Hyperconjugation
In
Applications
Hyperconjugation can be used to rationalize a variety of chemical phenomena, including the
Effect on chemical properties
Hyperconjugation affects several properties.[6][10]
- hydrocarbons. For butadiene, this can be explained as normal conjugation of the two alkenyl parts. But for propyne, it is generally accepted that this is due to hyperconjugation between the alkyl and alkynyl parts.
- Dipole moments: The large increase in dipole moment of 1,1,1-trichloroethane as compared with chloroformcan be attributed to hyperconjugated structures.
- The heat of formation of molecules with hyperconjugation are greater than sum of their bond energies and the heats of hydrogenation per double bond are less than the heat of hydrogenation of ethylene.
- Stability of carbocations:
- (CH3)3C+ > (CH3)2CH+ > (CH3)CH2+ > CH3+
- The three C–H σ bonds of the methyl group(s) attached to the carbocation can undergo the stabilization interaction but only one of them can be aligned perfectly with the empty p-orbital, depending on the conformation of the carbon–carbon bond. Donation from the two misaligned C–H bonds is weaker.[11] The more adjacent methyl groups there are, the larger hyperconjugation stabilization is because of the increased number of adjacent C–H bonds.
Hyperconjugation in unsaturated compounds
Hyperconjugation was suggested as the reason for the increased stability of carbon-carbon double bonds as the degree of substitution increases. Early studies in hyperconjugation were performed by in the research group of
One set of experiments by Kistiakowsky involved collected heats of
A portion of Kistiakowsky's work involved a comparison of other unsaturated compounds in the form of CH2=CH(CH2)n-CH=CH2 (n=0,1,2). These experiments revealed an important result; when n=0, there is an effect of conjugation to the molecule where the ΔH value is lowered by 3.5
Despite the thoroughness of Kistiakowsky's work, it was not complete and needed further evidence to back up his findings. His work was a crucial first step to the beginnings of the ideas of hyperconjugation and conjugation effects.
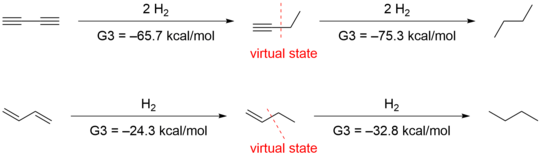
Stabilization of 1,3-butadiyne and 1,3-butadiene
The
Another group led by Houk
Deleting the hyperconjugative interactions gives virtual states that have energies that are 4.9 and 2.4 kcal/mol higher than those of
Trends in hyperconjugation
A relatively recent work (2006) by Fernández and Frenking (2006) summarized the trends in hyperconjugation among various groups of acyclic molecules, using energy decomposition analysis or EDA. Fernández and Frenking define this type of analysis as "...a method that uses only the pi orbitals of the interacting fragments in the geometry of the molecule for estimating pi interactions.[16]" For this type of analysis, the formation of bonds between various molecular moieties is a combination of three component terms. ΔEelstat represents what Fernández and Frenking call a molecule's “quasiclassical electrostatic attractions.[16]” The second term, ΔEPauli, represents the molecule's Pauli repulsion. ΔEorb, the third term, represents stabilizing interactions between orbitals, and is defined as the sum of ΔEpi and ΔEsigma. The total energy of interaction, ΔEint, is the result of the sum of the 3 terms.[16]
A group whose ΔEpi values were very thoroughly analyzed were a group of
Fernández and Frenking reported that the
Rotational barrier of ethane
An instance where hyperconjugation may be overlooked as a possible chemical explanation is in rationalizing the

In their 2001 paper, however, Pophristic and Goodman
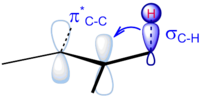
Goodman also conducted studies to determine the contribution of vicinal (between two methyl groups) vs. geminal (between the atoms in a single methyl group) interactions to hyperconjugation. In separate experiments, the geminal and vicinal interactions were removed, and the most stable conformer for each interaction was deduced.[8]
| Deleted interaction | Torsional angle | Corresponding conformer |
|---|---|---|
| None | 60° | Staggered |
| All hyperconjugation | 0° | Eclipsed |
| Vicinal hyperconjugation | 0° | Eclipsed |
| Geminal hyperconjugation | 60° | Staggered |
From these experiments, it can be concluded that hyperconjugative effects delocalize charge and stabilize the molecule. Further, it is the vicinal hyperconjugative effects that keep the molecule in the staggered conformation.[8] Thanks to this work, the following model of the stabilization of the staggered conformation of ethane is now more accepted:

Hyperconjugation can also explain several other phenomena whose explanations may also not be as intuitive as that for the rotational barrier of ethane.[18]
The matter of the rotational barrier of ethane is not settled within the scientific community. An analysis within quantitative molecular orbital theory shows that 2-orbital-4-electron (steric) repulsions are dominant over hyperconjugation.[19] A valence bond theory study also emphasizes the importance of steric effects.[20]
See also
References
- ISBN 0-534-07968-7
- S2CID 222197582.
- ^ The mixed orbital of antibonding character is, in fact, raised in energy compared to the original antibonding orbital. However, since the antibonding orbital remains unpopulated in most cases, this does not usually affect the energy of the system.
- ^ .
- ISBN 9789332901070.
- ^ S2CID 205017635.
- S2CID 9812878.
- .
- doi:10.1002/poc.3382.
- PMID 19562814.
- ^ .
- PMID 12841733.
- PMID 15547994.
- ^ PMID 16502455.
- ^ Refer to Reference 12 for the graph and its full analysis
- ^ PMID 12370897.
- PMID 14502731.
- PMID 15065281.