Hydrogen fluoride
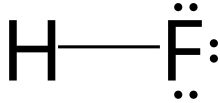
| |||
| |||
| Names | |||
|---|---|---|---|
| Other names
Fluorane
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.028.759 | ||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1052 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| HF | |||
| Molar mass | 20.006 g·mol−1 | ||
| Appearance | colourless gas or colourless liquid (below 19.5 °C) | ||
| Odor | unpleasant | ||
| Density | 1.15 g/L, gas (25 °C) 0.99 g/mL, liquid (19.5 °C) 1.663 g/mL, solid (–125 °C) | ||
| Melting point | −83.6 °C (−118.5 °F; 189.6 K) | ||
| Boiling point | 19.5 °C (67.1 °F; 292.6 K) | ||
| completely miscible (liquid) | |||
| Vapor pressure | 783 mmHg (20 °C)[1] | ||
| Acidity (pKa) | 3.17 (in water),
15 (in DMSO) [2] | ||
Conjugate acid
|
Fluoronium | ||
Conjugate base
|
Fluoride | ||
Refractive index (nD)
|
1.00001 | ||
| Structure | |||
| Linear | |||
| 1.86 D | |||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
8.687 J/g K (gas) | ||
Std enthalpy of (ΔfH⦵298)formation |
−13.66 kJ/g (gas) −14.99 kJ/g (liquid) | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Highly toxic, corrosive, irritant | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H300+H310+H330, H314 | |||
| P260, P262, P264, P270, P271, P280, P284, P301+P310, P301+P330+P331, P302+P350, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P321, P322, P330, P361, P363, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | none | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
17 ppm (rat, oral) | ||
LC50 (median concentration)
|
1276 ppm (rat, 1 hr) 1774 ppm (monkey, 1 hr) 4327 ppm (guinea pig, 15 min)[3] | ||
LCLo (lowest published)
|
313 ppm (rabbit, 7 hr)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 3 ppm[1] | ||
REL (Recommended)
|
TWA 3 ppm (2.5 mg/m3) C 6 ppm (5 mg/m3) [15-minute][1] | ||
IDLH (Immediate danger) |
30 ppm[1] | ||
| Related compounds | |||
Other anions
|
Hydrogen chloride Hydrogen bromide Hydrogen iodide Hydrogen astatide | ||
Other cations
|
Sodium fluoride Potassium fluoride Rubidium fluoride Caesium fluoride | ||
Related compounds
|
Water Ammonia | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydrogen fluoride (fluorane) is an
Hydrogen fluoride is an extremely dangerous gas, forming
History
In 1771
Structure and reactions

HF is diatomic in the gas-phase. As a liquid, HF forms relatively strong hydrogen bonds, hence its relatively high boiling point. Solid HF consists of zig-zag chains of HF molecules. The HF molecules, with a short covalent H–F bond of 95 pm length, are linked to neighboring molecules by intermolecular H–F distances of 155 pm.[4] Liquid HF also consists of chains of HF molecules, but the chains are shorter, consisting on average of only five or six molecules.[5]
Comparison with other hydrogen halides
Hydrogen fluoride does not boil until 20 °C in contrast to the heavier hydrogen halides, which boil between −85 °C (−120 °F) and −35 °C (−30 °F).[6][7][8] This hydrogen bonding between HF molecules gives rise to high viscosity in the liquid phase and lower than expected pressure in the gas phase.
Aqueous solutions
HF is
| HF and H2O similarities | |

|
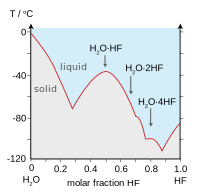
|
| Boiling points of the hydrogen halides (blue) and hydrogen chalcogenides (red): HF and H2O break trends. | Freezing point of HF/ H2O mixtures: arrows indicate compounds in the solid state. |
Aqueous solutions of HF are called
- 3 HF ⇌ H2F+ + HF−2
which forms an extremely acidic liquid (H0 = −15.1).
Reactions with Lewis acids
Like water, HF can act as a weak base, reacting with
Production
Hydrogen fluoride is typically produced by the reaction between sulfuric acid and pure grades of the mineral fluorite:[14]
- CaF2 + H2SO4 → 2 HF + CaSO4
About 20% of manufactured HF is a byproduct of fertilizer production, which generates hexafluorosilicic acid. This acid can be degraded to release HF thermally and by hydrolysis:
- H2SiF6 → 2 HF + SiF4
- SiF4 + 2 H2O → 4 HF + SiO2
Use
In general, anhydrous hydrogen fluoride is more common industrially than its aqueous solution,
Precursor to organofluorine compounds
HF reacts with chlorocarbons to give fluorocarbons. An important application of this reaction is the production of
- CHCl3 + 2 HF → CHClF2 + 2 HCl
Pyrolysis of chlorodifluoromethane (at 550-750 °C) yields TFE.
HF is a reactive solvent in the
1,1-Difluoroethane is produced by adding HF to acetylene using mercury as a catalyst.[15]
- HC≡CH + 2 HF → CH3CHF2
The intermediate in this process is vinyl fluoride or fluoroethylene, the monomeric precursor to polyvinyl fluoride.
Precursor to metal fluorides and fluorine
The electrowinning of aluminium relies on the electrolysis of aluminium fluoride in molten cryolite. Several kilograms of HF are consumed per ton of Al produced. Other metal fluorides are produced using HF, including uranium tetrafluoride.[14]
HF is the precursor to elemental fluorine, F2, by electrolysis of a solution of HF and potassium bifluoride. The potassium bifluoride is needed because anhydrous HF does not conduct electricity. Several thousand tons of F2 are produced annually.[16]
Catalyst
HF serves as a
Solvent
Hydrogen fluoride is an excellent solvent. Reflecting the ability of HF to participate in hydrogen bonding, even proteins and carbohydrates dissolve in HF and can be recovered from it. In contrast, most non-fluoride inorganic chemicals react with HF rather than dissolving.[17]
Health effects

Hydrogen fluoride is highly corrosive and a powerful contact poison. Exposure requires immediate medical attention.
References
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0334". National Institute for Occupational Safety and Health (NIOSH).
- ^ Evans, D. A. "pKa's of Inorganic and Oxo-Acids" (PDF). Retrieved June 19, 2020.
- ^ a b "Hydrogen fluoride". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- .
- PMID 15065271.
- ISBN 978-0-8014-0333-0.
- ISBN 978-1097774678.
- ^ Emsley, John (1981). "The hidden strength of hydrogen". New Scientist. 91 (1264): 291–292. Retrieved 25 December 2012.
- ISBN 0-7506-3365-4.
- ^ C. E. Housecroft and A. G. Sharpe Inorganic Chemistry, p. 221.
- ^ F. A. Cotton and G. Wilkinson Advanced Inorganic Chemistry, p. 111.
- ISBN 0-07-032768-8.
- ISBN 0-471-84997-9. p. 109.
- ^ ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - ^ ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - ISBN 978-3527306732.).
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - ^ Greenwood and Earnshaw, "Chemistry of the Elements", pp. 816–819.
- ^ a b Facts About Hydrogen Fluoride (Hydrofluoric Acid)
External links
- Fluorides, Hydrogen Fluoride, and Fluorine at ATSDR. Retrieved September 30, 2019
- CDC - NIOSH Pocket Guide to Chemical Hazards
- Hydrogen Fluoride Fact Sheet at Toxics Use Reduction Institute



