Interferon beta-1b
| Clinical data | |
|---|---|
| Trade names | Betaseron, Actoferon, Extavia |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601151 |
| License data |
|
Subcutaneous | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
ECHA InfoCard | 100.207.515 |
| Chemical and physical data | |
| Formula | C908H1408N246O253S6 |
| Molar mass | 20011.08 g·mol−1 |
| | |
Interferon beta-1b is a
Mechanism of action
Interferon beta balances the expression of pro- and anti-inflammatory agents in the brain, and reduces the number of inflammatory cells that cross the
Side effects
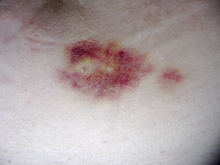
Interferon beta-1b is available only in injectable forms, and can cause skin reactions at the injection site that may include cutaneous necrosis. Skin reactions vary greatly in their clinical presentation.[2] They usually appear within the first month of treatment albeit their frequence and importance diminish after six months of treatment.[2] Skin reactions are more prevalent in women.[2] Mild skin reactions usually do not impede treatment whereas necroses appear in around 5% of patients and lead to the discontinuation of the therapy.[2] Also over time, a visible dent at the injection site due to the local destruction of fat tissue, known as lipoatrophy, may develop.
The injection-site reactions can be mitigated by rotating injection sites or by using one of the medications that requires less frequent injections. Side effects are often onerous enough that many patients ultimately discontinue taking Interferons (or glatiramer acetate, a comparable disease-modifying therapies requiring regular injections).
Efficacy
Clinically isolated syndrome
The earliest clinical presentation of relapsing-remitting multiple sclerosis is the clinically isolated syndrome (CIS), that is, a single attack of a single symptom. During a CIS, there is a subacute attack suggestive of
Relapsing-remitting MS
Medications are modestly effective at decreasing the number of attacks in relapsing-remitting multiple sclerosis
While more studies of the long-term effects of the drugs are needed,[4][11] some data on the effects of interferons indicate that early-initiated long-term therapy is safe and it is related to better outcomes.[11] More recent data suggest that interferon betas does not hasten disability.[12]
Interferon-β exacerbates Th17-mediated inflammatory disease.[13]
Commercial formulations
Betaferon/Betaseron is marketed today by
References
External links
- "Interferon beta-1b". Drug Information Portal. U.S. National Library of Medicine.
