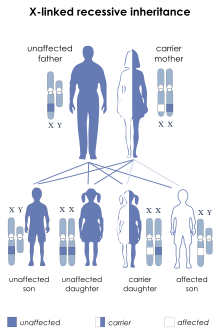
Since Hunter syndrome is an X-linked recessive disorder, it preferentially affects male patients. The IDS gene is located on the X chromosome. The IDS gene encodes for an enzyme called iduronate-2-sulfatase (I2S). A lack of this enzyme leads to a buildup of GAGs, which cause the symptoms of MPS II.[6]
If a female inherits one copy of the mutant
Pathophysiology
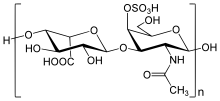
The
The biochemistry of Hunter syndrome is related to a problem in a part of the connective tissue known as the
In MPS II, the problem concerns the breakdown of two GAGs: dermatan sulfate and heparan sulfate. The first step in the breakdown of dermatan sulfate and heparan sulfate requires the lysosomal enzyme iduronate-2-sulfatase, or I2S. In people with MPS II, this enzyme is either partially or completely inactive. As a result, GAGs build up in cells throughout the body, particularly in tissues that contain large amounts of dermatan sulfate and heparan sulfate. The rate of GAGs buildup is not the same for all people with MPS II, resulting in a wide spectrum of medical problems.[citation needed]
Diagnosis
The first laboratory screening test for an MPS disorder is a
Treatment
Because of the wide variety of phenotypes, the treatment for this disorder is specifically determined for each patient. Until recently, no effective therapy for MPS II was available, so palliative care was used. Recent advances, though, have led to medications that can improve survival and well-being in people with MPS II.[citation needed]
Enzyme replacement therapy
Idursulfase, a purified form of the missing lysosomal enzyme, underwent clinical trial in 2006[6] and was subsequently approved by the United States Food and Drug Administration as an enzyme replacement treatment for MPS II. Idursulfase beta, another enzyme replacement treatment, was approved in Korea by the Ministry of Food and Drug Safety.
Recent advances in enzyme replacement therapy (ERT) with idursulfase have been proven to improve many signs and symptoms of MPS II, especially if started early in the disease. After administration, it can be transported into cells to break down GAGs, but as the medication cannot cross the blood–brain barrier, it is not expected to lead to cognitive improvement in patients with severe central nervous system symptoms. Even with ERT, treatment of various organ problems from a wide variety of medical specialists is necessary.[2]
Bone marrow and stem cell transplantation
Bone marrow transplantation and hematopoietic stem cell transplantation (HSCT) have been used as treatments in some studies.[7][8] While transplantation has provided benefits for many organ systems, it has not been shown to improve the neurological symptoms of the disease. Although HSCT has shown promise in the treatment of other MPS disorders, its results have been unsatisfactory so far in the treatment of MPS II. ERT has been shown to lead to better outcomes in MPS II patients.[2]
Gene editing therapy
In February 2019, medical scientists working with
Prognosis
Earlier onset of symptoms is linked to a worse prognosis. For children who exhibit symptoms between the ages of 2 and 4, death usually occurs by the age of 15 to 20 years. The cause of death is usually due to neurological complications, obstructive airway disease, and cardiac failure. If patients have minimal neurologic involvement, they may survive into their 50s or beyond.[1][6]
Epidemiology
An estimated 2,000 people have MPS II worldwide, 500 of whom live in the United States.[11]
A study in the United Kingdom indicated an incidence among males around one in 130,000 male live births.[12]
History
The syndrome is named after physician Charles A. Hunter (1873–1955), who first described it in 1917.[13][14]
Research
Beginning in 2010, a phase I/II clinical trial evaluated
In 2017, a 44-year-old[18] patient with MPS II was treated with gene therapy in an attempt to prevent further damage by the disease. This is the first case of gene editing being used in vivo in humans.[19] The study was extended to six patients in 2018.[20]
Society
On 24 July 2004, Andrew Wragg, 38, of Worthing, West Sussex, England, suffocated his 10-year-old son Jacob with a pillow, because of the boy's disabilities related to MPS II. A military security specialist, Wragg also claimed that he was under stress after returning from the Iraq War. He denied murdering Jacob, but pleaded guilty to manslaughter by reason of diminished capacity. Mrs Justice Anne Rafferty called the case "exceptional", gave Wragg a two-year prison sentence for manslaughter, then suspended his sentence for two years. Rafferty said "nothing [was] to be gained" from sending Wragg to prison for the crime.[21][22][23]
See also
- Hurler syndrome (MPS I)
- Sanfilippo syndrome (MPS III)
- Morquio syndrome (MPS IV)
- Prenatal testing
- Genetic counseling
References
- ^ a b c d "Mucopolysaccharidoses Fact Sheet". National Institute of Neurological Disorders and Stroke. 15 November 2017. Retrieved 11 May 2018.
- ^ PMID 18038146.
- ISBN 978-0-7216-2921-6.
- ^ McGraw-Hill. p. 117.
- S2CID 23119106.
- ^ PMID 16912578.
- PMID 19167723.
- PMID 24056375.
- AP News. Retrieved 7 February 2019.
- U.S. National Library of Medicine. Retrieved 7 February 2019.
- ^ LaTercera.com (in Spanish)[permanent dead link]
- S2CID 9667145.
- Who Named It?
- PMID 19979883.
- ^ "A Phase I/II, Randomized, Safety and Ascending Dose Ranging Study of Intrathecal Idursulfase-IT Administered in Conjunction With Intravenous Elaprase in Pediatric Patients With Hunter Syndrome and Cognitive Impairment". Clinicaltrials.gov. U.S. National Institutes of Health. 15 June 2009. Retrieved 22 July 2018.
- ^ "A Safety and Dose Ranging Study of Idursulfase (Intrathecal) Administration Via an Intrathecal Drug Delivery Device in Pediatric Patients With Hunter Syndrome Who Have Central Nervous System Involvement and Are Receiving Treatment With Elaprase® - Results". Clinicaltrials.gov. U.S. National Institutes of Health. 31 October 2013. Retrieved 20 July 2014.
- ^ "Study of Intrathecal Idursulfase-IT Administered in Conjunction With Elaprase® in Pediatric Patients With Hunter Syndrome and Early Cognitive Impairment (AIM-IT)". Clinicaltrials.gov. U.S. National Institutes of Health. July 2014. Retrieved 20 July 2014.
- ^ Marchione, Marilynn (15 November 2017). "US scientists try 1st gene editing in the body". Associated Press. Retrieved 16 November 2017.
- ^ Marchione, Marilynne (14 November 2017). "Scientists Attempt First Gene Editing Inside a Patient". Time. Archived from the original on 15 November 2017. Retrieved 15 November 2017.
- ^ Marchione, Mailynn (5 September 2018). "Early results boost hopes for historic gene editing attempt". AP News. Retrieved 6 September 2018.
- ^ NEWS.BBC.co.uk, "Father cleared of murdering son", BBC News
- ^ Guardian.co.uk, "Former SAS soldier who smothered terminally ill son walks free" The Guardian
- ^ NEWS.BBC.co.uk, "Review 'will clarify murder laws'" BBC News
External links
 Media related to Hunter syndrome at Wikimedia Commons
Media related to Hunter syndrome at Wikimedia Commons- GeneReview/NIH/UW entry on Mucopolysaccharidosis Type II
