Idiopathic pulmonary fibrosis
| Idiopathic pulmonary fibrosis | |
|---|---|
| Other names | Fibrosing alveolitis, cryptogenic fibrosing alveolitis, diffuse fibrosing alveolitis, usual interstitial pneumonitis, diffuse interstitial pneumonitis |
viral infections, family history[1] | |
| Diagnostic method | CT scan, lung biopsy[3] |
| Differential diagnosis | Sarcoidosis, other interstitial lung diseases, hypersensitivity pneumonitis[4] |
| Treatment | Pulmonary rehabilitation, supplemental oxygen, lung transplantation[1] |
| Medication | Pirfenidone, nintedanib[2] |
| Prognosis | Life expectancy ~ 4 years[1] |
| Frequency | 12 per 100,000 people per year[4] |
Idiopathic pulmonary fibrosis (IPF), or (formerly
The cause is unknown, hence the term
People often benefit from pulmonary rehabilitation and supplemental oxygen.[1] Certain medications like pirfenidone (Esbriet) or nintedanib (Ofev) may slow the progression of the disease.[2] Lung transplantation may also be an option.[1]
About 5 million people are affected globally.[7] The disease newly occurs in about 12 per 100,000 people per year.[4] Those in their 60s and 70s are most commonly affected.[4] Males are affected more often than females.[4] Average life expectancy following diagnosis is about four years.[1] Updated international guidelines were published in 2022, which some simplification in diagnosis and the removal of antacids as a possible adjunct therapy.[8]
Signs and symptoms

In many people, symptoms are present for a considerable time before diagnosis.[7] The most common clinical features of IPF include the following:[3][9][10]
- Age over 50 years
- Dry, non-productive cough on exertion
- Progressive exertional dyspnea (shortness of breath with exercise)
- Dry, inspiratory bibasilar "velcro-like" crackles on auscultation (a crackling sound in the lungs during inhalation similar to Velcro being torn apart slowly, heard with a stethoscope).[3][11][12]
- Clubbing of the digits, a disfigurement of the finger tips or toes (see image)
- Abnormal pulmonary function testresults, with evidence of restriction and impaired gas exchange.
Some of these features are due to chronic hypoxemia (oxygen deficiency in the blood), are not specific for IPF, and can occur in other pulmonary disorders. IPF should be considered in all patients with unexplained chronic exertional dyspnea who present with cough, inspiratory bibasilar crackles, or finger clubbing.[3]
Assessment of "velcro" crackles on lung auscultation is a practical way to improve the earlier diagnosis of IPF. Fine crackles are easily recognized by clinicians and are characteristic of IPF.[13]
If bilateral fine crackles are present throughout the inspiratory time and are persisting after several deep breaths, and if remaining present on several occasions several weeks apart in a subject aged ≥60 years, this should raise the suspicion of IPF and lead to consideration of an HRCT scan of the chest which is more sensitive than a
Causes
The cause of IPF is unknown but certain environmental factors and exposures have been shown to increase the risk of getting IPF.
Pathogenesis

Despite extensive investigation, the cause of IPF remains unknown.[3] The fibrosis in IPF has been linked to cigarette smoking, environmental factors (e.g. occupational exposure to gases, smoke, chemicals or dusts), other medical conditions including gastroesophageal reflux disease (GERD), or to genetic predisposition (familial IPF). However, none of these is present in all people with IPF and therefore do not provide a completely satisfactory explanation for the disease.[3][16]
IPF is believed to be the result of an aberrant wound healing process including/involving abnormal and excessive deposition of
It is hypothesized that the initial or repetitive injury in IPF occurs to the lung cells, called alveolar epithelial cells (AECs, pneumocytes), which line the majority of the alveolar surface.
In the past, it was thought that
Familial IPF accounts for less than 5% of the total of patients with IPF and is clinically and histologically indistinguishable from sporadic IPF.
Diagnosis
An earlier diagnosis of IPF is a prerequisite for earlier treatment and, potentially, improvement of the long-term clinical outcome of this progressive and ultimately fatal disease.[3] If IPF is suspected, diagnosis can be challenging but a multidisciplinary approach involving a pulmonologist, radiologist and pathologist expert in interstitial lung disease has been shown to improve the accuracy of IPF diagnosis.[3][29][30]
A Multidisciplinary Consensus Statement on the Idiopathic Interstitial Pneumonias published by the American Thoracic Society (ATS) and the European Respiratory Society (ERS) in 2000 proposed specific major and minor criteria for establishing the diagnosis of IPF.[3] However, in 2011, new simplified and updated criteria for the diagnosis and management of IPF were published by the ATS, ERS, together with the Japanese Respiratory Society (JRS) and Latin American Thoracic Association (ALAT).[3] Currently, a diagnosis of IPF requires:
- Exclusion of known causes of ILD, e.g., domestic and occupational environmental exposures, connective tissue disorders, or drug exposure/toxicity
- The presence of a typical radiological pattern of usual interstitial pneumonia (UIP) on high-resolution computed tomography (HRCT).
In the right clinical setting, it is possible to make the diagnosis of IPF by HRCT alone, obviating the need for surgical lung biopsy.[3][9]
Various technologies using
Differential diagnosis
Recognizing IPF in clinical practice can be challenging as
Classification

Idiopathic pulmonary fibrosis (IPF) belongs to a large group of more than 200 lung diseases known as
The 2002
The diagnosis of IIPs requires exclusion of known causes of ILD. Examples of ILD of known cause include
Radiology
Typical HRCT of the chest of IPF demonstrates fibrotic changes in both lungs, with a predilection for the bases and the periphery. According to the joint ATS/ERS/JRS/ALAT 2011 guidelines, HRCT is an essential component of the diagnostic pathway in IPF which can identify UIP by the presence of:[3]
- Reticular opacities, often associated with traction bronchiectasis
- Honeycombing manifested as cluster cystic airspaces, typically of comparable diameters (3–10 mm) but occasionally large. Usually sub-pleural and characterized by well-defined walls and disposed in at least two lines. Generally one line of cysts is not sufficient to define honeycombing
- Ground-glass opacitiesare common but less extensive than the reticulation
- Distribution characteristically basal and peripheral though often patchy.

Histology
According to the updated 2011 guidelines, in the absence of a typical UIP pattern on HRCT, a surgical lung biopsy is required for confident diagnosis.[3]
Histologic specimens for the diagnosis of IPF must be taken at least in three different places and be large enough that the pathologist can comment on the underlying lung architecture. Small biopsies, such as those obtained via transbronchial lung biopsy (performed during bronchoscopy) are usually not sufficient for this purpose. Hence, larger biopsies obtained surgically via a thoracotomy or thoracoscopy are usually necessary.[3][9]
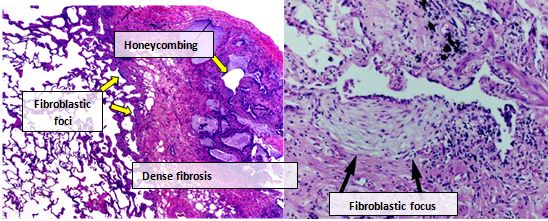
Lung tissue from people with IPF usually show a characteristic histopathologic UIP pattern and is therefore the pathologic counterpart of IPF.[3] Although a pathologic diagnosis of UIP often corresponds to a clinical diagnosis of IPF, a UIP histologic pattern can be seen in other diseases as well, and fibrosis of known origin (rheumatic diseases for example).[1][3] There are four key features of UIP including interstitial fibrosis in a 'patchwork pattern', interstitial scarring, honeycomb changes and fibroblast foci.[citation needed]
Fibroblastic foci are dense collections of myofibroblasts and scar tissue and, together with honeycombing, are the main pathological findings that allow a diagnosis of UIP.
Bronchoalveolar lavage
Pulmonary function tests
Spirometry classically reveals a reduction in the vital capacity (VC) with either a proportionate reduction in airflows, or increased airflows for the observed vital capacity. The latter finding reflects the increased lung stiffness (reduced lung compliance) associated with pulmonary fibrosis, which leads to increased lung elastic recoil.[34]
Measurement of static lung volumes using
The diffusing capacity for carbon monoxide (DLCO) is invariably reduced in IPF and may be the only abnormality in mild or early disease. Its impairment underlies the propensity of patients with IPF to exhibit oxygen desaturation with exercise which can also be evaluated using the 6-minute walk test (6MWT).[3]
Terms such as 'mild', 'moderate', and 'severe' are sometimes used for staging disease and are commonly based on resting pulmonary function test measurements.[3] However, there is no clear consensus regarding the staging of IPF patients and what are the best criteria and values to use. Mild-to-moderate IPF has been characterized by the following functional criteria:[35][36][37][38]
- Forced vital capacity(FVC) of ≥50%
- DLCO of ≥30%
- 6MWT distance ≥150 meters.
Treatment
The goals of treatment in IPF are essentially to reduce the symptoms, stop disease progression, prevent acute exacerbations, and prolong survival. Preventive care (e.g. vaccinations) and symptom-based treatment should be started early in every patient.[39]
Oxygen therapy
In the 2011 IPF guidelines, oxygen therapy, or supplementary oxygen for home use, became a strong recommendation for use in those patients with significantly low oxygen levels at rest. Although oxygen therapy has not been shown to improve survival in IPF, some data indicate an improvement in exercise capacity.[3][40]
Pulmonary rehabilitation
Fatigue and loss of muscular mass are common and disabling problems for patients with IPF. Pulmonary rehabilitation may alleviate the overt symptoms of IPF and improve functional status by stabilizing and/or reversing the extrapulmonary features of the disease.[41][42] The number of published studies on the role of pulmonary rehabilitation in idiopathic pulmonary fibrosis is small, but most of these studies have found significant short-term improvements in functional exercise tolerance, quality of life, and dyspnea on exertion.[43] Typical programs of rehabilitation include exercise training, nutritional modulation, occupational therapy, education and psychosocial counseling. In the late phase of disease, IPF patients tend to discontinue physical activity due to increasing dyspnea. Whenever possible, this should be discouraged.[citation needed]
Medications
A number of treatments have been investigated in the past for IPF, including
Pirfenidone
A
N-acetylcysteine and triple therapy
More recently, a large randomized, controlled trial (PANTHER-IPF) was undertaken by the National Institutes of Health (NIH) in the US to evaluate triple therapy and NAC monotherapy in IPF patients. This study found that the combination of prednisone, azathioprine, and NAC increased the risk of death and hospitalizations[50] and the NIH announced in 2012 that the triple-therapy arm of the PANTHER-IPF study had been terminated early.[51]
This study also evaluated NAC alone and the results for this arm of the study were published in May 2014 in the
Nintedanib
Lung transplantation
Lung transplantation may be suitable for those patients physically eligible to undergo a major transplant operation. In IPF patients, lung transplant has been shown to reduce the risk of death by 75% as compared with patients who remain on the waiting list.[56] Since the introduction of the lung allocation score (LAS), which prioritizes transplant candidates based on survival probability, IPF has become the most common indication for lung transplantation in the USA.[41]
Symptomatic patients with IPF younger than 65 years of age and with a body mass index (BMI) ≤26 kg/m2 should be referred for lung transplantation, but there are no clear data to guide the precise timing for LTx. Although controversial, the most recent data suggest that bilateral lung transplantation is superior to single lung transplantation in patients with IPF.[57] Five-year survival rates after lung transplantation in IPF are estimated at between 50 and 56%.[3][58][59]
Palliative care
Palliative care also includes relief of physical and emotional suffering and psychosocial support for patients and caregivers.[3] With disease progression, patients may experience fear, anxiety and depression and psychological counseling should therefore be considered. In a recent study of outpatients with ILDs, including IPF, depression score, functional status (as assessed by walk test), as well as pulmonary function, all contributed to the severity of dyspnea.[60]
In selected cases of particularly severe dyspnea morphine could be considered. It can reduce dyspnea, anxiety and cough without significant decrease in oxygen saturation.[61]
Follow-up
IPF is often misdiagnosed, at least until physiological and/or imaging data suggest the presence of an ILD leading to delay in accessing appropriate care.[41] Considering that IPF is a disease with a median survival of three years after diagnosis, early referral to a center with specific expertise should therefore be considered for any patient with suspected or known ILD. On the basis of the complex differential diagnostic, multidisciplinary discussion between pulmonologists, radiologists, and pathologists experienced in the diagnosis of ILD is of the utmost importance to an accurate diagnosis.[3]
Those with IPF have higher chances of getting lung cancer, at a rate of 13.5% where the most common cancer type is Squamous-cell carcinoma of the lung.[62] a routine evaluation every 3 to 6 months, including spirometry (body plethysmography), diffusion capacity testing, chest X-rays, 6MWT, assessment of dyspnea, quality of life, oxygen requirement is mandatory.[citation needed]
In addition, the increasing awareness of complications and common concomitant conditions frequently associated with IPF requires a routinely evaluation of comorbidities, most of them simply reflecting concurrent diseases of aging, and medications with their interaction and side effects.
Acute exacerbations
Acute exacerbations of IPF (AE-IPF) are defined as an unexplained worsening or development of dyspnea within 30 days with new radiological infiltrates at HRCT abnormality often superimposed on a background consistent with UIP pattern. The yearly incidence of AE-IPF is between 10 and 15% of all patients. The prognosis of AE-IPF is poor, with mortality ranging from 78% to 96%.[63] Other causes of AE-IPF such as pulmonary embolism, congestive heart failure, pneumothorax, or infection need to be excluded. Pulmonary infection have to be ruled out by endotracheal aspirate or BAL.
Many patients experiencing acute deterioration require intensive care treatment, particularly when respiratory failure is associated with hemodynamic instability, significant comorbidities or severe hypoxemia.[64] However, mortality during hospitalization is high.[63] Mechanical ventilation should be introduced only after carefully weighing the person's long-term prognosis and, whenever possible, the person's wishes. However, current guidelines discourage the use of mechanical ventilation in patients with respiratory failure secondary to IPF.[3]
Prognosis

The clinical course of IPF can be unpredictable.[3][65][66] IPF progression is associated with an estimated median survival time of 2 to 5 years following diagnosis.[1][3] The 5-year survival for IPF ranges between 20 and 40%,[66] a mortality rate higher than that of a number of malignancies, including colon cancer, multiple myeloma and bladder cancer.[65][66]
Recently a multidimensional index and staging system has been proposed to predict mortality in IPF.[67] The name of the index is GAP and is based on gender [G], age [A], and two lung physiology variables [P] (FVC and DLCO) that are commonly measured in clinical practice to predict mortality in IPF. The highest stage of GAP (stage III) has been found to be associated with a 39% risk of mortality at 1 year.[67] This model has also been evaluated in IPF and other ILDs and shown good performance in predicting mortality in all main ILD subtypes. A modified ILD-GAP Index has been developed for application across ILD subtypes to provide disease-specific survival estimates.[68] In IPF patients, the overall mortality at 5 years rate is high but the annual rate of all-cause mortality in patients with mild to moderate lung impairment is relatively low. This is the reason why change in lung function (FVC) is usually measured in 1-year clinical trials of IPF treatments rather than survival.[69]
In addition to clinical and physiological parameters to predict how rapidly patients with IPF might progress, genetic and molecular features are also associated with IPF mortality. For example, it has been shown that IPF patients who have a specific genotype in the mucin MUC5B gene polymorphism (see above) experience slower decline in FVC and significantly improved survival.[70][71] Even if such data are interesting from a scientific point of view, the application in the clinical routine of a prognostic model based on specific genotypes is still not possible.
Epidemiology
Although rare, IPF is the most common form of IIP.[9] The prevalence of IPF has been estimated between 14.0 and 42.7 per 100,000 persons based on a USA analysis of healthcare claims data, with variation depending on the case definitions used in this analyses.[11][72] IPF is more common in men than in women and is usually diagnosed in people over 50 years of age.[3]
The incidence of IPF is difficult to determine as uniform diagnostic criteria have not been applied consistently.[3][11] A recent study from the US estimated the incidence of IPF to be between 6.8 and 16.3 per 100,000 persons. In the 27 European Union countries, a range of sources estimate an incidence of 4.6–7.4 people per 100,000 of the population,[73][74] suggesting that approximately 30,000–35,000 new patients will be diagnosed with IPF each year.[72][75]
A recent single-centre, retrospective, observational cohort study including incident patients diagnosed with ILD at Aarhus University Hospital (Denmark) between 2003 and 2009 revealed an incidence of 4.1 per 100,000 inhabitants/year for ILD. IPF was the most common diagnosis (28%) followed by connective tissue disease-related ILD (14%), hypersensitivity pneumonitis (7%) and non-specific interstitial pneumonia (NSIP) (7%). IPF incidence was 1.3 per 100,000 inhabitants/year.[76]
Due to a heterogeneous distribution of the disease across European countries, epidemiological data needs to be updated through a Europe-wide registry for ILD and IPF.
Other animals
IPF has been recognized in several breeds of both dogs and cats, Prognosis is generally poor.
Research
A number of agents are currently being investigated in
mir-29 microRNA precursor investigations in mice have produced reversal of induced IPF. MRG-201 is currently being tested as-of 2016, but not in IPF patients yet, and no human trials for IPF use have been scheduled as of January 2016[update].[81]
A machine learning algorithm has been proposed that discovers subtle patterns in individual history of medical encounters to reliably estimate the risk of a future IPF diagnosis, up to four years before current medical practice.[84] The algorithm outputs a score (ZCoR) using medical history on file with no new tests, and might be deployable as a universal IPF screening tool in primary care. ZCoR has been trained and validated on nearly 3 million patients across multiple databases, achieving high predictive performance in out-of-sample data (positive likelihood ratio > 30 with 99% specificity). The authors conclude that past respiratory disorders maximally contribute to IPF risk, followed by known IPF comorbidities, metabolic diseases, cardiovascular abnormalities, and diseases of the eye, with the overall pattern of the importance ranking substantially invariant across the sexes.
References
- ^ a b c d e f g h i j k l m n o p q "Idiopathic Pulmonary Fibrosis". NHLBI. Retrieved 21 January 2018.
- ^ PMID 26177183.
- ^ PMID 21471066.
- ^ ISBN 9780323529570.
- PMID 22824096.
- ^ "Idiopathic Pulmonary Fibrosis". NHLBI, NIH. Retrieved 5 December 2020.
- ^ PMID 18366757.
- S2CID 248432749.
- ^ PMID 24032382.
- ^ PMID 12773329.
- ^ PMID 16809633.
- ^ PMID 22941541.
- PMID 2060395.
- ^ PMID 22365244.
- S2CID 22704874.
- ]
- ^ S2CID 21633787.
- PMID 30616998.
- PMID 31451866.
- PMID 32686219.
- ^ PMID 23602652.
- S2CID 52130702.
- ^ S2CID 17026757.
- PMID 12379275.
- S2CID 10955241.
- ^ a b "OMIM Entry – # 178500 – PULMONARY FIBROSIS, IDIOPATHIC; IPF". Omim.org. Retrieved 7 June 2018.
- PMID 25022318.
- PMID 24504062.
- PMID 15256390.
- PMID 17255566.
- ^ Lynch DA, Sverzellati N, Travis WD, Brown KK, Colby TV, Galvin JR, et al. Diagnostic criteria for idiopathic pulmonary fibrosis: a Fleischner Society White Paper. Lancet Respir Med. 2018;6:138–53
- ^ Taiki Furukawa et al. A comprehensible machine learning tool to differentially diagnose idiopathic pulmonary fibrosis from other chronic interstitial lung diseases. Respirology, 27(9) pp.739-746. First published: 13 June 2022 https://doi.org/10.1111/resp.14310
- PMID 19246718.
- PMID 16264058.
- ^ S2CID 10119356.
- S2CID 24224976.
- ^ PMID 24836312.
- ^ PMID 24836310.
- S2CID 11918582.
- PMID 1643945.
- ^ PMID 23146172.
- S2CID 11918582.
- PMID 23942353.
- S2CID 2432490.
- S2CID 3190634.
- S2CID 115745592.
- PMID 22561965.
- PMID 20824834.
- S2CID 30279834.
- PMID 22607134.
- ^ "Commonly used three-drug regimen for idiopathic pulmonary fibrosis found harmful". NIH. 21 October 2011. Retrieved 11 April 2013.
- PMID 24836309.
- ^ "BIBF 1120 Fact Sheet" (PDF). Dl.groovygecko.net. Archived from the original (PDF) on 3 March 2016. Retrieved 8 April 2014.
- ^ "FDA Approval Package for Nintedanib" (PDF). www.accessdata.fda.gov. Retrieved 7 January 2019.
- ^ "Ofev | European Medicines Agency". www.ema.europa.eu. 17 September 2018. Retrieved 7 January 2019.
- PMID 19820072.
- PMID 17888957.
- PMID 22006881.
- PMID 19249537.
- S2CID 34116718. Archived from the original(PDF) on 1 August 2020.
- S2CID 12999693.
- PMID 30114238.
- ^ PMID 18471669.
- PMID 11451841.
- ^ S2CID 13942321. Archived from the original(PDF) on 27 February 2019.
- ^ PMID 16738191.
- ^ S2CID 207536377.
- PMID 24114524.
- PMID 24476390.
- PMID 23695349.
- PMID 23321605.
- ^ a b Pulmonary Fibrosis Foundation. "Prevalence and Incidence". Pulmonaryfibrosis.org. Retrieved 11 April 2013
- PMID 16844727.
- ^ "Eurostat News Release. European demography. 110/2010. 27 July 2010" (PDF). Epp.eurostat.ec.europa.eu. Retrieved 7 June 2018.
- PMID 24636811.
- PMID 15189952.
- PMID 12240528.
- ^ "AKC Canine Health Foundation". Akcchf.org. Retrieved 7 June 2018.
- ^ "Active Clinical Trials and Investigational Research in IPF". Archived from the original on 4 September 2014. Retrieved 4 September 2014.
- ^ "Research Demonstrates Reversal Of Pulmonary Fibrosis With miRagen Therapeutics Synthetic microRNA-29 Mimic (promiR-29)". Pulmonaryfibrosisnews.com. 23 September 2014. Retrieved 8 June 2018.
- PMID 25986617.
- ^ "Stem cell therapy for lung fibrosis conditions". Sciencedaily.com. Retrieved 8 June 2018.
- S2CID 252622515.
External links
- Idiopathic pulmonary fibrosis at Curlie

