Hyperforin
 | |
 | |
| Clinical data | |
|---|---|
| Dependence liability | None |
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status | |
Hepatic and CYP3A & CYP2B | |
| Identifiers | |
| |
JSmol) | |
| Melting point | 79–80 °C (174–176 °F) |
| Solubility in water | 0.66 mg/mL (20 °C) |
| |
| |
| | |
Hyperforin is a phytochemical produced by some of the members of the plant genus Hypericum, notably Hypericum perforatum (St John's wort).[2] Hyperforin may be involved in the pharmacological effects of St. John's wort,[2] specifically in its antidepressant effects.[3][4][5]
Occurrence
Hyperforin has only been found in significant amounts in
Chemistry
Hyperforin is a
Hyperforin is unstable in the presence of light and oxygen.[14] Frequent oxidized forms contain a C3 to C9 hemiketal/heterocyclic bridge or will form furan/pyran derivatives.[7][8]
Pharmacokinetics
Some
In healthy male volunteers, 612 mg dry extract of St. John's wort produced hyperforin pharmacokinetics characterised by a half life of 19.64 hours.[16]
Pharmacodynamics
Hyperforin may be a constituent responsible for the
| Neurotransmitter | IC50 (nanomoles)[18] |
|---|---|
| Norepinephrine | 80 ± 24 |
| Dopamine | 102 ± 19 |
| GABA | 184 ± 41 |
| 5-HT | 205 ± 45 |
| Glutamate | 829 ± 687 |
| Choline | 8500 |
| Receptor | Ki (nanomoles)
|
|---|---|
D1 |
595.8[20] |
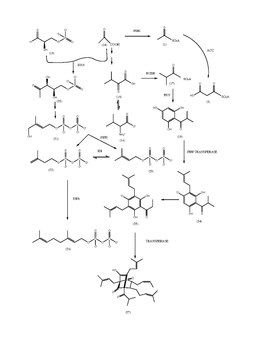
Hyperforin Biosynthesis
Hyperforin is a polyprenylated acylphloroglucinol (PPAP) derivative with a pholoroisobutyrophenone bicyclic core. Isobutryl-CoA (17) has been determined to be one of the initial primary metabolite starter molecules in the biosynthesis of the hyperforin core structure. Isobutryl-CoA is derived from an a-ketoisovalerate intermediate (15). The bicyclic structure suggests that it has elements of meroterpenoid origin. The nucleus of hyperforin is formed in a sequence condensation of one molecule of isobutyryl-CoA and three molecules of malonyl-CoA, both catalyzed by Isobutyrophenone synthase. Type III PKS enzymes will catalyze the decarboxylative condensation of enzyme active sites to generate scaffolding.
These enzymes preferred a different substrate and did not produce identical products. The cell-free extracts from the cell cultures were incubated with isobutyryl-CoA and malonyl-CoA, phlorisobutyrophenone was formed (18). The enzymatic reaction was identified as BUS. PIVP is a similar function of enzyme in glandular hairs of hop cones. Two acylphloroglucinoal cores PICP and PIBP formed are formed by claisen condensation but will differ in substrate and enzyme specificities. PIVP will use isovaleryl-CoA in the presence of an enzyme VSP, and PIBP will use isobutyryl-CoA in the presence of bus resulting in the production of adhyperforin and hyperforin.
However, hyperforin is an easily degradable compound highly sensitive to heat and light in its powder form or within a solution, making it difficult to determine a true synthesis route for hyperforin making this synthesis route a possible route.[21]
- Natural and semi-synthetic analogues of Hyperforin
-
Aristoforin
-
Hyperforin trimethoxybenzoate
-
Tetrahydrohyperforin
-
Hyperforin nicotinate
Antidepressant research
Two meta-analyses of preliminary clinical trials evaluating the efficacy of St. John's wort for treating mild-to-moderate depression indicated a response similar to selective serotonin reuptake inhibitors and with better tolerance, although the long-term generalization of study results was limited by the short duration (4–12 weeks) of reviewed studies.[22][23]
See also
References
- ^ St John's Wort available again Archived 2020-08-02 at the Wayback Machine. Irishhealth.com (2015-10-13). Retrieved on 2020-02-11.
- ^ a b c d "Hyperforin". PubChem, US National Library of Medicine. 8 September 2018. Retrieved 13 September 2018.
- ^ S2CID 4701643.
- ^ PMID 16967926.
- PMID 15464835.
- PMID 16973193.
- ^ PMID 12837358.
- ^ PMID 10346967.
- PMID 16599273.
- ^ PMID 23270309.
- PMID 1248360.
- .
- PMID 20063336.
- PMID 15708671.
- S2CID 24700980.
- S2CID 6880438.
- ISBN 978-0-85369-474-8.
- ^ PMID 9718074.
- PMID 10852961.
- ^ "Hyperforin". BindingDB. Retrieved 5 March 2015.
- PMID 33081197.
- PMID 28064110.
27 clinical trials with a total of 3808 patients were reviewed [...] For patients with mild-to-moderate depression, St John's wort has comparable efficacy and safety when compared to SSRIs. Follow-up studies carried out over a longer duration should be planned to ascertain its benefits.
- PMID 27468236.
A total of 3,126 patients with depression were included. St John's wort extract did not differ from SSRIs in clinical response, remission, and mean reduction in Hamilton Rating Scale for Depression score. [...] Both St John's wort extract and SSRIs are effective in treating mild-to-moderate depression. St John's wort extract is safer than SSRIs.
External links
 Media related to Hyperforin at Wikimedia Commons
Media related to Hyperforin at Wikimedia Commons





