Henoch–Schönlein purpura
| Henoch–Schönlein purpura | |
|---|---|
| Other names | IgA vasculitis,[1] anaphylactoid purpura,[2] purpura rheumatica,[2] Schönlein–Henoch purpura[2] |
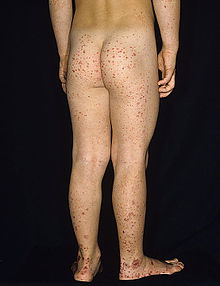 | |
| Typical purpura on lower legs and buttocks | |
| Pronunciation | |
| Specialty | Rheumatology, Immunology |
Henoch–Schönlein purpura (HSP), also known as IgA vasculitis, is a disease of the
HSP is a systemic vasculitis (inflammation of blood vessels) and is characterized by deposition of immune complexes containing the antibody immunoglobulin A (IgA); the exact cause for this phenomenon is unknown. In children, it usually resolves within several weeks and requires no treatment apart from symptom control but may relapse in a third of cases and cause irreversible kidney damage in about one in a hundred cases. In adults, the prognosis is different from in children. The average duration of cutaneous lesions is 27.9 months.[3] For many, it tends to be relapsing–remitting over a long period of time, rather than self-limiting and there tend to be more complications.[4]
Signs and symptoms


Purpura, arthritis, and abdominal pain are known as the "classic triad" of Henoch–Schönlein purpura.[5] Purpura occur in all cases, joint pains and arthritis in 80%, and abdominal pain in 62%. Some include gastrointestinal hemorrhage as a fourth criterion; this occurs in 33% of cases, sometimes, but not necessarily always, due to intussusception.[6] The purpura typically appear on the legs and buttocks, but may also be seen on the arms, face and trunk. The abdominal pain is colicky in character, and may be accompanied by nausea, vomiting, constipation or diarrhea. There may be blood or mucus in the stools.[7] The joints involved tend to be the ankles, knees, and elbows, but arthritis in the hands and feet is possible; the arthritis is nonerosive and hence causes no permanent deformity.[5] Forty percent have evidence of kidney involvement, mainly in the form of hematuria (blood in the urine), but only a quarter will have this in sufficient quantities to be noticeable without laboratory tests.[6] Problems in other organs, such as the central nervous system (brain and spinal cord) and lungs may occur, but is much less common than in the skin, bowel and kidneys.[8]
Of the 40% of patients who develop kidney involvement, almost all have evidence (visible or on urinalysis) of blood in the urine. More than half also have proteinuria (protein in the urine), which in one eighth is severe enough to cause nephrotic syndrome (generalised swelling due to low protein content of the blood). While abnormalities on urinalysis may continue for a long time, only 1% of all HSP patients develop chronic kidney disease.[8] Hypertension (high blood pressure) may occur. Protein loss and high blood pressure, as well as the features on biopsy of the kidney if performed, may predict progression to advanced kidney disease. Adults are more likely than children to develop advanced kidney disease.[8][9]
Pathophysiology
Henoch–Schönlein purpura is a small-vessel vasculitis in which complexes of immunoglobulin A (IgA) and complement component 3 (C3) are deposited on arterioles, capillaries, and venules (hence it is a type III hypersensitivity reaction). The activation of the alternative complement pathway results in the deposition of IgA aggregates or IgA complexes in target organs (with deposition of C3).[10] This leads to the production of inflammatory mediators, including vascular prostaglandins like prostacyclin, which may play a key role in the development of IgAV and its organ-specific clinical manifestations.[11] As with IgA nephropathy, serum levels of IgA are high in HSP and there are identical findings on renal biopsy; however, IgA nephropathy has a predilection for young adults while HSP is more predominant among children. Further, IgA nephropathy typically only affects the kidneys while HSP is a systemic disease. HSP involves the skin and connective tissues, scrotum, joints, gastrointestinal tract and kidneys.[12] The genetic basis remains unclear except for involvement of the human leukocyte antigen region of the genome.[13] It is hypothesized to involve autoimmunity triggered by infections. Streptococcus strains and Parainfluenza virus are the most commonly associated pathogens, and in children Human Parvovirus B19 is a frequent viral trigger[14]
Diagnosis

The diagnosis is based on the combination of the symptoms, as very few other diseases cause the same symptoms together.
If there is doubt about the cause of the skin lesions, a
On the basis of symptoms, it is possible to distinguish HSP from
Biopsy of the
HSP can develop after infections with
The exact cause of HSP is unknown, but most of its features are due to the deposition of abnormal antibodies in the wall of blood vessels, leading to
Classification
Multiple standards exist for defining Henoch–Schönlein purpura, including the 1990
More recent classifications, the 2006
Differential diagnosis
Henoch–Schönlein purpura may present with an atypical manifestation, which can be confused with
Treatment
As of 2017, the optimal way to treat Henoch–Schönlein purpura remains controversial.
Evidence of worsening kidney damage would normally prompt a kidney biopsy. Treatment may be indicated on the basis of the appearance of the biopsy sample; various treatments may be used, ranging from steroids by mouth to a combination of intravenous
There is no good evidence that treating children who have HSP with antiplatelet agent prevents persistent kidney disease.[25][needs update] There is also no evidence that treating children or adults with cyclophosphamide prevents severe kidney disease.[25] Heparin treatment is not justified.[25]
Prognosis
Overall prognosis is good in most patients, with one study showing recovery occurring in 94% and 89% of children and adults, respectively (some having needed treatment).[26] In children under ten, the condition recurs in about a third of all cases, usually within the four months of the initial attack.[6] Recurrence is more common in older children and adults.[8]
Kidney involvement
In adults, kidney involvement progresses to
The findings on renal biopsy correlate with the severity of symptoms: those with asymptomatic hematuria may only have focal mesangial proliferation while those with proteinuria may have marked cellular proliferation or even crescent formation. The number of crescentic glomeruli is an important prognostic factor in determining whether the patient will develop chronic renal disease.[8]
In ESKD, some eventually need
Epidemiology
HSP occurs more often in children than in adults,[26] and usually follows an upper respiratory tract infection. Half of affected patients are below the age of six, and 90% are under ten. It occurs about twice as often in boys as in girls.[8] The incidence of HSP in children is about 20 per 100,000 children per year, making it the most common vasculitis in children.[28]
Cases of HSP may occur anytime throughout the year, but some studies have found that fewer cases occur during the summer months.[29]
History
The disease is named after Eduard Heinrich Henoch (1820–1910), a German pediatrician (nephew of Moritz Heinrich Romberg) and his teacher Johann Lukas Schönlein (1793–1864), who described it in the 1860s. Schönlein associated the purpura and arthritis, and Henoch the purpura and gastrointestinal involvement. The English physician William Heberden (1710–1801) and the dermatologist Robert Willan (1757–1812) had already described the disease in 1802 and 1808, respectively, but the name Heberden–Willan disease has fallen into disuse. William Osler was the first to recognise the underlying allergic mechanism of HSP.[30]
See also
References
- PMID 23045170.
- ^ ISBN 978-1-4160-2999-1.
- PMID 9521029.
- ^ Collins TR (August 2016). "Treatment Challenges, Uncertainty Abound with IgA Vasculitis". The Rheumatologist.
- ^ PMID 9713395. Archived from the originalon 2011-06-06. Retrieved 2007-12-15.
- ^ PMID 10575422.
- ISBN 978-0-07-079454-2.
- ^ S2CID 32647673.
- ^ PMID 16565522.
- PMID 34858429.
- PMID 2420289.
- ^ PMID 10589705.
- PMID 29353097.
- PMID 28654132.
- PMID 1613701.
- PMID 2202310.
- ^ American College of Rheumatology. "1990 criteria for the classification of Henoch–Schönlein purpura". Archived from the original on 27 July 2011. Retrieved 15 December 2007.
- PMID 8129773.
- S2CID 45849349.
- PMID 16322081.
- PMID 18697972.
- ^ PMID 28654132.
- PMID 17974746.
- PMID 36853224.
- ^ PMID 26258874.
- ^ PMID 9153547.
- PMID 22235302.
- S2CID 25018798.
- S2CID 4714914. Archived from the originalon 2020-03-27. Retrieved 2012-08-26.
- Who Named It?
External links
