Hydroperoxide

Hydroperoxides or peroxols are
Properties
The O−O
Hydroperoxides are typically more volatile than the corresponding
- tert-BuOOH (b.p. 36 °C) vs tert-BuOH (b.p. 82-83 °C)
- CH3OOH (b.p. 46 °C) vs CH3OH (b.p. 65 °C)
- cumene hydroperoxide (b.p. 153 °C) vs cumyl alcohol (b.p. 202 °C)
Miscellaneous reactions
Hydroperoxides are mildly
Hydroperoxides can be reduced to alcohols with lithium aluminium hydride, as described in this idealized equation:
- 4 ROOH + LiAlH4 → LiAlO2 + 2 H2O + 4 ROH
This reaction is the basis of methods for analysis of organic peroxides.[5] Another way to evaluate the content of peracids and peroxides is the volumetric titration with alkoxides such as sodium ethoxide.[6] The phosphite esters and tertiary phosphines also effect reduction:
- ROOH + PR3 → OPR3 + ROH
Uses
Precursors to epoxides
"The single most important synthetic application of alkyl hydroperoxides is without doubt the metal-catalysed epoxidation of alkenes." In the
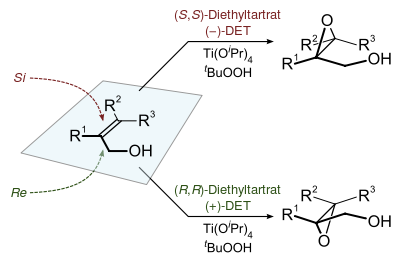
Of specialized interest, chiral epoxides are prepared using hydroperoxides as reagents in the Sharpless epoxidation.[8]
Production of cyclohexanone and caprolactone
Hydroperoxides are intermediates in the production of many organic compounds in industry. For example, the cobalt catalyzed oxidation of cyclohexane to cyclohexanone:[9]
- C6H12 + O2 → (CH2)5C=O + H2O
Drying oils, as found in many paints and varnishes, function via the formation of hydroperoxides.
Hock processes

Compounds with

Formation
By autoxidation
The most important (in a commercial sense) peroxides are produced by
- R−H + O2 → R−OOH
Although ether hydroperoxide often form adventitiously (i.e. autoxidation), they can be prepared in high yield by the acid-catalyzed addition of hydrogen peroxide to vinyl ethers:[13]
- C2H5OCH=CH2 + H2O2 → C2H5OCH(OOH)CH3
From hydrogen peroxide
Many industrial peroxides are produced using hydrogen peroxide. Reactions with aldehydes and ketones yield a series of compounds depending on conditions. Specific reactions include addition of hydrogen peroxide across the C=O double bond:
- R2C=O + H2O2 → R2C(OH)OOH
In some cases, these hydroperoxides convert to give cyclic diperoxides:
- [R2C(O2H)]2O2 → [R2C]2(O2)2 + 2 H2O
Addition of this initial adduct to a second equivalent of the carbonyl:
- R2C=O + R2C(OH)OOH → [R2C(OH)]2O2
Further replacement of alcohol groups:
- [R2C(OH)]2O2 + 2 H2O2 → [R2C(O2H)]2O2 + 2 H2O
Triphenylmethanol reacts with hydrogen peroxide gives the unusually stable hydroperoxide, Ph3COOH.[14]
Naturally occurring hydroperoxides
Many hydroperoxides are derived from fatty acids, steroids, and terpenes. The biosynthesis of these species is affected extensively by enzymes.
References
- ISBN 978-3527306732.
- .
- ISBN 978-0-470-77173-0.
- ISBN 978-3527306732.
- .
- .
- ^ .
- .
- ISBN 978-3527306732.
- ..
- ISBN 3-8274-1579-9(in German)
- ^ ISBN 3-527-29985-8pp. 206–207
- .
- PMID 16595187.