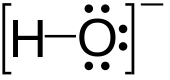
Hydroxide
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hydroxide
| |||
| Systematic IUPAC name
Oxidanide (not recommended) | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| OH− | |||
| Molar mass | 17.007 g·mol−1 | ||
Conjugate acid
|
Water | ||
Conjugate base
|
Oxide anion | ||
| Related compounds | |||
Related compounds
|
O2H+ OH• O22− H2O | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydroxide is a
Many
Hydroxide ion
The hydroxide ion is naturally produced from water by the self-ionization reaction:[1]
- H3O+ + OH− ⇌ 2H2O
The equilibrium constant for this reaction, defined as
- Kw = [H+][OH−][note 1]
has a value close to 10−14 at 25 °C, so the
4, which decreases the hydrogen cation concentration, which increases the hydroxide ion concentration. pOH can be kept at a nearly constant value with various buffer solutions

In
3O−
2 has been characterized in the solid state. This compound is centrosymmetric and has a very short hydrogen bond (114.5 pm) that is similar to the length in the bifluoride ion HF−
2 (114 pm).[2] In aqueous solution the hydroxide ion forms strong hydrogen bonds with water molecules. A consequence of this is that concentrated solutions of sodium hydroxide have high viscosity due to the formation of an extended network of hydrogen bonds as in hydrogen fluoride
In solution, exposed to air, the hydroxide ion reacts rapidly with atmospheric carbon dioxide, acting as an acid, to form, initially, the bicarbonate ion.
- OH− + CO2 ⇌ HCO−
3
The equilibrium constant for this reaction can be specified either as a reaction with dissolved carbon dioxide or as a reaction with carbon dioxide gas (see Carbonic acid for values and details). At neutral or acid pH, the reaction is slow, but is catalyzed by the enzyme carbonic anhydrase, which effectively creates hydroxide ions at the active site.
Solutions containing the hydroxide ion attack
The hydroxide ion can function as a typical electron-pair donor ligand, forming such complexes as tetrahydroxoaluminate/tetrahydroxidoaluminate [Al(OH)4]−. It is also often found in mixed-ligand complexes of the type [MLx(OH)y]z+, where L is a ligand. The hydroxide ion often serves as a bridging ligand, donating one pair of electrons to each of the atoms being bridged. As illustrated by [Pb2(OH)]3+, metal hydroxides are often written in a simplified format. It can even act as a 3-electron-pair donor, as in the tetramer [PtMe3(OH)]4.[4]
When bound to a strongly electron-withdrawing metal centre, hydroxide ligands tend to ionise into oxide ligands. For example, the bichromate ion [HCrO4]− dissociates according to
- [O3CrO–H]− ⇌ [CrO4]2− + H+
with a pKa of about 5.9.[5]
Vibrational spectra
The
When the OH group is bound to a metal ion in a coordination complex, an M−OH bending mode can be observed. For example, in [Sn(OH)6]2− it occurs at 1065 cm−1. The bending mode for a bridging hydroxide tends to be at a lower frequency as in [(bipyridine)Cu(OH)2Cu(bipyridine)]2+ (955 cm−1).[7] M−OH stretching vibrations occur below about 600 cm−1. For example, the tetrahedral ion [Zn(OH)4]2− has bands at 470 cm−1 (Raman-active, polarized) and 420 cm−1 (infrared). The same ion has a (HO)–Zn–(OH) bending vibration at 300 cm−1.[8]
Applications
Solutions containing the hydroxide ion are generated when a salt of a
- CO2−
3 + H2O ⇌ HCO−
3 + OH− (pKa2= 10.33 at 25 °C and zero ionic strength)
Although the base strength of sodium carbonate solutions is lower than a concentrated sodium hydroxide solution, it has the advantage of being a solid. It is also manufactured on a vast scale (42 million tonnes in 2005) by the
Bauxite, a basic hydroxide of aluminium, is the principal ore from which the metal is manufactured.[11] Similarly, goethite (α-FeO(OH)) and lepidocrocite (γ-FeO(OH)), basic hydroxides of iron, are among the principal ores used for the manufacture of metallic iron.[12]
Inorganic hydroxides
Alkali metals
Aside from NaOH and KOH, which enjoy very large scale applications, the hydroxides of the other alkali metals also are useful.
- 2 LiOH + CO2 → Li2CO3 + H2O
The hydroxide of lithium is preferred to that of sodium because of its lower mass.
Alkaline earth metals


Water molecules attached to Be are omitted
The solubility in water of the other hydroxides in this group increases with increasing
3 + OH− illustrates the basicity of calcium hydroxide. Soda lime
Boron group elements
The simplest hydroxide of boron B(OH)3, known as boric acid, is an acid. Unlike the hydroxides of the alkali and alkaline earth hydroxides, it does not dissociate in aqueous solution. Instead, it reacts with water molecules acting as a Lewis acid, releasing protons.
- B(OH)3 + H2O ⇌ B(OH)−
4 + H+
A variety of oxyanions of boron are known, which, in the protonated form, contain hydroxide groups.[19]
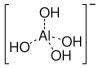
aluminate(III) ion
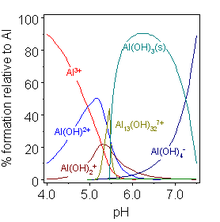
Aluminium hydroxide Al(OH)3 is amphoteric and dissolves in alkaline solution.[16]
- Al(OH)3 (solid) + OH− (aq) ⇌ Al(OH)−
4 (aq)
In the Bayer process[20] for the production of pure aluminium oxide from bauxite minerals this equilibrium is manipulated by careful control of temperature and alkali concentration. In the first phase, aluminium dissolves in hot alkaline solution as Al(OH)−
4, but other hydroxides usually present in the mineral, such as iron hydroxides, do not dissolve because they are not amphoteric. After removal of the insolubles, the so-called red mud, pure aluminium hydroxide is made to precipitate by reducing the temperature and adding water to the extract, which, by diluting the alkali, lowers the pH of the solution. Basic aluminium hydroxide AlO(OH), which may be present in bauxite, is also amphoteric.
In mildly acidic solutions, the hydroxo/hydroxido complexes formed by aluminium are somewhat different from those of boron, reflecting the greater size of Al(III) vs. B(III). The concentration of the species [Al13(OH)32]7+ is very dependent on the total aluminium concentration. Various other hydroxo complexes are found in crystalline compounds. Perhaps the most important is the basic hydroxide AlO(OH), a polymeric material known by the names of the mineral forms
Carbon group elements
Carbon forms no simple hydroxides. The
- C(OH)4 → HCO−
3 + H3O+ - HCO−
3 + H+ ⇌ H2CO3
Carbon dioxide is also known as carbonic anhydride, meaning that it forms by dehydration of carbonic acid H2CO3 (OC(OH)2).[23]
Silicic acid is the name given to a variety of compounds with a generic formula [SiOx(OH)4−2x]n.[24][25] Orthosilicic acid has been identified in very dilute aqueous solution. It is a weak acid with pKa1 = 9.84, pKa2 = 13.2 at 25 °C. It is usually written as H4SiO4, but the formula Si(OH)4 is generally accepted.[5][dubious ] Other silicic acids such as metasilicic acid (H2SiO3), disilicic acid (H2Si2O5), and pyrosilicic acid (H6Si2O7) have been characterized. These acids also have hydroxide groups attached to the silicon; the formulas suggest that these acids are protonated forms of polyoxyanions.
Few hydroxo complexes of germanium have been characterized. Tin(II) hydroxide Sn(OH)2 was prepared in anhydrous media. When tin(II) oxide is treated with alkali the pyramidal hydroxo complex Sn(OH)−
3 is formed. When solutions containing this ion are acidified, the ion [Sn3(OH)4]2+ is formed together with some basic hydroxo complexes. The structure of [Sn3(OH)4]2+ has a triangle of tin atoms connected by bridging hydroxide groups.[26] Tin(IV) hydroxide is unknown but can be regarded as the hypothetical acid from which stannates, with a formula [Sn(OH)6]2−, are derived by reaction with the (Lewis) basic hydroxide ion.[27]
Hydrolysis of Pb2+ in aqueous solution is accompanied by the formation of various hydroxo-containing complexes, some of which are insoluble. The basic hydroxo complex [Pb6O(OH)6]4+ is a cluster of six lead centres with metal–metal bonds surrounding a central oxide ion. The six hydroxide groups lie on the faces of the two external Pb4 tetrahedra. In strongly alkaline solutions soluble plumbate ions are formed, including [Pb(OH)6]2−.[28]
Other main-group elements
 |
 |
 |
 |
 |
 |
| Phosphorous acid | Phosphoric acid | Sulfuric acid | Telluric acid | Ortho-periodic acid | Xenic acid |
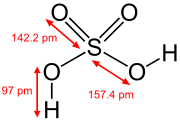
In the higher oxidation states of the pnictogens, chalcogens, halogens, and noble gases there are oxoacids in which the central atom is attached to oxide ions and hydroxide ions. Examples include phosphoric acid H3PO4, and sulfuric acid H2SO4. In these compounds one or more hydroxide groups can dissociate with the liberation of hydrogen cations as in a standard Brønsted–Lowry acid. Many oxoacids of sulfur are known and all feature OH groups that can dissociate.[29]
Telluric acid is often written with the formula H2TeO4·2H2O but is better described structurally as Te(OH)6.[30]
Ortho-periodic acid
As is common outside of the alkali metals, hydroxides of the elements in lower oxidation states are complicated. For example, phosphorous acid H3PO3 predominantly has the structure OP(H)(OH)2, in equilibrium with a small amount of P(OH)3.[32][33]
The oxoacids of
Transition and post-transition metals
The hydroxides of the
4 in strongly alkaline solution.[16]
Numerous mixed ligand complexes of these metals with the hydroxide ion exist. In fact, these are in general better defined than the simpler derivatives. Many can be made by deprotonation of the corresponding metal aquo complex.
- LnM(OH2) + B ⇌ LnM(OH) + BH+ (L = ligand, B = base)
Basic salts containing hydroxide
In some cases, the products of partial hydrolysis of metal ion, described above, can be found in crystalline compounds. A striking example is found with
The mineral
Structural chemistry
The hydroxide ion appears to rotate freely in crystals of the heavier alkali metal hydroxides at higher temperatures so as to present itself as a spherical ion, with an effective
The hydroxide ion displays cylindrical symmetry in hydroxides of divalent metals Ca, Cd, Mn, Fe, and Co. For example, magnesium hydroxide Mg(OH)2 (brucite) crystallizes with the cadmium iodide layer structure, with a kind of close-packing of magnesium and hydroxide ions.[39][42]
The
All theseA number of mixed hydroxides are known with stoichiometry A3MIII(OH)6, A2MIV(OH)6, and AMV(OH)6. As the formula suggests these substances contain M(OH)6 octahedral structural units.[46] Layered double hydroxides may be represented by the formula [Mz+
1−xM3+
x(OH)
2]q+(Xn−)
q⁄n·yH
2O. Most commonly, z = 2, and M2+ = Ca2+, Mg2+, Mn2+, Fe2+, Co2+, Ni2+, Cu2+, or Zn2+; hence q = x.
In organic reactions
Potassium hydroxide and sodium hydroxide are two well-known reagents in organic chemistry.
Base catalysis
The hydroxide ion may act as a
- RC(O)CH2R' + B ⇌ RC(O)CH−R' + BH+
The base should have a pKa value not less than about 4 log units smaller, or the equilibrium will lie almost completely to the left.
The hydroxide ion by itself is not a strong enough base, but it can be converted in one by adding sodium hydroxide to ethanol
- OH− + EtOH ⇌ EtO− + H2O
to produce the ethoxide ion. The pKa for self-dissociation of ethanol is about 16, so the alkoxide ion is a strong enough base.[49] The addition of an alcohol to an aldehyde to form a hemiacetal is an example of a reaction that can be catalyzed by the presence of hydroxide. Hydroxide can also act as a Lewis-base catalyst.[50]
As a nucleophilic reagent

The hydroxide ion is intermediate in
hydrolysis under alkaline conditions (also known as base hydrolysis)- R1C(O)OR2 + OH− ⇌ R1CO(O)H + −OR2 ⇌ R1CO2− + HOR2
is an example of a nucleophilic acyl substitution with the hydroxide ion acting as a nucleophile.[52]
Early methods for manufacturing soap treated triglycerides from animal fat (the ester) with lye.
Other cases where hydroxide can act as a nucleophilic reagent are
Notes
- ^ [H+] denotes the concentration of hydrogen cations and [OH−] the concentration of hydroxide ions
- activity
- ^ pOH signifies the minus the logarithm to base 10 of [OH−], alternatively the logarithm of 1/[OH−]
- ^ In this context proton is the term used for a solvated hydrogen cation
- ^ In aqueous solution the ligands L are water molecules, but they may be replaced by other ligands
- ^ The name is not derived from "period", but from "iodine": per-iodic acid (compare iodic acid, perchloric acid), and it is thus pronounced per-iodic /ˌpɜːraɪˈɒdɪk/ PUR-eye-OD-ik, and not as /ˌpɪərɪ-/ PEER-ee-.
- ^ Crystal structures are illustrated at Web mineral: Gibbsite, Bayerite, Norstrandite and Doyleite
References
- S2CID 1081091. Archived from the original(PDF) on 2007-06-25. Retrieved 2017-10-25.
- ^ .
- PMID 20170203.
- ^ Greenwood, p. 1168
- ^ a b IUPAC SC-Database Archived 2017-06-19 at the Wayback Machine A comprehensive database of published data on equilibrium constants of metal complexes and ligands
- ISBN 978-0-471-16394-7.
- ^ Nakamoto, Part B, p. 57
- ^ Adams, D.M. (1967). Metal–Ligand and Related Vibrations. London: Edward Arnold. Chapter 5.
- ISBN 978-3527306732.
- ^ Kostick, Dennis (2006). "Soda Ash", chapter in 2005 Minerals Yearbook, United States Geological Survey.
- ISBN 978-0-19-850340-8.
- ISBN 978-0-19-850340-8.
- ^ Lew. Kristi., Acids and Bases (Essential Chemistry). Infobase Publishing (2009). p43.
- ^ Jaunsen, JR (1989). "The Behavior and Capabilities of Lithium Hydroxide Carbon Dioxide Scrubbers in a Deep Sea Environment". US Naval Academy Technical Report. USNA-TSPR-157. Archived from the original on 2009-08-24. Retrieved 2008-06-17.
{{cite journal}}: CS1 maint: unfit URL (link) - ^ Holleman, p. 1108
- ^ ISBN 0-8031-2066-4p. 100
- S2CID 93623985.
- ^ Housecroft, p. 241
- ^ Housectroft, p. 263
- ^ Bayer process chemistry
- ISBN 0-12-356786-6, p. 764
- ISSN 1097-461X.
- ^ Greenwood, p. 310
- ^ Greenwood, p. 346
- ISBN 0-471-02404-X
- ^ Greenwood, p. 384
- ^ Greenwood, pp. 383–384
- ^ Greenwood, p. 395
- ^ Greenwood, p. 705
- ^ Greenwood, p. 781
- ^ Greenwood, pp. 873–874
- S2CID 93718865.
- ^ Holleman, pp. 711–718
- ^ Greenwood, p. 853
- .
- ISBN 1-4020-1242-X, p. 4
- ^ a b c Wells, p. 561
- ^ Wells, p. 393
- ^ a b c d Wells, p. 548
- ISBN 0-19-851790-4, p. 276
- .
- .
- ISBN 0-470-58768-7pp. 15 ff
- .
- ^ Wells, p. 557
- ^ Wells, p. 555
- ISBN 978-0-444-98655-9.
- ISBN 0-02-390171-3.
- ISBN 978-0-07-050115-7.
- PMID 18236505.
- ^ Hardinger, Steven A. (2017). "Illustrated Glossary of Organic Chemistry: Saponification". Department of Chemistry & Biochemistry, UCLA. Retrieved April 10, 2023.
Bibliography
- Holleman, A.F.; Wiberg, E.; Wiberg, N. (2001). Inorganic Chemistry. Academic press. ISBN 978-0-12-352651-9.
- Housecroft, C. E.; Sharpe, A. G. (2008). Inorganic Chemistry (3rd ed.). Prentice Hall. ISBN 978-0-13-175553-6.
- ISBN 978-0-08-037941-8.
- Shriver, D.F; Atkins, P.W (1999). Inorganic Chemistry (3rd ed.). Oxford: Oxford University Press. ISBN 978-0-19-850330-9.
- Wells, A.F (1962). Structural Inorganic Chemistry (3rd. ed.). Oxford: Clarendon Press. ISBN 978-0-19-855125-6.


