Hypertrophic cardiomyopathy
| Hypertrophic cardiomyopathy | |
|---|---|
| Other names | Asymmetric septal hypertrophy; idiopathic hypertrophic subaortic stenosis; |
| Prognosis | Less than 1% per year risk of death (with treatment)[9] |
| Frequency | Up to 1 in 200 people[8] |
Hypertrophic cardiomyopathy (HCM, or HOCM when obstructive) is a condition in which muscle tissues of the heart become thickened without an obvious cause.[8] The parts of the heart most commonly affected are the interventricular septum and the ventricles.[10] This results in the heart being less able to pump blood effectively and also may cause electrical conduction problems.[3] Specifically, within the bundle branches that conduct impulses through the interventricular septum and into the Purkinje fibers, as these are responsible for the depolarization of contractile cells of both ventricles.[11]
People who have HCM may have a range of symptoms. People may be asymptomatic, or may have
HCM is most commonly
Treatment may depend on symptoms and other risk factors. Medications may include the use of
HCM affects up to one in 200 people.[8] People of all ages may be affected.[12] The first modern description of the disease was by Donald Teare in 1958.[13][14]
Signs and symptoms
Many people are asymptomatic or mildly symptomatic, and many of those carrying disease genes for HCM do not have clinically detectable disease.
Major risk factors for sudden death in individuals with HCM include prior history of cardiac arrest or ventricular fibrillation, spontaneous sustained ventricular tachycardia, abnormal exercise blood pressure and non-sustained ventricular tachycardia,[20][21] unexplained syncope, family history of premature sudden death, and LVW thickness greater than 15 mm to 30 mm, on echocardiogram.
HCM also presents with a
Pulsus bisferiens may occasional be found during examination.[23]
Genetics
| Gene | Locus | Type |
|---|---|---|
| MYH7 | 14q12 | CMH1 ( 192600) |
| TNNT2 | 1q32 | CMH2 ( 115195) |
| TPM1 | 15q22.1 | CMH3 ( 115196) |
MYBPC3
|
11p11.2 | CMH4 ( 115197) |
| ? | ? | CMH5 |
| PRKAG2 | 7q36 | CMH6 ( 600858) |
| TNNI3 | 19q13.4 | CMH7 ( 613690) |
| MYL3 | 3p | CMH8 ( 608751) |
| TTN | 2q24.3 | CMH9 ( 613765) |
| MYL2 | 12q23-q24 | CMH10 ( 608758) |
| ACTC1 | 15q14 | CMH11 ( 612098) |
| CSRP3 | 11p15.1 | CMH12 ( 612124) |
Familial hypertrophic cardiomyopathy is inherited as an
Currently, about 50–60% of people with a high index of clinical suspicion for HCM will have a mutation identified in at least one of nine sarcomeric genes. Approximately 40% of these mutations occur in the β-
Over 1400 mutations have been identified in genes known to lead to HCM.[28] Some mutations could have more harmful potential compared to others (β-myosin heavy chain). For example, troponin T mutations were originally associated with a 50% mortality before the age of 40. However, a more recent and larger study found a similar risk to other sarcomeric protein mutations.[29] The age at disease onset of HCM with MYH7 is earlier and leads to more severe symptoms.[30] Moreover, mutations on troponin C can alter Ca+2 sensibility on force development in cardiac muscle, these mutations are named after the amino acid that was changed after the location in which it happened, such as A8V, A31S, C84Y and D145E.[31]
Pathophysiology
Ventricular hypertrophy causes a dynamic pressure gradient across the left ventricular outflow tract (LVOT), which is associated with further narrowing of the outflow during systole. Pulling of the mitral valve leaflets towards the septum contributes to the outflow obstruction. This pulling is thought to occur by several proposed mechanisms, including that flow of blood through the narrowed outflow tract results in it having a higher velocity, and less pressure through the Venturi effect.[17] This low pressure then causes the anterior leaflet of the mitral valve to be pulled into the outflow tract, resulting in further obstruction.[32]
Diagnosis
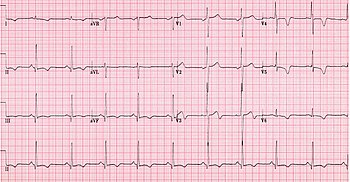
A
ECG is the most sensitive diagnostic test.
Variants
Depending on whether the distortion of normal heart anatomy causes an obstruction of the outflow of blood from the left ventricle of the heart, HCM can be classified as obstructive or non-obstructive.[35] The obstructive variant of HCM is hypertrophic obstructive cardiomyopathy (HOCM), also historically known as idiopathic hypertrophic subaortic stenosis (IHSS) or asymmetric septal hypertrophy (ASH).[36] The diagnosis of left ventricular outflow tract obstruction is usually made by echocardiographic assessment and is defined as a peak left ventricular outflow tract gradient of ≥ 30 mmHg.[37]
Another, non-obstructive variant of HCM is apical hypertrophic cardiomyopathy (AHCM or ApHCM),[38] also called Yamaguchi syndrome. It was first described in individuals of Japanese descent. Sakamoto is the first to report the condition's ECG pattern in 1976. Yamaguchi is the first to characterize the syndrome adn its ventriculargrophic feature in 1979.[39] Yamaguchi syndrome is an infrequent variant of HCM in the European population. ApHCM is thought to be autosomal dominant, with the majority of mutations occurring in the genes encoding for the sarcomere. In the thick myofilaments of the heart tissue, the predominant gene mutations occur in “myosin-binding protein C (MYBPC3)” and “myosin heavy chain (MYH7).” Myocardial infarction, atrial fibrillation, ventricular fibrillation, embolic events, and/or congestive heart failure are all possible outcomes of this condition. On ausculation, the presence of fourth heart sound(S4), deep negative T waves on ECG notably in the precordial leads, and a "spade-like" structure of the left ventricular chamber at end-diastole on the left ventriculography and also in 2D echo are all common signs of apical HCM. Sometimes, its ECG and clinical presentation may be misdiagnosed as an acute coronary syndrome. The apical HCM has lower mortality rate and better prognosis than other variant types of HCM.[40]
Cardiac catheterization
Upon
The Brockenbrough–Braunwald–Morrow sign is observed in individuals with HCM with outflow tract gradient. This sign can be used to differentiate HCM from aortic stenosis. In individuals with aortic stenosis, after a premature ventricular contraction (PVC), the following ventricular contraction will be more forceful, and the pressure generated in the left ventricle will be higher. Because of the fixed obstruction that the stenotic aortic valve represents, the post-PVC ascending aortic pressure will increase as well. In individuals with HCM, however, the degree of obstruction will increase more than the force of contraction will increase in the post-PVC beat. The result of this is that the left ventricular pressure increases and the ascending aortic pressure decreases, with an increase in the LVOT gradient.[citation needed]
While the Brockenbrough–Braunwald–Morrow sign is most dramatically demonstrated using simultaneous intra-cardiac and intra-aortic catheters, it can be seen on routine physical examination as a decrease in the pulse pressure in the post-PVC beat in individuals with HCM.[citation needed]
Screening
Although HCM may be asymptomatic, affected individuals may present with symptoms ranging from mild to critical heart failure and sudden cardiac death at any point from early childhood to seniority.[15][41] HCM is the leading cause of sudden cardiac death in young athletes in the United States, and the most common genetic cardiovascular disorder.[4] One study found that the incidence of sudden cardiac death in young competitive athletes declined in the Veneto region of Italy by 89% since the 1982 introduction of routine cardiac screening for athletes, from an unusually high starting rate.[42] As of 2010, however, studies have shown that the incidence of sudden cardiac death, among all people with HCM, has declined to one percent or less.[43] Screen-positive individuals who are diagnosed with cardiac disease are usually told to avoid competitive athletics.[44]
HCM can be detected with an
United States
There are several potential challenges associated with routine screening for HCM in the United States.[48] First, the U.S. athlete population of 15 million is almost twice as large as Italy's estimated athlete population.[48] Second, these events are rare, with fewer than 100 deaths in the U.S. due to HCM in competitive athletes per year,[49] or about 1 death per 220,000 athletes.[50] Lastly, genetic testing would provide a definitive diagnosis; however, due to the numerous HCM-causing mutations, this method of screening is complex and is not cost-effective.[15] Therefore, genetic testing in the United States is limited to individuals who exhibit clear symptoms of HCM, and their family members. This ensures that the test is not wasted on detecting other causes of ventricular hypertrophy (due to its low sensitivity), and that family members of the individual are educated on the potential risk of being carriers of the mutant gene(s).[51]
Canada
Canadian genetic testing guidelines and recommendations for individuals diagnosed with HCM are as follows:[33]
- The main purpose of genetic testing is for screening family members.
- According to the results, at-risk relatives may be encouraged to undergo extensive testing.
- Genetic testing is not meant for confirming a diagnosis.
- If the diagnosed individual has no relatives that are at risk, then genetic testing is not required.
- Genetic testing is not intended for risk assessment or treatment decisions.
- Evidence only supports clinical testing in predicting the progression and risk of developing complications of HCM.
For individuals suspected of having HCM:
- Genetic testing is not recommended for determining other causes of left ventricular hypertrophy (such as "athlete's heart", hypertension, and cardiac amyloidosis).
- HCM may be differentiated from other hypertrophy-causing conditions using clinical history and clinical testing.
Treatment
Asymptomatic people
A significant number of people with hypertrophic cardiomyopathy do not have any symptoms and will have a normal life expectancy, although they should avoid particularly strenuous activities or competitive athletics. Asymptomatic people should be screened for risk factors for sudden cardiac death. In people with resting or inducible outflow obstructions, situations that will cause dehydration or vasodilation (such as the use of vasodilatory or diuretic blood pressure medications) should be avoided. Septal reduction therapy is not recommended in asymptomatic people.[7]
Medications
The primary goal of medications is to relieve symptoms such as chest pain, shortness of breath, and palpitations.
Mavacamten was approved for medical use in the United States in April 2022.[52]
Surgical septal myectomy
Surgical
Alcohol septal ablation
Alcohol septal ablation, introduced by Ulrich Sigwart in 1994, is a percutaneous technique that involves injection of alcohol into one or more septal branches of the left anterior descending artery. This is a catheter technique with results similar to the surgical septal myectomy procedure but is less invasive, since it does not involve general anaesthesia and opening of the chest wall and pericardium (which are done in a septal myectomy). In a select population with symptoms secondary to a high outflow tract gradient, alcohol septal ablation can reduce the symptoms of HCM. In addition, older individuals and those with other medical problems, for whom surgical myectomy would pose increased procedural risk, would likely benefit from the less-invasive septal ablation procedure.[15][57]
When performed properly, an alcohol septal ablation induces a controlled heart attack, in which the portion of the interventricular septum that involves the left ventricular outflow tract is infarcted and will contract into a scar. There is debate over which people are best served by surgical myectomy, alcohol septal ablation, or medical therapy.[58]
Mitral clip
Since 2013,
Implantable pacemaker or defibrillator
The use of a
The procedure includes an incision on the anterolateral area below the clavicle. Two leads are then inserted; one into the right atrium and the other into the right ventricular apex via the subclavian veins. Once in place, they are secured and attached to the generator which will remain inside the fascia, anterior to the pectoral muscle.[41] Complications of this procedure include infection, electrical lead and generator malfunction which will require replacement.[41]
For people with HCM who exhibit one or more of the major risk factors for sudden cardiac death, an implantable cardioverter-defibrillator (ICD) or a combination pacemaker/ICD all-in-one unit may be recommended as an appropriate precaution.[7][20][62][63] In 2014, European Society of Cardiology suggested a practical risk score to calculate that risk.[64]
Cardiac transplantation
In cases that are unresponsive to all other forms of treatment,
Prognosis
A systematic review from 2002 concluded that: "Overall, HCM confers an annual mortality rate of about 1%... HCM may be associated with important symptoms and premature death but more frequently with no or relatively mild disability and normal life expectancy."[15]
Children
Even though hypertrophic cardiomyopathy (HCM) may be present early in life and is most likely congenital, it is one of the most-uncommon cardiac malformations encountered in pediatric cardiology, largely because the presentation of symptoms is usually absent, incomplete, or delayed into adulthood. Most of the current information pertaining to HCM arises from studies in adult populations, and the implication of these observations for pediatric population is often uncertain.[65] Nonetheless, recent studies in pediatric cardiology have revealed that HCM accounts for 42% of childhood cardiomyopathies, with an annual incidence rate of 0.47/100,000 in children.[66] Further, in asymptomatic cases, sudden death is considered one of the most-feared complications associated with the disease in select pediatric populations. Consequently, the recommended practice is to screen children of affected individuals throughout childhood to detect cardiac abnormalities at an early stage, in the hope of preventing further complications of the disease.[65]
Generally, the diagnosis of HCM in a pediatric population is made during assessment for murmur, congestive heart failure, physical exhaustion, and genetic testing of children of affected individuals.[65] Specifically, echocardiogram (ECHO) has been used as a definitive noninvasive diagnostic tool in nearly all children. ECHO assesses cardiac ventricular size, wall thickness, systolic and diastolic function, and outflow obstruction. Thus, ECHO has been chosen as an ideal means to detect excessive wall thickening of cardiac muscle in HCM.[65]
For children with HCM, treatment strategies aim to reduce disease symptoms and lower the risk of sudden death.[67] Due to the heterogeneity of the disease, treatment is usually modified according to individual's needs.[67] β-blockers improve left ventricular filling and relaxation and thereby lessen symptoms. In some children, β–blockers (e.g., propranolol) were shown effective to reduce the risk of sudden death.[67] Further, calcium channel blockers (verapamil) and antiarrhythmic drugs may be used as an adjunct therapy to β-blockers in symptomatic children. Nonetheless, further testing is needed to determine their definitive benefits.[67]
Epidemiology
The prevalence of HCM in the general population around the world is 0.2% (1 in 500 adults), as determined from echocardiographic studies.[17] HCM is more common in males than in females.[17] The most common presentation of HCM is in the third decade of life, though it can present at any age, from newborns to the elderly.[17]
Other animals


Cats
Feline hypertrophic cardiomyopathy (HCM) is the most common
Clinically, cats with hypertrophic cardiomyopathy commonly have a systolic anterior motion (SAM) of the mitral valve (see graphic).[78] Cats with severe HCM often develop left heart failure (pulmonary edema; pleural effusion) because of severe diastolic dysfunction of the left ventricle. They may also develop a left atrial thrombus that embolizes, most commonly, to the terminal aorta creating acute pain and rear limb paralysis (see below). Sudden death can also occur but appears to be uncommon.[79][80]
Ultrasound of the heart (echocardiography) is necessary to diagnose HCM in cats.[81][82][83] Measurement of circulating cardiac biomarkers, like N‐terminal‐proBNP (NT‐proBNP)[84][85] and troponin I (TnI) may be used in cats to strengthen the suspicion of cardiac disease.[86] There is a Point-of-care test for feline NT-proBNP available which can be used at the veterinary clinic when echocardiography is not possible to perform.[87][88][89]
Cats that are tachycardic (>220) and/or have outflow obstruction (SAM) on echo should probably be treated but there is no cure for feline HCM. Many but not all cats have a heart murmur. Many cats that have a heart murmur do not have HCM. Frequently the first signs that a cat has HCM are tachypnea/dyspnea due to heart failure or acute pain and paralysis due to systemic thromboembolism. While medication is commonly given to cats with HCM that have no clinical signs, no medication has been shown to be helpful at this stage and it has been shown that an ACE inhibitor is not beneficial until heart failure is present[90] (at which time a diuretic is most beneficial). Diltiazem generally produces no demonstrable benefit. Atenolol is commonly administered when a severe systolic anterior motion of the mitral valve is present.[citation needed]
Feline arterial thromboembolism (FATE) is a relatively common and devastating complication of feline HCM and other feline cardiomyopathies. The thrombus generally forms in the left atrium, most commonly the left auricle. The formation is thought to be primarily due to blood flow stasis. Classically, the thromboembolism lodges at the iliac trifurcation of the aorta, occluding either one or both of the common iliac arteries. Because this split is called the saddle, and is the most frequent location for the thrombus, FATE is commonly known as saddle thrombus.[91] Clinically this presents as a cat with complete loss of function in one or both hind limbs. The hind limbs are cold and the cat is in considerable pain. Emboli may, rarely, lodge in other locations, most commonly the right front limb and the renal arteries.[citation needed]
Clopidogrel is used to try to prevent left atrial thrombus formation in cats with HCM and a large left atrium. The FATCAT study at Purdue University demonstrated that it is superior to aspirin for the prevention of a second thrombus from forming in cats that have already experienced a clot. Thrombolytic agents (e.g., tissue plasminogen activator) have been used with some success to break down an existing aortic thromboembolism, but their cost is high and outcome appears to be no better than giving a cat time (48–72 hours) to break down its own clot. Pain management is extremely important. The prognosis for cats with FATE is often poor as they are likely to have significant HCM already and a recurrent bout of FATE is likely.[92] For this reason, euthanasia is often a valid consideration.[citation needed]
Gorillas
In July 2013, Rigo, a 42-year-old western lowland gorilla, resident in Melbourne Zoo and father of Mzuri, the first gorilla born by artificial insemination, died unexpectedly as a result of HCM. The condition is not uncommon in male gorillas over the age of 30, and in many cases, there is no sign of the disease until the individual's sudden death.[93]
References
- ^ "Other Names for Cardiomyopathy". NHLBI. June 22, 2016. Archived from the original on 28 July 2016. Retrieved 31 August 2016.
- ^ a b c "What Are the Signs and Symptoms of Cardiomyopathy?". NHLBI. 22 June 2016. Archived from the original on 28 July 2016. Retrieved 10 November 2017.
- ^ a b c "What Is Cardiomyopathy?". NHLBI. 22 June 2016. Archived from the original on 10 November 2017. Retrieved 10 November 2017.
- ^ S2CID 25887172.
- ^ ISBN 9780323529570. Archivedfrom the original on 2017-11-10. Retrieved 2017-11-10.
- ^ a b c "What Causes Cardiomyopathy?". NHLBI. 22 June 2016. Archived from the original on 5 October 2017. Retrieved 10 November 2017.
- ^ PMID 22093712.
- ^ ISBN 978-3-030-24176-6. Archivedfrom the original on 2023-01-10. Retrieved 2022-10-20.
- ^ PMID 24998133.
- ^ from the original on 2021-04-06. Retrieved 2020-09-20.
- PMID 37125268.
- ^ "Types of Cardiomyopathy". NHLBI. 22 June 2016. Archived from the original on 4 October 2017. Retrieved 10 November 2017.
- PMID 13499764.
- from the original on 2017-11-11. Retrieved 2017-02-06.
- ^ S2CID 19887576.
- PMID 18212300.
- ^ PMID 28613539. Retrieved 2023-10-25.
- ISBN 978-1-4160-0014-3.
- PMID 7671349.
- ^ PMID 14607462.
- S2CID 1726747.
- ISBN 9781439825457. Archivedfrom the original on 2018-10-23. Retrieved 2018-10-22.
- ISBN 9781482215649. Archivedfrom the original on 2018-01-03. Retrieved 2018-01-02.
- from the original on 2023-08-09. Retrieved 2023-04-02.
- from the original on 2017-01-18. Retrieved 2017-02-11.
- PMID 15314809.
- S2CID 39088276.
- S2CID 38333896.
- PMID 22144547.
- S2CID 23723131.
- PMID 26851561.
- ^ "Venturi effect". www.healio.com. Retrieved 2023-10-25.
- ^ PMID 21459272.
- PMID 29343659.
- S2CID 227078319.
- ISBN 978-1-4160-6391-9. Archivedfrom the original on 2023-08-09. Retrieved 2022-12-02.
- S2CID 227078319.
- PMID 8685759.
- ^ Yamaguchi H, Ishimura T, Nishiyama S, Nagasaki F, Nakanishi S, Takatsu F, Nishijo T, Umeda T, Machii K. Hypertrophic nonobstructive cardiomyopathy with giant negative T waves (apical hypertrophy): ventriculographic and echocardiographic features in 30 patients. Am J Cardiol. 1979 Sep;44(3):401-12. doi: 10.1016/0002-9149(79)90388-6. PMID: 573056.
- license.
- ^ S2CID 8041261.
- PMID 17018804.
- PMID 28770721.
- PMID 28795011.
- PMID 27519172.
- ^ PMID 17174192.
- ^ S2CID 318803.
- ^ S2CID 37588552.
- PMID 19221222.
- PMID 9857867.
- PMID 19254666.
- ^ "U.S. Food and Drug Administration Approves Camzyos (mavacamten) for the Treatment of Adults With Symptomatic New York Heart Association Class II-III Obstructive Hypertrophic Cardiomyopathy (HCM) to Improve Functional Capacity and Symptoms" (Press release). Bristol Myers Squibb. 28 April 2022. Retrieved 29 April 2022 – via Business Wire.
- PMID 12607696.
- PMID 8067875.
- PMID 7586394.
- ^ PMID 15975370.
- S2CID 32959772.
- PMID 17239718.
- PMID 27247935.
- PMID 27311518.
- PMID 10400010.
- PMID 17652294.
- ^ "ICDs and Pacemakers". Hypertrophic Cardiomyopathy Association. Archived from the original on November 15, 2016. Retrieved November 14, 2016.
- ^ "2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy". Archived from the original on 2020-12-05. Retrieved 2020-11-22.
- ^ PMID 20869644.
- PMID 12711739.
- ^ PMID 22222117.
- (PDF) from the original on 2022-01-24. Retrieved 2020-09-28.
- from the original on 2023-08-09. Retrieved 2020-09-14.
- from the original on 2023-08-09. Retrieved 2023-01-24.
- ^ "Hypertrophic Cardiomyopathy (HCM) in Cats". Cornell University Hospital for Animals. Archived from the original on 22 January 2018. Retrieved 24 February 2017.
- PMID 10377082.
- ^ Kittleson M, Gompf R, Little S. "Feline Hypertrophic Cardiomyopathy: Advice for Breeders". Cat Fancier's Association. Archived from the original on May 13, 2008.
- PMID 16236761.
- ^ "Genetics: Maine Coon Cat Hypertrophic Cardiomyopathy". North Carolina State University, College of Veterinary Medicine. Archived from the original on September 3, 2019. Retrieved December 29, 2016.
- PMID 17521870.
- ^ "Genetics: Ragdoll Cat Hypertrophic Cardiomyopathy". North Carolina State University, College of Veterinary Medicine. Archived from the original on December 14, 2016. Retrieved December 29, 2016.
- PMID 20185379.
- PMID 29660848.
- PMID 31605422.
- (PDF) from the original on 2021-01-22. Retrieved 2020-09-28.
- PMID 26776573.
- PMID 32243654.
- PMID 21985136.
- PMID 21434959.
- (PDF) from the original on 2020-03-07. Retrieved 2020-09-28.
- PMID 32200578.
- PMID 25456274.
- PMID 28617995.
- PMID 17063701.
- ^ "The Fragile Fate of FATEs: The Management and Prognosis of Feline Aortic Thromboembolism". Massachusetts Society for the Prevention of Cruelty to Animals-Angell. Archived from the original on October 9, 2018. Retrieved October 8, 2016.
- PMID 24237457.
- ^ Smith B (2013-07-26). "Silverback gorilla Rigo died of heart failure at Melbourne Zoo". The Age. Archived from the original on 2017-01-03. Retrieved 2013-07-26.
