Hydrocortisone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | A-hydrocort, Cortef, Solu-cortef, others[1] |
| Other names | Cortisol; 11β,17α,21-Trihydroxypregn-4-ene-3,20-dione; 11β,17α,21-Trihydroxyprogesterone |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682206 |
| License data | |
| Pregnancy category |
|
| Drug class | Corticosteroid; Glucocorticoid; Mineralocorticoid |
| ATC code | |
| Legal status | |
| Legal status | |
Tmax)[11] | |
| Elimination half-life | 1.2–2.0 hours[11][12] |
| Duration of action | 8–12 hours[13] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Hydrocortisone is the name for the hormone
Side effects may include mood changes, increased risk of
Hydrocortisone was patented in 1936 and approved for medical use in 1941.
Medical uses
Hydrocortisone is the pharmaceutical term for cortisol used in oral administration, intravenous injection, or topical application. It is used as an immunosuppressive drug, given by injection in the treatment of severe allergic reactions such as anaphylaxis and angioedema, in place of prednisolone in patients needing steroid treatment but unable to take oral medication, and perioperatively in patients on long-term steroid treatment to prevent an adrenal crisis. It may also be injected into inflamed joints resulting from diseases such as gout.
It may be used topically for allergic rashes,
It may also be used rectally in suppositories to relieve the swelling, itch, and irritation in
It may be used as an acetate form (hydrocortisone acetate), which has slightly different pharmacokinetics and pharmacodynamics.[7][22]
-
Cortisol for injection
-
A tube of hydrocortisone cream, purchasedover the counter
-
Hydrocortisone 10 mg oral tablets (depicted a package for Russian market)
Pharmacology
Pharmacodynamics
Hydrocortisone is a corticosteroid, acting specifically as both a glucocorticoid and as a mineralocorticoid. That is, it is an agonist of the glucocorticoid and mineralocorticoid receptors.
Hydrocortisone has low
The
The total and/or free concentrations of cortisol/hydrocortisone required for various glucocorticoid effects have been determined.[11]
Pharmacokinetics
Absorption
The
The
Distribution
Most cortisol in the blood (all but about 4%) is bound to proteins, including corticosteroid binding globulin (CBG) and serum albumin. A pharmacokinetic review stated that 92% ± 2% (SD) (92–93%) of hydrocortisone is plasma protein-bound.[11] Free cortisol passes easily through cellular membranes.[32] Inside cells it interacts with corticosteroid receptors.[33]
Metabolism
Hydrocortisone is
Elimination
The
Chemistry
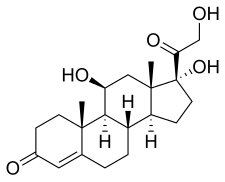
Hydrocortisone, also known as 11β,17α,21-trihydroxypregn-4-ene-3,20-dione, is a
Society and culture
Legal status
In March 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Efmody, intended for the treatment of congenital adrenal hyperplasia (CAH) in people aged twelve years and older.[37] The applicant for this medicinal product is Diurnal Europe BV.[37] Hydrocortisone (Efmody) was approved for medical use in the European Union, in May 2021, for the treatment of congenital adrenal hyperplasia (CAH) in people aged twelve years and older.[9]
Anti-competitive practices
In the UK, the
Research
COVID-19
Hydrocortisone was found to be effective in reducing mortality rate of critically ill COVID-19 patients when compared to other usual care or a placebo.[39]
References
- ^ a b c d e f g h "Hydrocortisone". Drugs.com. American Society of Health-System Pharmacists. 9 February 2015. Archived from the original on 20 September 2016. Retrieved 30 August 2016.
- ^ "Prescribing medicines in pregnancy database". Therapeutic Goods Administration (TGA). Archived from the original on 20 December 2016. Retrieved 21 February 2021.
- ^ "Hydrocortisone Notice of enforcement policy" (PDF). FDA. Archived (PDF) from the original on 12 March 2023. Retrieved 31 December 2022.
- ^ "Ala-cort- hydrocortisone cream". DailyMed. Archived from the original on 27 October 2020. Retrieved 21 February 2021.
- ^ "Ala-scalp- hydrocortisone lotion". DailyMed. Archived from the original on 21 April 2021. Retrieved 21 February 2021.
- ^ "Alkindi Sprinkle- hydrocortisone granule". DailyMed. Archived from the original on 10 February 2022. Retrieved 21 February 2021.
- ^ a b c "Anusol HC- hydrocortisone acetate suppository". DailyMed. Archived from the original on 10 February 2022. Retrieved 21 February 2021.
- ^ "Cortef- hydrocortisone tablet". DailyMed. Archived from the original on 17 April 2021. Retrieved 21 February 2021.
- ^ a b "Efmody EPAR". European Medicines Agency (EMA). 24 March 2021. Archived from the original on 14 June 2021. Retrieved 14 June 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Efmody Product information". Union Register of medicinal products. Archived from the original on 5 March 2023. Retrieved 3 March 2023.
- ^ S2CID 24458998.
- ^ S2CID 73248541.
- ^ PMID 23947590.
- ISBN 978-0-7817-1750-2. Archivedfrom the original on 14 September 2016.
- ISBN 978-1-284-05756-0.
- ^ "Hydrocortisone Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 20 September 2016. Retrieved 1 September 2016.
- ^ U.S. patent 2,183,589
- ISBN 978-3-527-60749-5. Archivedfrom the original on 10 January 2023. Retrieved 7 September 2020.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Hydrocortisone - Drug Usage Statistics". ClinCalc. Archived from the original on 12 April 2020. Retrieved 14 January 2024.
- PMID 18345402.
- ^ "Dexamethasone". drugs.com. Archived from the original on 21 June 2013. Retrieved 14 June 2013.
- S2CID 13986916.
- ISBN 978-3-318-05840-6. Archivedfrom the original on 27 April 2023. Retrieved 10 April 2023.
- OCLC 1020003143. Archivedfrom the original on 10 April 2023. Retrieved 10 April 2023.
- ^ PMID 8299376.
- S2CID 220439810.
- ^ S2CID 132864025.
- S2CID 33367289.
- S2CID 30076779.
- PMID 11250647.
- ISBN 978-1-4377-1753-2.
- S2CID 230716310.
- ^ ISBN 978-1-4757-2085-3. Archivedfrom the original on 8 September 2017.
- ^ ISBN 978-3-88763-075-1. Archivedfrom the original on 10 January 2023. Retrieved 19 June 2020.
- ^ a b "Efmody: Pending EC decision". European Medicines Agency (EMA). 25 March 2021. Archived from the original on 4 May 2021. Retrieved 27 March 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^
 This article incorporates text published under the British Open Government Licence: Competition and Markets Authority, Decision: Hydrocortisone tablets. Excessive and unfair pricing and Anti-competitive agreements, published 31 March 2022, accessed 1 June 2023
This article incorporates text published under the British Open Government Licence: Competition and Markets Authority, Decision: Hydrocortisone tablets. Excessive and unfair pricing and Anti-competitive agreements, published 31 March 2022, accessed 1 June 2023
- PMID 32876694.