Hydroxylamine
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Azinous acid
| |||
| Preferred IUPAC name
Hydroxylamine (only preselected[1]) | |||
Other names
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3DMet | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.029.327 | ||
| EC Number |
| ||
| 478 | |||
| KEGG | |||
| MeSH | Hydroxylamine | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| NH2OH | |||
| Molar mass | 33.030 g·mol−1 | ||
| Appearance | Vivid white, opaque crystals | ||
| Density | 1.21 g cm−3 (at 20 °C)[2] | ||
| Melting point | 33 °C (91 °F; 306 K) | ||
| Boiling point | 58 °C (136 °F; 331 K) /22 mm Hg (decomposes) | ||
| Soluble | |||
| log P | −0.758 | ||
| Acidity (pKa) | 6.03 ([NH3OH]+) | ||
| Basicity (pKb) | 7.97 | ||
| Structure | |||
| Tricoordinated at N, dicoordinated at O | |||
Trigonal pyramidal at N, bent at O
| |||
| 0.67553 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
46.47 J/(K·mol) | ||
Std molar
entropy (S⦵298) |
236.18 J/(K·mol) | ||
Std enthalpy of (ΔfH⦵298)formation |
−39.9 kJ/mol | ||
| Hazards | |||
| GHS labelling: | |||
    
| |||
| Danger | |||
| H200, H290, H302, H312, H315, H317, H318, H335, H351, H373, H400 | |||
| P201, P202, P234, P260, P261, P264, P270, P271, P272, P273, P280, P281, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P312, P314, P321, P322, P330, P332+P313, P333+P313, P362, P363, P372, P373, P380, P390, P391, P401, P403+P233, P404, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 129 °C (264 °F; 402 K) | ||
| 265 °C (509 °F; 538 K) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
408 mg/kg (oral, mouse); 59–70 mg/kg (intraperitoneal mouse, rat); 29 mg/kg (subcutaneous, rat)[3] | ||
| Safety data sheet (SDS) | ICSC 0661 | ||
| Related compounds | |||
Related hydroxylammonium salts
|
|||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Hydroxylamine (also known as hydroxyammonia) is an
History
Hydroxylamine was first prepared as hydroxylammonium chloride in 1865 by the German chemist Wilhelm Clemens Lossen (1838-1906); he reacted tin and hydrochloric acid in the presence of ethyl nitrate.[6] It was first prepared in pure form in 1891 by the Dutch chemist Lobry de Bruyn and by the French chemist Léon Maurice Crismer (1858-1944).[7][8] The coordination complex ZnCl2(NH2OH)2 (zinc dichloride di(hydroxylamine)), known as Crismer's salt, releases hydroxylamine upon heating.[9]
Production
Hydroxylamine or its
From nitric oxide
NH2OH is mainly produced as its
Raschig process
Another route to NH2OH is the
This anion is then
- N(OH)(SO−3)2 + H2O → NH(OH)(SO−3) + HSO−4
- 2 NH(OH)(SO−3) + 2 H2O → [NH3OH]2SO4 + SO2−4
Solid NH2OH can be collected by treatment with
A base then frees the hydroxylamine from the salt:
- [NH3OH]Cl + NaOCH2CH2CH2CH3 → NH2OH + NaCl + CH3CH2CH2CH2OH[4]
Other methods
- HNO3 + 3 H2 → NH2OH + 2 H2O
Hydroxylamine can also be produced by the reduction of nitrous acid or potassium nitrite with bisulfite:
- HNO2 + 2 HSO−3 → N(OH)(OSO−2)2 + H2O → NH(OH)(OSO−2) + HSO−4
- NH(OH)(OSO−2) + [H3O]+ → [NH3OH]+ + HSO−4 (100 °C, 1 h)
A direct production of hydroxylamine from molecular nitrogen is also possible in water plasma.[13]
Reactions
Hydroxylamine reacts with
atoms:- R−X + NH2OH → R−O−NH2 + HX
- R−X + NH2OH → R−NH−OH + HX
The reaction of NH2OH with an aldehyde or ketone produces an oxime.
This reaction is useful in the purification of ketones and aldehydes: if hydroxylamine is added to an aldehyde or ketone in solution, an oxime forms, which generally precipitates from solution; heating the precipitate with an inorganic acid then restores the original aldehyde or ketone.[14]
Oximes such as dimethylglyoxime are also employed as ligands.
NH2OH reacts with
- HO−S(=O)2−Cl + NH2OH → NH2−O−S(=O)2−OH + HCl
When heated, hydroxylamine explodes. A detonator can easily explode aqueous solutions concentrated above 80% by weight, and even 50% solution might prove detonable if tested in bulk.[16][17] In air, the combustion is rapid and complete:
- 4 NH2OH + O2 → 2 N2 + 6 H2O
Absent air, pure hydroxylamine requires stronger heating and the detonation does not complete combustion:
- 3 NH2OH → N2 + NH3 + 3 H2O
Partial
Conjugate oxoacids
Hydroxylamine yields via
More hydroxyl radicals substitute the hydrogen atoms to yield azonous acid & azorous acid.
Functional group

Similarly to amines, one can distinguish hydroxylamines by their degree of substitution: primary, secondary and tertiary. When stored exposed to air for weeks, secondary hydroxylamines degrade to nitrones.[19]
N-organylhydroxylamines, R−NH−OH, where R is an
- R−NH−OH (Zn, HCl) → R−NH2 + ZnO
Synthesis
Amine oxidation with benzoyl peroxide is the most common method to synthesize hydroxylamines. Care must be taken to prevent over-oxidation to a nitrone. Other methods include:
- Hydrogenation of an oxime
- Alkylating a precursor hydroxylamine
- Amine oxide pyrolysis (the Cope reaction)
Uses
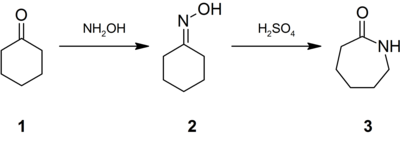
Conversion of cyclohexanone to caprolactam involving the Beckmann rearrangement.
Approximately 95% of hydroxylamine is used in the synthesis of cyclohexanone oxime, a precursor to Nylon 6.[10] The treatment of this oxime with acid induces the Beckmann rearrangement to give caprolactam (3).[21] The latter can then undergo a ring-opening polymerization to yield Nylon 6.[22]
Laboratory uses
Hydroxylamine and its salts are commonly used as reducing agents in myriad organic and inorganic reactions. They can also act as antioxidants for fatty acids.
High concentrations of hydroxylamine are used by biologists to introduce mutations by acting as a DNA nucleobase amine-hydroxylating agent.[23] In is thought to mainly act via hydroxylation of cytidine to hydroxyaminocytidine, which is misread as thymidine, thereby inducing C:G to T:A transition mutations.[24] But high concentrations or over-reaction of hydroxylamine in vitro are seemingly able to modify other regions of the DNA & lead to other types of mutations.[24] This may be due to the ability of hydroxylamine to undergo uncontrolled free radical chemistry in the presence of trace metals and oxygen, in fact in the absence of its free radical affects Ernst Freese noted hydroxylamine was unable to induce reversion mutations of its C:G to T:A transition effect & even considered hydroxylamine to be the most specific mutagen known.[25] Practically, it has been largely surpassed by more potent mutagens such as EMS, ENU, or nitrosoguanidine, but being a very small mutagenic compound with high specificity, it found some specialized uses such as mutation of DNA packed within bacteriophage capsids,[26] & mutation of purified DNA in vitro.[27]

This route also involves the Beckmann Rearrangement, like the conversion from cyclohexanone to caprolactam.
An
Some non-chemical uses include removal of hair from animal hides and photographic developing solutions.[2] In the semiconductor industry, hydroxylamine is often a component in the "resist stripper", which removes photoresist after lithography.
Hydroxylamine can also be used to better characterize the nature of a post-translational modification onto proteins. For example, poly(ADP-Ribose) chains are sensitive to hydroxylamine when attached to glutamic or aspartic acids but not sensitive when attached to serines.[28] Similarly, Ubiquitin molecules bound to serines or threonines residues are sensitive to hydroxylamine, but those bound to lysine (isopeptide bond) are resistant.[29]
Biochemistry
In biological nitrification, the oxidation of NH3 to hydroxylamine is mediated by the
Hydroxylamine can also be used to highly selectively cleave
Safety and environmental concerns
With a theoretical decomposition energy of about 5 kJ/g, hydroxylamine is an explosive, and aqueous solutions above 80% can be easily detonated by detonator or strong heating under confinement.[16] [17] At least two factories dealing in hydroxylamine have been destroyed since 1999 with loss of life.[33] It is known, however, that ferrous and ferric iron salts accelerate the decomposition of 50% NH2OH solutions.[34] Hydroxylamine and its derivatives are more safely handled in the form of salts.
It is an irritant to the
See also
References
- ISBN 978-0-85404-182-4.
- ^ ISBN 0-8493-0487-3.
- ISBN 978-1-903996-65-2.
- ^ a b c Greenwood and Earnshaw. Chemistry of the Elements. 2nd Edition. Reed Educational and Professional Publishing Ltd. pp. 431–432. 1997.
- ^ PMID 24523098.
- ^ W. C. Lossen (1865) "Ueber das Hydroxylamine" (On hydroxylamine), Zeitschrift für Chemie, 8 : 551-553. From p. 551: "Ich schlage vor, dieselbe Hydroxylamin oder Oxyammoniak zu nennen." (I propose to call it hydroxylamine or oxyammonia.)
- ^ C. A. Lobry de Bruyn (1891) "Sur l'hydroxylamine libre" (On free hydroxylamine), Recueil des travaux chimiques des Pays-Bas, 10 : 100-112.
- ^ L. Crismer (1891) "Préparation de l'hydroxylamine cristallisée" (Preparation of crystalized hydroxylamine), Bulletin de la Société chimique de Paris, series 3, 6 : 793-795.
- ISBN 9780470132401.
- ^ ISBN 978-3527306732.
- ^ James Hale, Arthur (1919). The Manufacture of Chemicals by Electrolysis (1st ed.). New York: D. Van Nostrand Co. p. 32. Retrieved 5 June 2014.
manufacture of chemicals by electrolysis hydroxylamine 32.
- ^ Osswald, Philipp; Geisler, Walter (1941). Process of preparing hydroxylamine hydrochloride (US2242477) (PDF). U.S. Patent Office.
- PMID 38378822.
- ^ Ralph Lloyd Shriner, Reynold C. Fuson, and Daniel Y. Curtin, The Systematic Identification of Organic Compounds: A Laboratory Manual, 5th ed. (New York: Wiley, 1964), chapter 6.
- ISBN 978-0-12-352651-9.
- ^ ISSN 0950-4230.
- ^ ISBN 9780081009710. Retrieved 2023-08-28.
- PMID 20449284.
- .
- ^ Smith, Michael and Jerry March. March's advanced organic chemistry : reactions, mechanisms, and structure. New York. Wiley. p. 1554. 2001.
- ISBN 978-0-19-927029-3.
- .
- PMID 16406304.
- ^ PMID 7042980.
- OCLC 851813793.
- PMID 4943557.
- ^ Forsberg, Susan. "Hydroxylamine Mutagenesis of plasmid DNA". PombeNet. University of Southern California. Retrieved 9 December 2021.
- PMID 34795260.
- PMID 31209050.
- PMID 8369308.
- PMID 27856762.
- )
- ^ Japan Science and Technology Agency Failure Knowledge Database Archived 2007-12-20 at the Wayback Machine.
- .
- ^ MSDS Sigma-Aldrich
Further reading
- Hydroxylamine[permanent dead link]
- Walters, Michael A. and Andrew B. Hoem. "Hydroxylamine." e-Encyclopedia of Reagents for Organic Synthesis. 2001.
- Schupf Computational Chemistry Lab
- M. W. Rathke A. A. Millard "Boranes in Functionalization of Olefins to Amines: 3-Pinanamine" Organic Syntheses, Coll. Vol. 6, p. 943; Vol. 58, p. 32. (preparation of hydroxylamine-O-sulfonic acid).