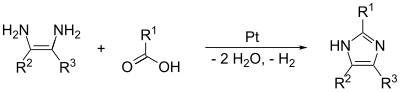Imidazole
| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1H-Imidazole[1] | |||
| Other names
1,3-Diazole
Glyoxaline (archaic) | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 103853 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.005.473 | ||
| EC Number |
| ||
| 1417 | |||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 3263 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H4N2 | |||
| Molar mass | 68.077 g/mol | ||
| Appearance | White or pale yellow solid | ||
| Density | 1.23 g/cm3, solid | ||
| Melting point | 89 to 91 °C (192 to 196 °F; 362 to 364 K) | ||
| Boiling point | 256 °C (493 °F; 529 K) | ||
| 633 g/L | |||
| Acidity (pKa) | 6.95 (for the conjugate acid) [2]
| ||
| UV-vis (λmax) | 206 nm | ||
| Structure | |||
Monoclinic
| |||
| Planar 5-membered ring | |||
| 3.61 D | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Corrosive | ||
| GHS labelling:[4] | |||
  
| |||
| Danger | |||
| H302, H314, H360D | |||
| P263, P270, P280, P301+P310, P305+P351+P338, P308+P313[3] | |||
| Flash point | 146 °C (295 °F; 419 K) | ||
| Safety data sheet (SDS) | External MSDS | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Imidazole (ImH) is an
.Many natural products, especially
When fused to a pyrimidine ring, it forms a purine, which is the most widely occurring nitrogen-containing heterocycle in nature.[10]
The name "imidazole" was coined in 1887 by the German chemist Arthur Rudolf Hantzsch (1857–1935).[11]
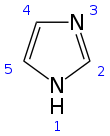
Structure and properties
Imidazole is a planar 5-membered ring, that exists in two equivalent tautomeric forms because hydrogen can be bound to one or another nitrogen atom. Imidazole is a highly polar compound, as evidenced by its electric dipole moment of 3.67 D,[12] and is highly soluble in water. The compound is classified as aromatic due to the presence of a planar ring containing 6 π-electrons (a pair of electrons from the protonated nitrogen atom and one from each of the remaining four atoms of the ring). Some resonance structures of imidazole are shown below:
Amphoterism
Imidazole is
Preparation
Imidazole was first reported in 1858 by the German chemist
In one
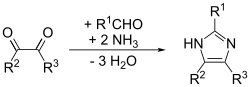
Imidazole can be synthesized by numerous methods besides the
Formation of one bond
The (1,5) or (3,4) bond can be formed by the reaction of an

Formation of two bonds
The (1,2) and (2,3) bonds can be formed by treating a 1,2-diamino
The (1,2) and (3,4) bonds can also be formed from N-substituted α-aminoketones and formamide with heat. The product will be a 1,4-disubstituted imidazole, but here since R1 = R2 = hydrogen, imidazole itself is the product. The yield of this reaction is moderate, but it seems to be the most effective method of making the 1,4 substitution.
Formation of four bonds
This is a general method that is able to give good yields for substituted imidazoles. In essence, it is an adaptation of the Debus method called the
Formation from other heterocycles
Imidazole can be synthesized by the
Imidazole can also be formed in a vapor-phase reaction. The reaction occurs with
- Van Leusen reaction[16]
The
Biological significance and applications
Imidazole is incorporated into many important biological compounds. The most pervasive is the
Imidazole-based histidine compounds play a very important role in intracellular buffering.
Imidazole substituents are found in many pharmaceuticals. Synthetic imidazoles are present in many
A number of substituted imidazoles, including clotrimazole, are selective inhibitors of nitric oxide synthase, which makes them interesting drug targets in inflammation, neurodegenerative diseases and tumors of the nervous system.[18][19] Other biological activities of the imidazole pharmacophore relate to the downregulation of intracellular Ca2+ and K+ fluxes, and interference with translation initiation.[20]
Pharmaceutical derivatives
The substituted imidazole derivatives are valuable in treatment of many systemic
.For comparison, another group of azoles is the triazoles, which includes fluconazole, itraconazole, and voriconazole. The difference between the imidazoles and the triazoles involves the mechanism of inhibition of the cytochrome P450 enzyme. The N3 of the imidazole compound binds to the heme iron atom of ferric cytochrome P450, whereas the N4 of the triazoles bind to the heme group. The triazoles have been shown to have a higher specificity for the cytochrome P450 than imidazoles, thereby making them more potent than the imidazoles.[22]
Some imidazole derivatives show effects on insects, for example
Industrial applications
Imidazole itself has few direct applications. It is instead a precursor to a variety of agrichemicals, including enilconazole, climbazole, clotrimazole, prochloraz, and bifonazole.[24]
Coordination chemistry
Imidazole and its derivatives have high affinity for metal cations. One of the applications of imidazole is in the purification of His-tagged proteins in immobilised metal affinity chromatography (IMAC). Imidazole is used to elute tagged proteins bound to nickel ions attached to the surface of beads in the chromatography column. An excess of imidazole is passed through the column, which displaces the His-tag from nickel coordination, freeing the His-tagged proteins.
Use in biological research
Imidazole is a suitable buffer for pH 6.2 to 7.8,[25]. Pure imidazole has essentially no absorbance at protein relevant wavelengths (280 nm),[26][27] however lower purities of imidazole can give notable absorbance at 280 nm. Imidazole can interfere with the Lowry protein assay.[28]
Salts of imidazole

Salts of imidazole where the imidazole ring is the
Related heterocycles
- Benzimidazole, an analog with a fused benzene ring
- Dihydroimidazole or imidazoline, an analog where the 4,5-double bondis saturated
- Pyrrole, an analog with only one nitrogen atom in position 1
- Oxazole, an analog with the nitrogen atom in position 1 replaced by oxygen
- Thiazole, an analog with the nitrogen atom in position 1 replaced by sulfur
- Pyrazole, an analog with two adjacent nitrogen atoms
- Triazoles, analogs with three nitrogen atoms
Safety
Imidazole has low acute toxicity as indicated by the LD50 of 970 mg/kg (Rat, oral).[24]
See also
References
- ISBN 978-0-85404-182-4.
- .
- ^ "Imidazole". molekula.com. Molekula Group. Archived from the original on 2018-10-19. Retrieved 2018-10-19.
- ^ "Imidazole". pubchem.ncbi.nlm.nih.gov. Archived from the original on 10 May 2023. Retrieved 17 February 2024.
- ISBN 978-0-08-042072-1.
- ISBN 978-0-08-053445-9.
- ISBN 978-94-011-4906-8.
- ISBN 978-0-471-96033-1.
- ISBN 978-0-582-01421-3.
- S2CID 12416667.
- ^ Hantzsch, A. and Weber, J. H. (1887) "Ueber Verbindungen des Thiazols (Pyridins der Thiophenreihe)" Archived 2020-05-30 at the Wayback Machine (On compounds of thiazole (pyridines of the thiophene series), Berichte der deutschen chemischen Gesellschaft, 20 : 3118–3132, see p. 3119. See also: Hantzsch, A. (1888) "Allegemeine Bemerkungen über Azole" Archived 2020-05-30 at the Wayback Machine (General observations about azoles), Annalen der Chemie, 249 : 1–6. Hantzsch proposed a reform of the nomenclature of azole compounds, including a proposal to call the heterocyclic ring C3H3(NH)N "imidazole" ; see pp. 2 and 4.
- S2CID 3522351.
- from the original on 2020-05-30. Retrieved 2016-10-01. From p. 205: "Die gereinigte Substanz stellt das oxalsaure Salz einer Basis dar, die ich mit Glyoxalin bezeichenen werde." (The purified substance constitutes the oxalic salt of a base, which I will designate as "glyoxaline".)
- .
- Arduengo, A. J., "Process for Manufacture of Imidazoles", issued 2001-01-23
- .
- ^ Hochachka, P. W.; Somero, G. N. (2002). Biochemical Adaptation: Mechanisms and Process in Physiological Evolution. New York: Oxford University Press.
- (PDF) from the original on 2023-03-06. Retrieved 2018-07-24.
- PMID 7518297.
- PMID 16121977.
- ISBN 9780781765619.
- ISBN 978-0-8138-2061-3.
- S2CID 135799368.
- ^ ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - OCLC 11865673.
- ^ "1H-Imidazole". Archived from the original on 25 April 2021. Retrieved 13 May 2021.
- ^ "Optimizing Purification of Histidine-Tagged Proteins". Archived from the original on 13 May 2021. Retrieved 13 May 2021.
- PMID 8851751.
- PMID 22409592.