Hydrochloric acid

| |||
| |||
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Chlorane[3]
| |||
| Other names | |||
| Identifiers | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.210.665 | ||
| EC Number |
| ||
| E number | E507 (acidity regulators, ...) | ||
PubChem CID
|
|||
| UNII | |||
| UN number | 1789 | ||
| Properties | |||
| HCl(aq) | |||
| Molar mass | 36.46 g·mol−1 | ||
| Appearance | Colorless, transparent liquid, fumes in air if concentrated | ||
| Odor | Pungent characteristic | ||
| Density | 1.18 g/cm3 | ||
| Melting point | Concentration-dependent – see table | ||
| Boiling point | Concentration-dependent – see table | ||
| log P | 0.00[4] | ||
| Acidity (pKa) | −5.9 (HCl gas)[5] | ||
| Pharmacology | |||
| A09AB03 (WHO) B05XA13 (WHO) | |||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger[6] | |||
| H290, H314, H335[6] | |||
| P260, P280, P303+P361+P353, P305+P351+P338[6] | |||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Other anions
|
|||
Related compounds
|
Hydrogen chloride | ||
| Supplementary data page | |||
| Hydrochloric acid (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride (HCl). It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical.[7][8]
Etymology
Because it was produced from
Gaseous HCl was called marine acid air. The name muriatic acid has the same origin (muriatic means "pertaining to brine or salt", hence
History
9th–10th century
In the early tenth century, the Persian physician and alchemist
Take equal parts of sweet salt, Bitter salt, Ṭabarzad salt, Andarānī salt, Indian salt, salt of Al-Qilī, and salt of Urine. After adding an equal weight of good crystallised Sal-ammoniac, dissolve by moisture, and distil (the mixture). There will distil over a strong water, which will cleave stone (sakhr) instantly.[13]
However, it appears that in most of his experiments al-Razi disregarded the gaseous products, concentrating instead on the color changes that could be effected in the residue.[14] According to Robert P. Multhauf, hydrogen chloride was produced many times without clear recognition that, by dissolving it in water, hydrochloric acid may be produced.[15]
11th–13th century
Drawing on al-Razi's experiments, the De aluminibus et salibus ("On Alums and Salts"), an eleventh- or twelfth-century Arabic text falsely attributed to al-Razi and
14th–15th century
Aqua regia
One important invention that resulted from the discovery of the mineral acids is aqua regia, a mixture of nitric acid and hydrochloric acid in a 1:3 proportion, capable of dissolving gold. This was first described in pseudo-Geber's De inventione veritatis ("On the Discovery of Truth", after c. 1300), where aqua regia was prepared by adding ammonium chloride to nitric acid.[18] The fact that aqua regia typically is defined as a mixture of nitric acid and hydrochloric acid does not mean that hydrochloric acid was discovered before or simultaneously with aqua regia. The isolation of hydrochloric acid happened about 300 years later. The production of hydrochloric acid itself (i.e., as an isolated substance rather than as already mixed with nitric acid) depended on the use of more efficient cooling apparatus, which would only develop in subsequent centuries.[19]
16th–17th century
Isolation of hydrochloric acid
From the point of view of Western history of chemistry, hydrochloric acid was the last of the three well-known mineral acids for which the method of its production appeared in the literature.[20] Recipes for its production started to appear in the late sixteenth century. The earliest recipes for the production of hydrochloric acid are found in Giovanni Battista Della Porta's (1535–1615) Magiae naturalis ("Natural Magic") and in the works of other contemporary chemists like Andreas Libavius (c. 1550–1616), Jean Beguin (1550–1620), and Oswald Croll (c. 1563–1609).[21] Among the historians who have written about this are German chemists Hermann Franz Moritz Kopp (1845) and Edmund Oscar von Lippmann (1938), mining engineer (and future U.S. president) Herbert Hoover with his wife geologist Lou Henry Hoover (1912), Dutch chemist Robert Jacobus Forbes (1948), American chemist Mary Elvira Weeks (1956), and British chemists F. Sherwood Taylor (1957) and J. R. Partington (1960). Italian chemist Ladislao Reti have summarized the result of their efforts thus:[22]
The first clear instance of the preparation of hydrochloric acid appears in the writings of Della Porta, (1589 and 1608), Libavius (1597), pseudo-Basil (1604), van Helmont (1646) and Glauber (1648). Less convincing earlier references are found in the Plichto of Rosetti (1540) and in Agricola (1558). As for the first practical method of preparation from vitriol and common salt, there is no doubt that pseudo-Basil precedes Glauber, but the latter has the unquestionable merit of having indicated the way of producing the acid later to be adopted by the chemical industry for large-scale operations.
— Ladislao Reti, How Old Is Hydrochloric Acid?
Dissolving metals
The knowledge of mineral acids such as hydrochloric acid would be of key importance to seventeenth-century chemists like Daniel Sennert (1572–1637) and Robert Boyle (1627–1691), who used their capability to rapidly dissolve metals in their demonstrations of the composite nature of bodies.[23]
Industrial developments
During the
In the 20th century, the Leblanc process was effectively replaced by the Solvay process without a hydrochloric acid by-product. Since hydrochloric acid was already fully settled as an important chemical in numerous applications, the commercial interest initiated other production methods, some of which are still used today. After 2000, hydrochloric acid is mostly made by absorbing by-product hydrogen chloride from industrial organic compounds production.[25][26][7]
Chemical properties
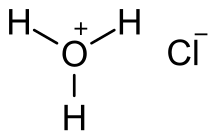
Gaseous hydrogen chloride is a molecular compound with a covalent bond between the hydrogen and chlorine atoms. In aqueous solutions dissociation is complete, with the formation of chloride ions and hydrated hydrogen ions (
The pKa value of hydrochloric acid in aqueous solution is estimated theoretically to be −5.9.
Physical properties
| Mass fraction |
Concentration | Density | Molarity
|
pH | Viscosity | Specific heat |
Vapour pressure |
Boiling point |
Melting point | |
|---|---|---|---|---|---|---|---|---|---|---|
| kg HCl/kg | kg HCl/m3 | Baumé | kg/L | mol/L | mPa·s | kJ/(kg·K) | kPa | °C | °C | |
| 10% | 104.80 | 6.6 | 1.048 | 2.87 | −0.5 | 1.16 | 3.47 | 1.95 | 103 | −18 |
| 20% | 219.60 | 13 | 1.098 | 6.02 | −0.8 | 1.37 | 2.99 | 1.40 | 108 | −59 |
| 30% | 344.70 | 19 | 1.149 | 9.45 | −1.0 | 1.70 | 2.60 | 2.13 | 90 | −52 |
| 32% | 370.88 | 20 | 1.159 | 10.17 | −1.0 | 1.80 | 2.55 | 3.73 | 84 | −43 |
| 34% | 397.46 | 21 | 1.169 | 10.90 | −1.0 | 1.90 | 2.50 | 7.24 | 71 | −36 |
| 36% | 424.44 | 22 | 1.179 | 11.64 | −1.1 | 1.99 | 2.46 | 14.5 | 61 | −30 |
| 38% | 451.82 | 23 | 1.189 | 12.39 | −1.1 | 2.10 | 2.43 | 28.3 | 48 | −26 |
| The reference temperature and pressure for the above table are 20 °C and 1 atmosphere (101.325 kPa). Vapour pressure values are taken from the International Critical Tables and refer to the total vapour pressure of the solution. | ||||||||||

Hydrochloric acid as the binary (two-component) mixture of HCl and H2O has a constant-boiling
Production
Hydrochloric acid is usually prepared industrially by dissolving hydrogen chloride in water. Hydrogen chloride can be generated in many ways, and thus several precursors to hydrochloric acid exist. The large-scale production of hydrochloric acid is almost always integrated with the industrial scale production of other chemicals, such as in the chloralkali process which produces hydroxide, hydrogen, and chlorine, the latter of which can be combined to produce HCl.[31][32]
Hydrogen chloride is produced by combining chlorine and hydrogen:
As the reaction is
Industrial market
Hydrochloric acid is produced in solutions up to 38% HCl (concentrated grade). Higher concentrations up to just over 40% are chemically possible, but the evaporation rate is then so high that storage and handling require extra precautions, such as pressurization and cooling. Bulk industrial-grade is therefore 30% to 35%, optimized to balance transport efficiency and product loss through evaporation. In the United States, solutions of between 20% and 32% are sold as muriatic acid. Solutions for household purposes in the US, mostly cleaning, are typically 10% to 12%, with strong recommendations to dilute before use. In the United Kingdom, where it is sold as "Spirits of Salt" for domestic cleaning, the potency is the same as the US industrial grade.[25] In other countries, such as Italy, hydrochloric acid for domestic or industrial cleaning is sold as "Acido Muriatico", and its concentration ranges from 5% to 32%.
Major producers worldwide include
Applications
Hydrochloric acid is a strong inorganic acid that is used in many industrial processes such as refining metal. The application often determines the required product quality.[25] Hydrogen chloride, not hydrochloric acid, is used more widely in industrial organic chemistry, e.g. for vinyl chloride and dichloroethane.[8]
Pickling of steel
One of the most important applications of hydrochloric acid is in the pickling of steel, to remove rust or iron oxide scale from iron or steel before subsequent processing, such as extrusion, rolling, galvanizing, and other techniques.[25][7] Technical quality HCl at typically 18% concentration is the most commonly used pickling agent for the pickling of carbon steel grades.
The
The steel pickling industry has developed hydrochloric acid regeneration processes, such as the spray roaster or the fluidized bed HCl regeneration process, which allow the recovery of HCl from spent pickling liquor. The most common regeneration process is the pyrohydrolysis process, applying the following formula:[25]
By recuperation of the spent acid, a closed acid loop is established.[7] The iron(III) oxide by-product of the regeneration process is valuable, used in a variety of secondary industries.[25]
Production of inorganic compounds
Akin to its use for pickling, hydrochloric acid is used to dissolve many metals, metal oxides and metal carbonates. The conversions are often depicted in simplified equations:
These processes are used to produce metal chlorides for analysis or further production.[31][32][7]
pH control and neutralization
Hydrochloric acid can be used to regulate the
In industry demanding purity (food, pharmaceutical, drinking water), high-quality hydrochloric acid is used to control the pH of process water streams. In less-demanding industry, technical quality hydrochloric acid suffices for
Regeneration of ion exchangers
High-quality hydrochloric acid is used in the regeneration of
Ion exchangers and demineralized water are used in all chemical industries, drinking water production, and many food industries.[25]
Laboratory use

Of the common strong
Hydrochloric acid is the preferred acid in
Other
Hydrochloric acid is used for a large number of small-scale applications, such as leather processing, household cleaning,[35] and building construction.[7] Oil production may be stimulated by injecting hydrochloric acid into the rock formation of an oil well, dissolving a portion of the rock, and creating a large-pore structure. Oil well acidizing is a common process in the North Sea oil production industry.[25]
Hydrochloric acid has been used for dissolving calcium carbonate, e.g. such things as de-scaling kettles and for cleaning mortar off brickwork. When used on brickwork the reaction with the mortar only continues until the acid has all been converted, producing calcium chloride, carbon dioxide, and water:
Many chemical reactions involving hydrochloric acid are applied in the production of food, food ingredients, and food additives. Typical products include aspartame, fructose, citric acid, lysine, hydrolyzed vegetable protein as food enhancer, and in gelatin production. Food-grade (extra-pure) hydrochloric acid can be applied when needed for the final product.[25][7]
Presence in living organisms

Gastric acid acts as a barrier against
The stomach itself is protected from the strong acid by the
Hydrochloric acid is also used by
Safety
Being a strong acid, hydrochloric acid is corrosive to living tissue and to many materials, but not to rubber. Typically, rubber protective gloves and related protective gear are used when handling concentrated solutions.[8]
Vapors or mists are a respiratory hazard, which can be partially mitigated by use of a respirator equipped with cartridges specifically designed to capture hydrochloric acid. Airborne acid is an irritant to the eyes, and may require the use of protective goggles or a facemask.[citation needed]
| Mass fraction |
Classification[41] | List of H-phrases |
|---|---|---|
| 10% ≤ C < 25% | Causes skin irritation, Causes serious eye irritation, | H315, H319 |
| C ≥ 10% | May cause respiratory irritation | H335 |
| C ≥ 25% | Causes severe skin burns and eye damage | H314 |
Legal status
Hydrochloric acid has been listed as a Table II precursor under the 1988 United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances because of its use in the production of heroin, cocaine, and methamphetamine.[42][43]
See also
- Chloride, inorganic salts of hydrochloric acid
- Hydrochloride, organic salts of hydrochloric acid from sodium hydroxide
References
- ^ a b "Hydrochloric Acid". Archived from the original on 15 October 2010. Retrieved 16 September 2010.
- ^ "spirits of salt". Retrieved 29 May 2012.
- ^ Favre HA, Powell WH, eds. (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. Cambridge: The Royal Society of Chemistry. p. 131.
- ^ "Hydrochloric acid". www.chemsrc.com.
- ^ S2CID 29697201.
- ^ a b c Sigma-Aldrich Co., Hydrochloric acid.
- ^ ISBN 978-0-08-037941-8.
- ^ ISBN 3527306730.
- ^ "Muriatic Acid" (PDF). PPG Industries. 2005. Archived from the original (PDF) on 2 July 2015. Retrieved 10 September 2010.
- ^ Gay-Lussac (1814) "Mémoire sur l'iode" (Memoir on iodine), Annales de Chemie, 91 : 5–160. From page 9: " ... mais pour les distinguer, je propose d'ajouter au mot spécifique de l'acide que l'on considère, le mot générique de hydro; de sorte que le combinaisons acide de hydrogène avec le chlore, l'iode, et le soufre porteraient le nom d'acide hydrochlorique, d'acide hydroiodique, et d'acide hydrosulfurique; ... " (... but in order to distinguish them, I propose to add to the specific suffix of the acid being considered, the general prefix hydro, so that the acidic combinations of hydrogen with chlorine, iodine, and sulfur will bear the name hydrochloric acid, hydroiodic acid, and hydrosulfuric acid; ...)
- ^ Multhauf 1966, pp. 141–142.
- OCLC 706947607. p. 333.
- ISBN 9781507778791. pp. 139–140.
- OCLC 977570829. pp. 141–142.
- ^ Multhauf 1966, p. 142. Multhauf refers to a number of recipes from the Kitāb al-Asrār translated by Ruska 1937, pp. 103–110, but does not seem to have noted the existence of the recipe in Ruska 1937, p. 182, §5 quoted above.
- ^ Multhauf 1966, pp. 160–162.
- ^ Multhauf 1966, pp. 162–163.
- ^ Karpenko, Vladimír; Norris, John A. (2002). "Vitriol in the History of Chemistry". Chemické listy. 96 (12): 997–1005. p. 1002.
- ^ Multhauf 1966, p. 204.
- ^ Reti 1965, p. 11.
- ^ Multhauf 1966, p. 208, note 29; cf. p. 142, note 79.
- JSTOR 27757245.
- ISBN 978-0226576961. p. 98.
- S2CID 221115202.
- ^ a b c d e f g h i j k l "Hydrochloric Acid". Chemicals Economics Handbook. SRI International. 2001. pp. 733.4000A–733.3003F.
- ^ ISBN 978-0-8122-1297-6.
- ISBN 978-0-13-014329-7.
- ISSN 1089-5639.
- ^ "Systemnummer 6 Chlor". Gmelins Handbuch der Anorganischen Chemie. Chemie Berlin. 1927.
- ^ "Systemnummer 6 Chlor, Ergänzungsband Teil B – Lieferung 1". Gmelins Handbuch der Anorganischen Chemie. Chemie Weinheim. 1968.
- ^ ISBN 978-0-8493-0481-1.
- ^ ISBN 978-0-07-049479-4.
- ^ a b Aspen Properties. binary mixtures modeling software (calculations by Akzo Nobel Engineering ed.). Aspen Technology. 2002–2003.
- ISBN 978-0-582-22628-9.
- ^ Simhon R (13 September 2003). "Household plc: really filthy bathroom". The Daily Telegraph. London. Archived from the original on 23 February 2009. Retrieved 31 March 2010.
- ^ ISBN 978-0-13-981176-0.
- ^ Haas E (6 December 2000). "Digestive Aids: Hydrochloric acid". healthy.net.
- ISBN 978-0-7216-8677-6.
- ^ Bowen R (18 March 2003). "Control and Physiologic Effects of Secretin". Colorado State University. Retrieved 16 March 2009.
- PMID 10639325. Retrieved 13 April 2024.
- ^ "Regulation (EC) No 1272/2008 of the European Parliament and of Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006". EUR-lex. Retrieved 16 December 2008.
- ^ List of precursors and chemicals frequently used in the illicit manufacture of narcotic drugs and psychotropic substances under international control (PDF) (Eleventh ed.). International Narcotics Control Board. January 2007. Archived from the original (PDF) on 2008-02-27.
- ^ Chemicals Required for the Illicit Manufacture of Drugs (PDF) (Report). UNDOC. 1998. Retrieved July 20, 2022.
External links
- NIST WebBook, general link
- Hydrochloric Acid – Part One and Hydrochloric Acid – Part Two at The Periodic Table of Videos(University of Nottingham)
- Calculators: surface tensions, and densities, molarities and molalities of aqueous HCl
General safety information
- EPA Hazard Summary
- Hydrochloric acid MSDS by Georgia Institute of Technology
- NIOSH Pocket Guide to Chemical Hazards